Clear Sky Science · en
Cytotoxic CD4+ T cells: origin, biological functions, diseases and therapeutic targets
When Helper Cells Turn into Killers
The immune system is usually described in simple roles: some cells spot trouble, others do the actual fighting. This paper focuses on a surprising group that blurs that line—cytotoxic CD4 T cells. Long known mainly as “helper” cells that coordinate defenses, certain CD4 T cells can themselves become armed killers. Understanding how these shape-shifting cells arise, what they do, and how they contribute to diseases such as cancer, viral infections, autoimmune disorders, and heart disease could open new ways to both boost immunity and calm harmful inflammation. 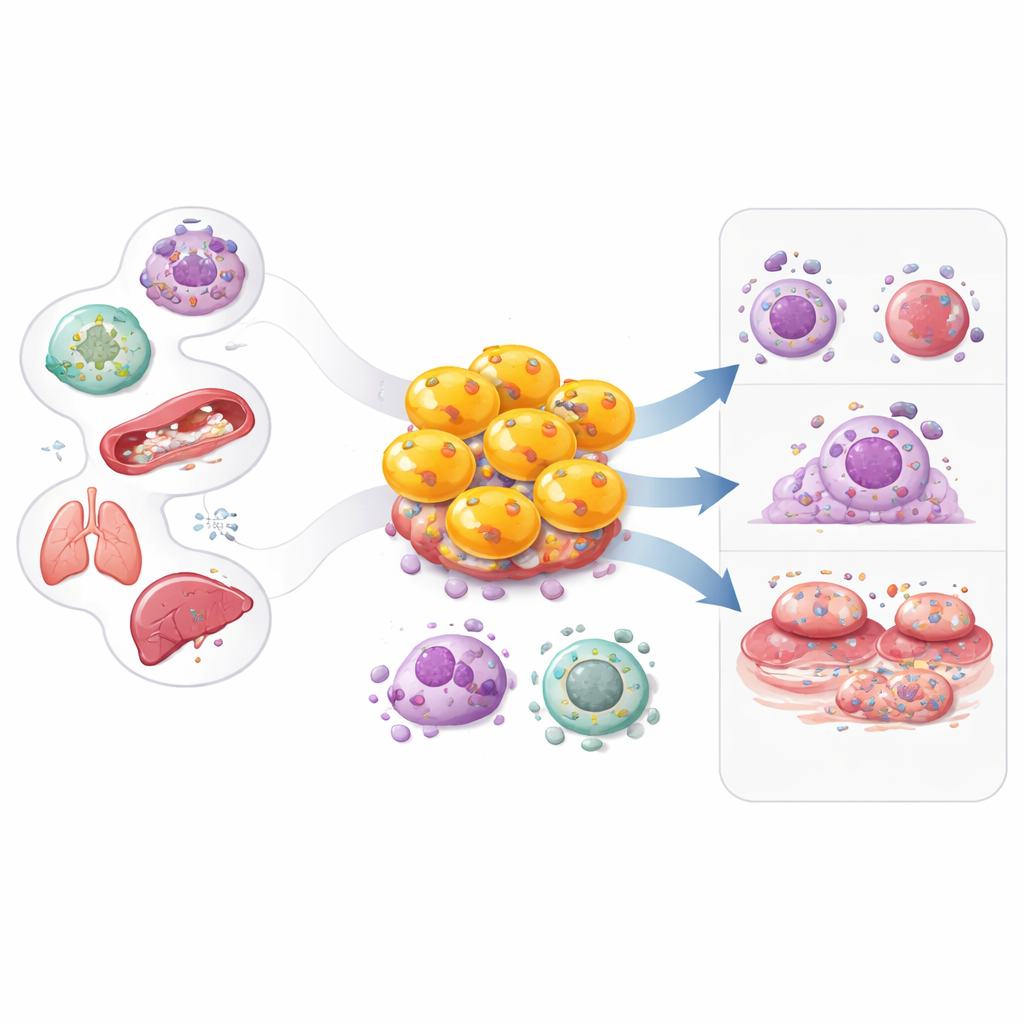
How a Flexible Defender Emerged
Early work in the late 1970s and 1980s showed that a fraction of CD4 T cells could directly kill other cells displaying the right immune flags, much like classic CD8 “killer” T cells. At first, many scientists dismissed these findings as artifacts of lab culture. Over the following decades, animal models of influenza and studies in people with rheumatoid arthritis, heart disease, chronic viral infections, and cancer confirmed that cytotoxic CD4 T cells are real and clinically important. Modern tools such as flow cytometry and single-cell RNA sequencing have since mapped their diversity, revealing that these cells expand in chronic infections, tumors, aging, and several inflammatory diseases, where they can persist for long periods.
