Clear Sky Science · en
Teriflunomide modulates the PD-1/PD-L1 axis and enhances antitumor immunity in colorectal cancer
Turning a Multiple Sclerosis Drug Against Colon Cancer
Many modern cancer treatments aim to wake up the immune system so it can better recognize and attack tumors. Yet for most people with colorectal (colon) cancer, current immune-based therapies offer limited benefit. This study explores an unexpected ally: teriflunomide, a long-approved pill for multiple sclerosis, and shows how it can rewire a key immune “brake” to help the body’s own killer T cells shrink colon tumors in experimental models. 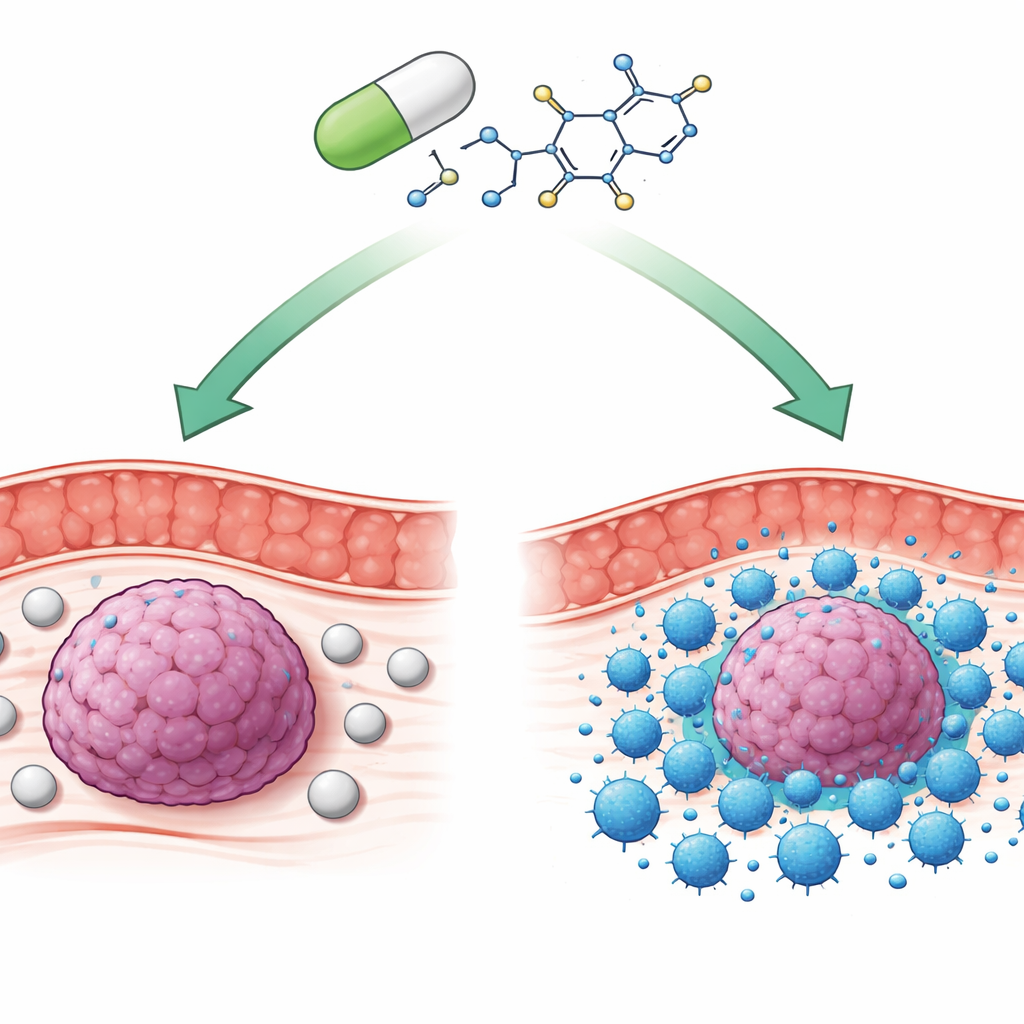
The Body’s Brakes on Immune Attack
Our immune system relies on a balance of gas pedals and brakes. One major brake is a molecular handshake between two proteins called PD-1, on immune T cells, and PD-L1, often displayed on tumor cells. When they connect, T cells become exhausted and stop attacking, allowing cancers to grow unnoticed. Existing drugs that block this handshake—largely antibody infusions given in the clinic—have transformed treatment for some cancers. But in most colorectal cancers, especially the common “microsatellite-stable” type, these drugs work poorly, and many patients either never respond or eventually become resistant.
An Old Drug with a New Mission
Teriflunomide is an oral drug used for years to treat multiple sclerosis by calming overactive immune cells. Because of its known safety in people, it is an attractive candidate for repurposing if it can be shown to help the immune system in cancer rather than suppress it. The researchers asked whether teriflunomide could act directly on the PD-1/PD-L1 system in colorectal cancer cells and T cells. Using several human colon cancer cell lines grown in the lab, they found that teriflunomide reduced the amount of PD-L1 protein on tumor cells without killing them outright at the doses tested. Interestingly, this effect did not come from turning off the PD-L1 gene; instead, the drug sped up the breakdown of the PD-L1 protein inside cellular “recycling centers” called lysosomes.
