Clear Sky Science · en
Inhibition of ZBTB7B-mediated ADPGK transcription by NEDD4 impedes glycolysis and progression of lung adenocarcinoma
Why this research matters
Lung adenocarcinoma is the most common form of lung cancer and is often found only after it has quietly advanced, when treatments work less well. One reason these tumors are so hard to control is that cancer cells rewire how they burn sugar, giving them extra fuel to grow, spread, and resist drugs. This study uncovers a previously unknown molecular "thermostat" that turns this sugar-burning system up or down in lung adenocarcinoma cells, pointing to new ways to starve tumors without harming the rest of the body.
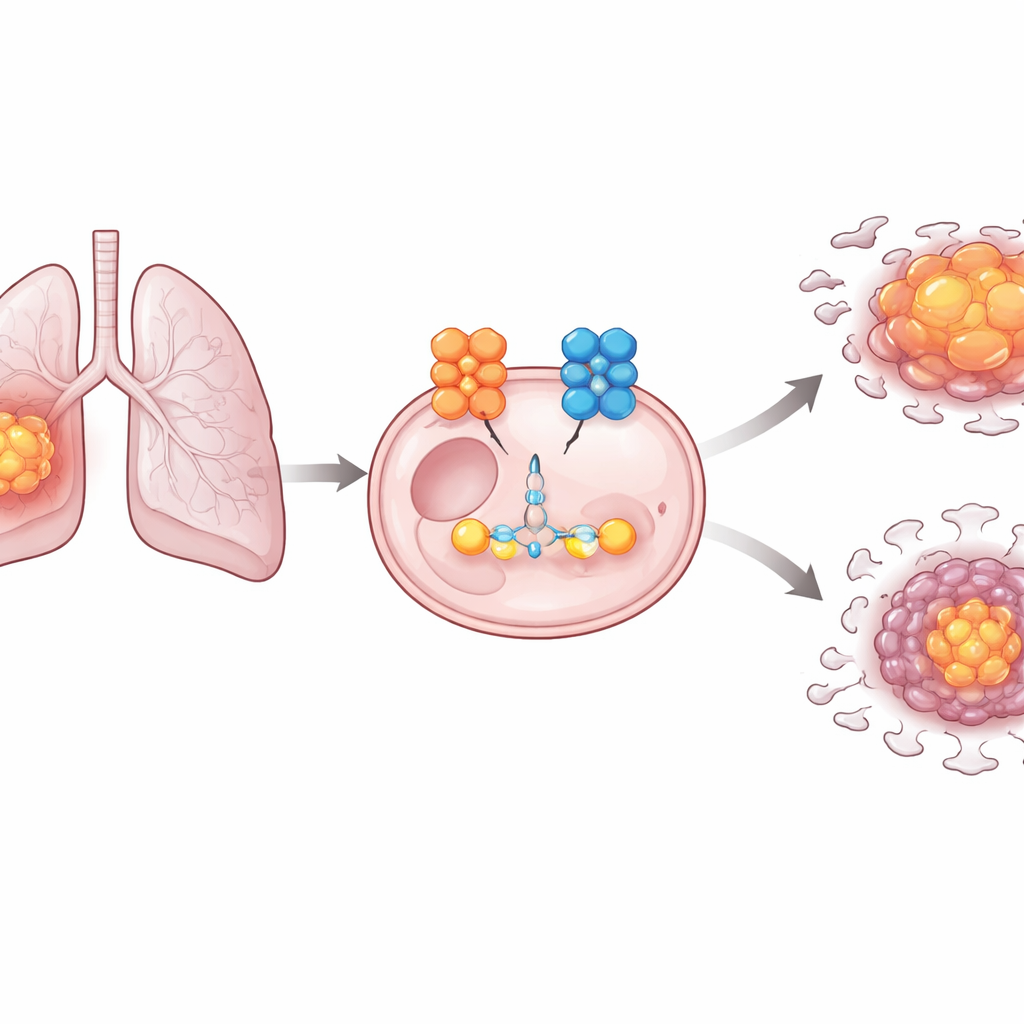
A cancer that lives on sugar
Cancer cells often depend heavily on glycolysis, a rapid but inefficient way of turning glucose into energy and building blocks, even when oxygen is plentiful. This metabolic shift helps tumors grow quickly and survive harsh conditions, such as poor blood supply or treatment stress. In lung adenocarcinoma, many of the individual enzymes that carry out glycolysis are known, but the switches that control when they are made and how strongly they are used have been less clear. The authors set out to identify key regulators that link gene control, protein stability, and altered sugar use in this disease.
An unexpected promoter of tumor growth
The researchers focused on a protein called ZBTB7B, previously known for roles in immune cells and fat metabolism. By analyzing large patient datasets and tumor samples, they found that ZBTB7B protein levels are consistently higher in lung adenocarcinoma tissue and cell lines than in normal lung cells, and that patients whose tumors carry more of this protein tend to do worse. When ZBTB7B was reduced in lung cancer cells grown in the lab, the cells divided more slowly, formed fewer colonies, and migrated less, all signs of weaker tumor behavior. These same cells also consumed less glucose, released less lactate, and produced less ATP, revealing that ZBTB7B helps drive the cancer’s sugar-hungry lifestyle.
A special sugar enzyme in the spotlight
To understand how ZBTB7B boosts glycolysis, the team combined genome-wide binding data with gene activity maps and homed in on a relatively unusual enzyme called ADPGK. Unlike typical hexokinases, which use ATP to start glycolysis, ADPGK can use ADP, allowing cells to keep processing glucose even when energy is low. The authors showed that ZBTB7B binds directly to a short region of the ADPGK gene’s control sequence and switches it on, increasing both its RNA and protein levels. When ADPGK was silenced, the growth, movement, and heightened glycolysis triggered by extra ZBTB7B largely disappeared, and restoring ADPGK revived these tumor-friendly traits. This indicates that ZBTB7B pushes lung cancer cells toward a high-glycolysis, high-growth state mainly through ADPGK.