Where These Killer Helpers Come From
The review explains that cytotoxic CD4 T cells likely arise in two main ways. One idea is that they partly resemble natural killer cells because they share many of the same surface receptors and cytotoxic molecules, hinting at a related developmental path. However, accumulating evidence favors another view: most of these cells start as ordinary helper CD4 T cells—especially Th1-like cells—that, under chronic antigen exposure and strong inflammatory signals, “retrain” into a terminally differentiated, cytotoxic state. This shift is controlled by a network of external cues (such as interleukins 2, 7, and 15) and internal switches (including Runx3, T-bet, Eomes, Blimp-1, and Hobit) that together turn on genes for perforin, granzymes, and death-inducing ligands.
How They Kill and Shape Inflammation
Functionally, cytotoxic CD4 T cells operate through two main attack routes. In the granule route, they form a tight contact with a target cell and release packets containing perforin and granzymes, which punch holes and trigger programmed cell death from within. In the death-ligand route, they display molecules such as Fas ligand and TRAIL on their surface, engaging matching receptors on target cells to launch an internal suicide program. Beyond direct killing, these cells pour out inflammatory messengers like interferon-gamma, tumor necrosis factor, and growth factors that recruit and activate other immune cells, reshape blood vessels, and alter local tissue behavior. Unlike classic helpers, they are often resistant to normal “brakes” such as regulatory T cells and are less prone to apoptosis, allowing them to accumulate in chronic disease. 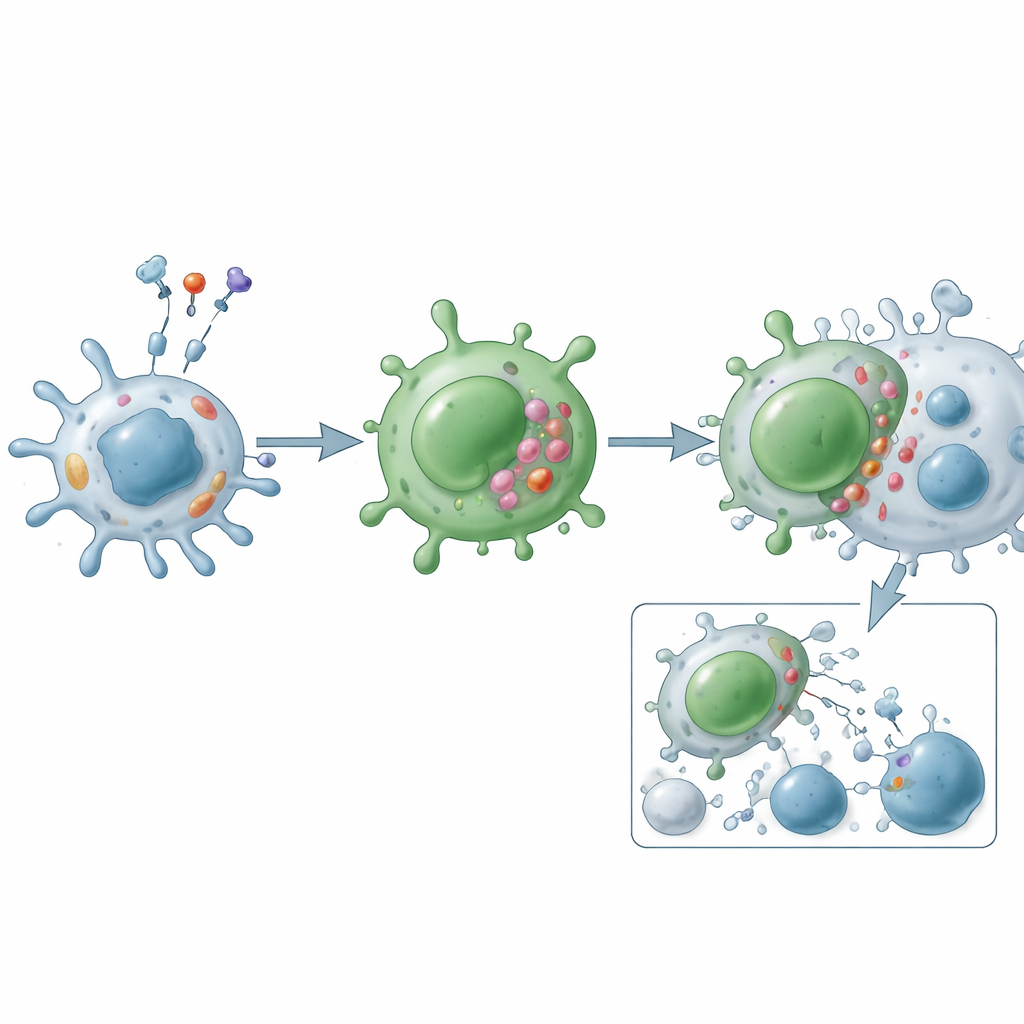
Friends in Infection and Cancer, Foes in Autoimmunity and Heart Disease
This double-edged behavior shows up across many conditions. In chronic hepatitis B, HIV, and COVID-19, cytotoxic CD4 T cells help control viruses when CD8 killers are exhausted, and their presence can track with better control or recovery. In solid tumors and blood cancers, they can directly recognize tumor cells that display the right immune markers or stress signals and may improve responses to immunotherapies such as checkpoint inhibitors and CAR-T cell treatments. At the same time, these same mechanisms can drive harm: in lupus, rheumatoid arthritis, inflammatory bowel disease, coronary artery disease, atrial fibrillation, giant cell arteritis, and transplant rejection, expanded cytotoxic CD4 T cells infiltrate tissues, damage blood vessels or organs, and amplify long-lasting inflammation, often correlating with worse prognosis.