Blocking the Immune Handshake and Powering Up T Cells
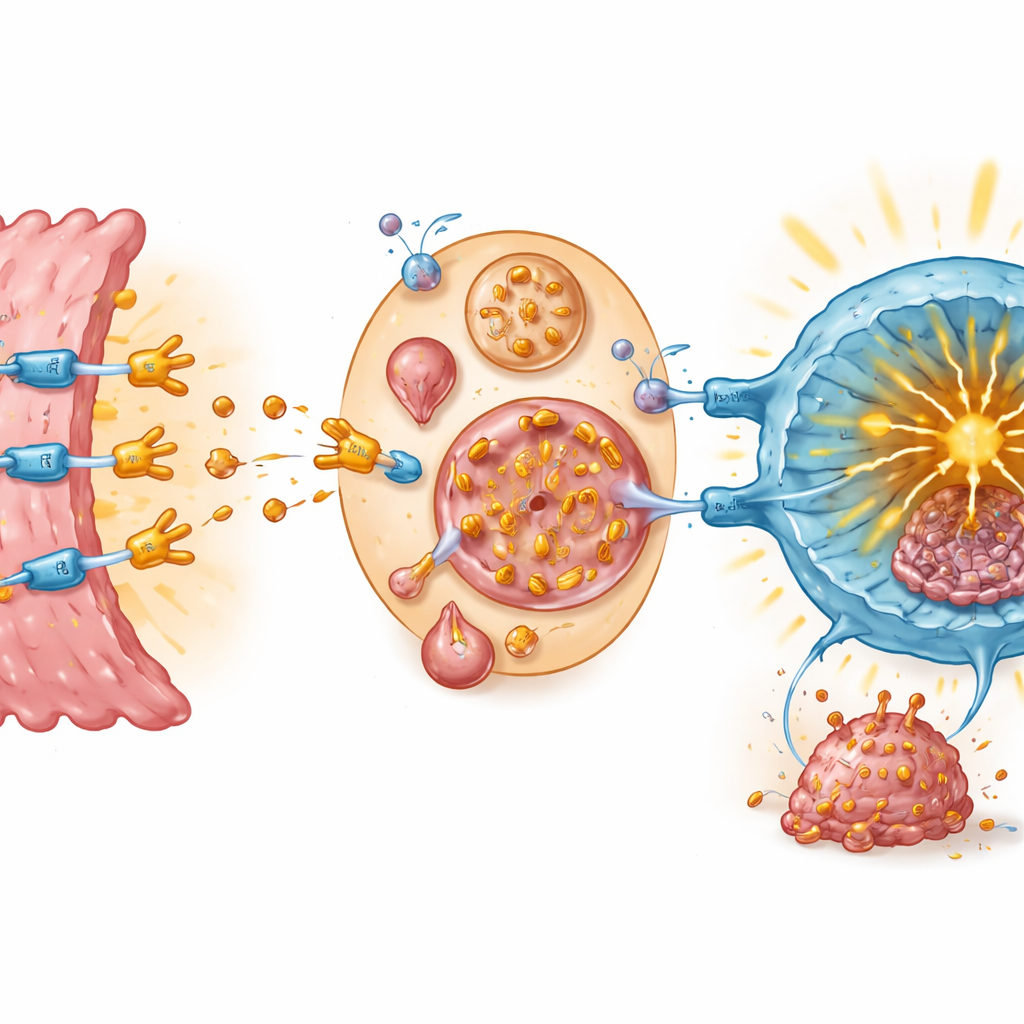
To probe how teriflunomide influenced the immune brake directly, the team studied its impact on the binding between PD-1 and PD-L1. Biochemical tests and computer modeling indicated that teriflunomide can attach to PD-1 itself, nestling into the very region PD-L1 normally grabs. This direct contact blocked the PD-1/PD-L1 interaction at very low drug concentrations. In engineered cell systems that mimic the immune checkpoint, adding teriflunomide restored signaling inside T cells, a sign that the brake was being released. When tumor cells and T cells were grown together, teriflunomide both lowered PD-L1 levels on cancer cells and boosted markers of T cell activation and killing ability.
From Petri Dish to Tumor-Bearing Mice
What This Could Mean for Future Cancer Care
To a non-specialist, the key message is that teriflunomide appears to work as a double-acting checkpoint blocker: it latches onto PD-1 on T cells to prevent a disabling handshake, and at the same time pushes tumor cells to dispose of their PD-L1 shield. Together, these actions let killer T cells flood into tumors and attack them more effectively in colorectal cancer models. Because teriflunomide is already approved and its safety profile is well mapped, it could move into cancer trials more quickly than an entirely new drug. While human studies are still needed, this work suggests that a familiar multiple sclerosis pill might one day become part of combination strategies to help people with colon cancer who do not benefit from current immunotherapies.
Citation: Han, J.H., Lee, EJ., Park, YH. et al. Teriflunomide modulates the PD-1/PD-L1 axis and enhances antitumor immunity in colorectal cancer. Oncogenesis 15, 15 (2026). https://doi.org/10.1038/s41389-026-00607-3
Keywords: colorectal cancer immunotherapy, PD-1 PD-L1 checkpoint, teriflunomide, CD8 T cells, drug repurposing