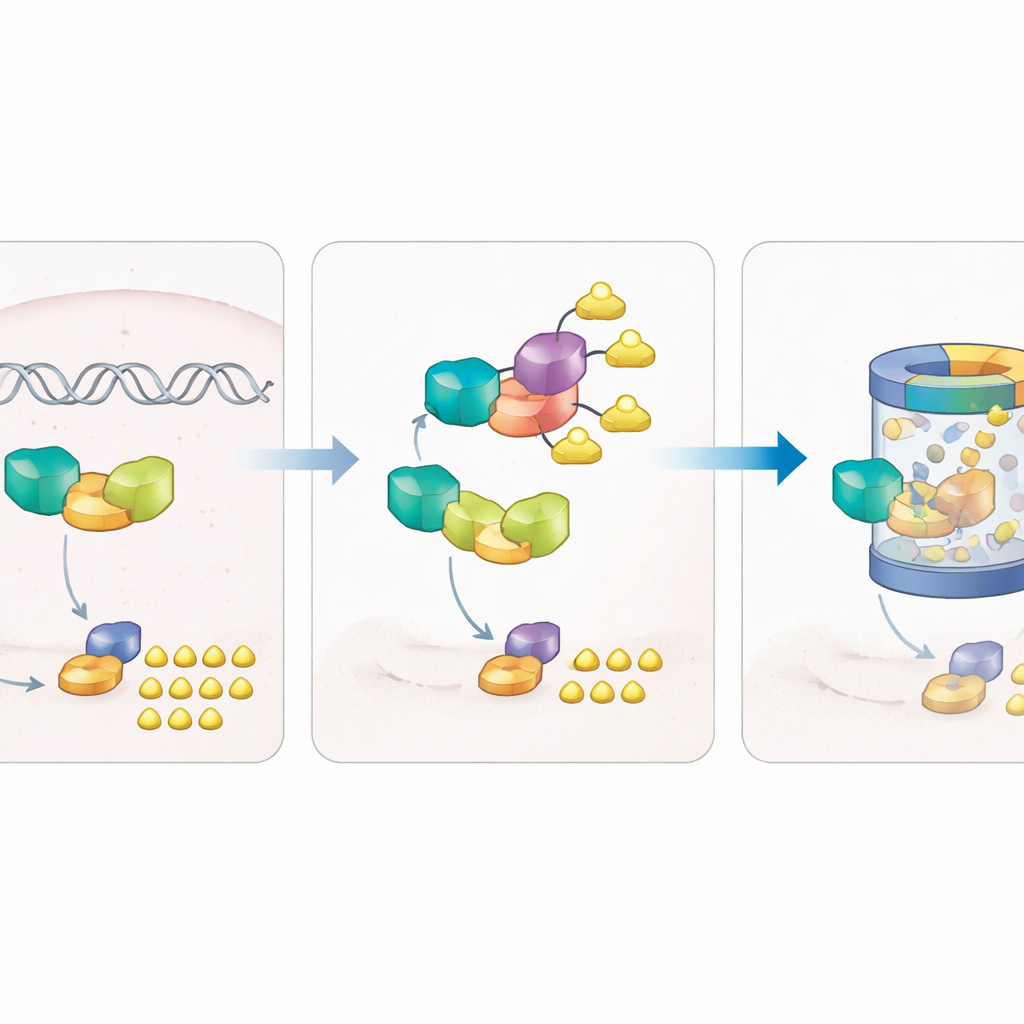
A built-in brake that targets the sugar switch
The puzzle deepened when the authors noticed that ZBTB7B RNA was not elevated in tumors, even though its protein was. This pointed to control after the RNA is made. They discovered that another protein, the E3 ligase NEDD4, physically binds to ZBTB7B and decorates it with ubiquitin tags at a specific site, marking it for destruction by the cell’s protein-recycling machinery. When NEDD4 was increased, ZBTB7B became less stable, ADPGK levels dropped, and glycolysis slowed; reducing NEDD4 had the opposite effect. A single change at the tagged site on ZBTB7B protected it from NEDD4 and prevented its breakdown. In mouse models, lung cancer cells engineered to produce more NEDD4 formed smaller, less glycolytic tumors and fewer lung metastases, but reintroducing ZBTB7B reversed this benefit.
What this means for future treatments
Together, these findings reveal a new control circuit in lung adenocarcinoma: ZBTB7B turns on ADPGK to fuel glycolysis and tumor progression, while NEDD4 acts as a natural brake by flagging ZBTB7B for disposal. For non-specialists, this can be thought of as a gas pedal (ZBTB7B–ADPGK) and a foot on the brake (NEDD4) that together determine how hard the tumor engine runs on sugar. By designing drugs that weaken the gas pedal—by blocking ZBTB7B or ADPGK—or strengthen the brake by enhancing NEDD4’s action on ZBTB7B, it may be possible to cut off a major energy supply to lung adenocarcinoma cells. While such therapies are still in the conceptual stage, this work maps out concrete molecular targets for future metabolism-focused cancer treatments.
Citation: Liu, FP., Huang, T., Yang, HY. et al. Inhibition of ZBTB7B-mediated ADPGK transcription by NEDD4 impedes glycolysis and progression of lung adenocarcinoma. Oncogenesis 15, 14 (2026). https://doi.org/10.1038/s41389-026-00605-5
Keywords: lung adenocarcinoma, cancer metabolism, glycolysis, ubiquitin ligase NEDD4, transcription factor ZBTB7B