Clear Sky Science · en
The oncogenic control of nucleotide synthesis
Why Cancer Cells Care About Building Blocks
Cancer is often described as cells that won’t stop dividing. But to copy themselves, these cells need a constant supply of tiny building blocks called nucleotides, which make up DNA and RNA and also help power many cell processes. This review explains how cancer-causing genes rewire cell chemistry to keep nucleotide production high, how tumors switch between different ways of getting these molecules, and how doctors can exploit these tricks with old and new drugs.
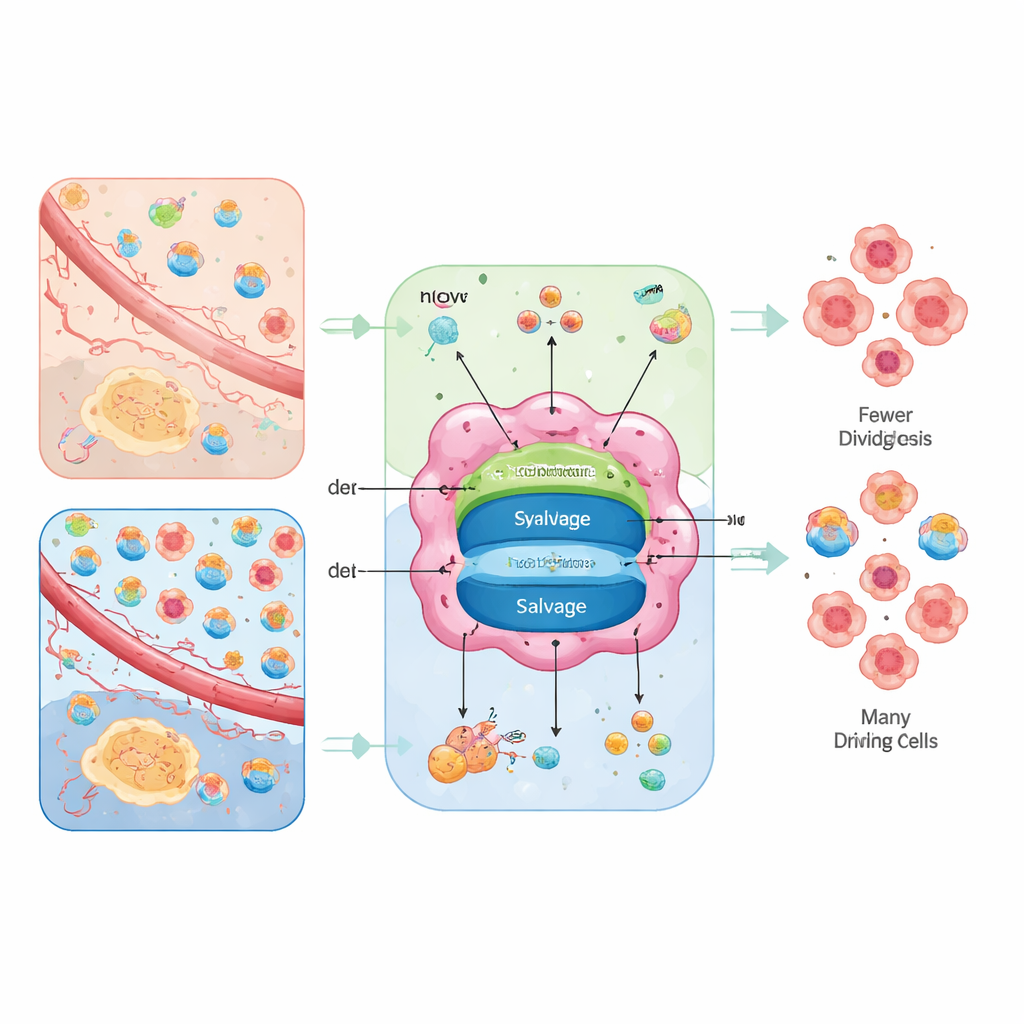
Two Roads to the Same Building Blocks
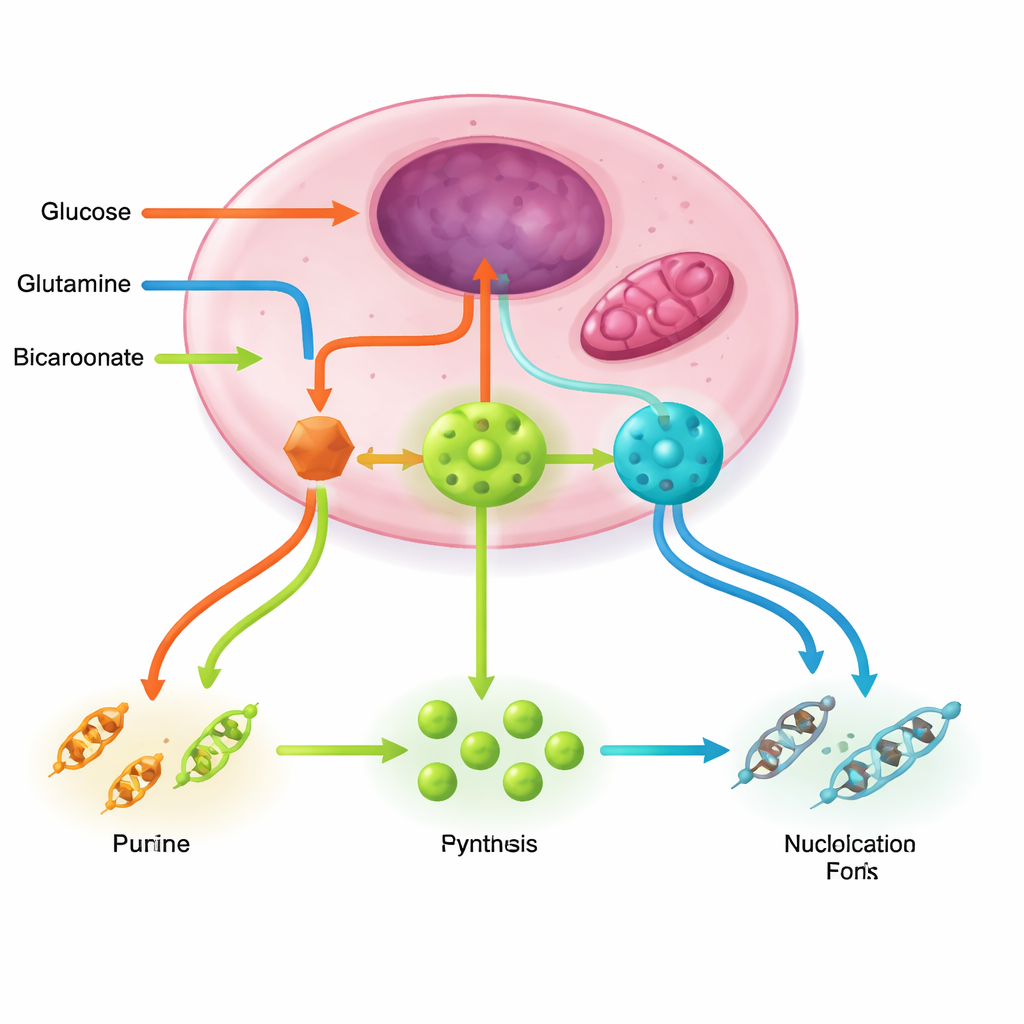
Cells can make nucleotides from scratch or recycle them. The "from scratch" route, called de novo synthesis, assembles rings from basic nutrients such as glucose, amino acids, carbon dioxide, and bicarbonate, using energy-hungry, multi-step reactions. The recycling route, called salvage, takes broken-down bases and nucleosides from outside or from cell waste and snaps them back into usable nucleotides at a much lower energy cost. Tumors use both options, but the balance between them depends on their internal power supply and what the surrounding tissue provides.
How the Tumor Environment Steers the Choice
The authors frame nucleotide supply as an "environment-shaped routing" problem. In poorly fed regions where amino acids are present but free nucleosides are scarce, cancer cells lean heavily on de novo pathways and become vulnerable to drugs that block these steps. In better-perfused regions, where the blood delivers plenty of nucleosides or where mitochondria are weak, tumors can rely more on salvage, becoming sensitive instead to transporter blockers that prevent nucleoside uptake. When both options are open, tumors show metabolic flexibility and can dodge single drugs; when both are closed, cells stall in DNA replication, accumulate damage, and may die or switch into more mature, less aggressive states.
Hidden Support Lines Feeding Nucleotide Supply