Turning a Risky Tool into a Therapy
Because cytotoxic CD4 T cells can be either protective or destructive, the paper highlights emerging therapeutic strategies that aim to fine-tune, rather than simply erase, this population. One approach is to dampen their harmful activity using selective inhibitors that target costimulatory receptors (such as OX40 or 4-1BB), inflammatory cytokines, ion channels, or survival pathways. Another is to harness their killing power in adoptive cell therapies, including gene-edited T cells and CAR-T cells, where CD4 cytotoxic cells often show better persistence than CD8-only products. A third strategy proposes redirecting long-lived, virus-specific CD4 killers toward tumors using vaccines or pathogen-derived peptides presented by cancer cells. Together, these lines of work position cytotoxic CD4 T cells not just as a biological curiosity, but as a central lever for future treatments that could more precisely boost defense against infections and cancer while reducing tissue-damaging inflammation.
A New Lever for Future Immune Therapies
For non-specialists, the key message is that “helper” CD4 T cells are far more versatile than once thought: some can switch into fully armed killers that protect us from persistent infections and cancer, but also help drive autoimmunity, vascular damage, and transplant rejection. The review argues that learning how to track these cells, predict when they will help or harm, and adjust their behavior with drugs or engineered therapies could reshape how we treat a wide range of chronic diseases—from hepatitis and HIV to arthritis, colitis, heart disease, and leukemia. Rather than simply turning the immune system up or down, future therapies may increasingly work by steering powerful cell types like cytotoxic CD4 T cells toward beneficial targets and away from our own tissues.
Citation: Lai, L., Ran, S., Li, Y. et al. Cytotoxic CD4+ T cells: origin, biological functions, diseases and therapeutic targets. Sig Transduct Target Ther 11, 85 (2026). https://doi.org/10.1038/s41392-025-02533-z
Keywords: cytotoxic CD4 T cells, immune cell plasticity, chronic inflammation, cancer immunotherapy, autoimmune disease