Keeping nucleotide factories running requires several supporting circuits. The pentose phosphate pathway provides the sugar backbone and reducing power; glutamine and the energy-producing cycle in mitochondria furnish nitrogen and aspartate; serine, glycine, and folate chemistry donate one-carbon units that finish the rings; bicarbonate brings in essential carbons. Cancer drivers such as PI3K–AKT–mTORC1, RAS–MAPK, and MYC dial up these pathways together, boost key enzymes, and even assemble multi-enzyme clusters that channel unstable intermediates efficiently. In contrast, stress sensors like LKB1–AMPK and p53 normally act as brakes, reducing nucleotide output when energy or DNA integrity is threatened—brakes that many tumors disable.
Old Chemotherapy, New Metabolic Logic
Because fast-growing cells depend so strongly on nucleotides, some of the earliest chemotherapies targeted this weakness. Classic drugs such as methotrexate, 5‑fluorouracil, cytarabine, gemcitabine, and various purine analogues either block folate use, inhibit specific enzymes, or masquerade as faulty building blocks that poison DNA and RNA. The review revisits these medicines through the lens of the routing model, explaining why side effects and resistance are common: noncancerous tissues also need nucleotides, and tumors can often reroute flux through salvage or parallel pathways when one node is blocked.
Next-Generation Strategies and Open Questions
Newer approaches aim to be more selective by striking at the most cancer-biased pieces of the network. Experimental drugs are being developed against mitochondrial one-carbon enzymes such as MTHFD2, the pyrimidine enzyme DHODH, and purine enzymes like IMPDH and PAICS, often in combinations that also block salvage transporters. The authors propose practical markers—such as transporter levels, blood-vessel density, mitochondrial fitness, and expression of specific metabolic enzymes—to decide which tumors rely more on de novo or salvage and to match patients with pathway-targeted therapies. Looking ahead, they highlight key questions, including how these routes shift over space and time inside real tumors, how surrounding immune and stromal cells shape nucleotide availability, and how to design tolerable drug schedules that exploit replication stress without overwhelming normal tissues. Together, this work argues that understanding and targeting nucleotide metabolism could turn a basic requirement of cell division into a powerful, personalized lever against cancer.
Citation: Vidal-Cruchez, O., Ben-Sahra, I. The oncogenic control of nucleotide synthesis. Oncogenesis 15, 17 (2026). https://doi.org/10.1038/s41389-026-00608-2
Keywords: nucleotide metabolism, cancer metabolism, de novo synthesis, salvage pathways, metabolic therapies