Clear Sky Science · en
Transcriptomic profiling of co-cultured cancer-host cells identifies hypoxia as a driver of the skeletal muscle cell’s anti-proliferative effect on cancer cells
Why some tissues resist cancer spread
Cancer usually becomes deadly when tumor cells escape their original site and settle in distant organs. Curiously, some tissues, like the lungs, are frequent landing spots, while others, such as skeletal muscle, are almost never colonized despite making up a large share of body weight. This study asks a simple but important question for patients and doctors alike: what is it about muscle that makes it such poor soil for cancer cells to grow in, and could understanding this natural defense reshape how we think about treating metastasis?
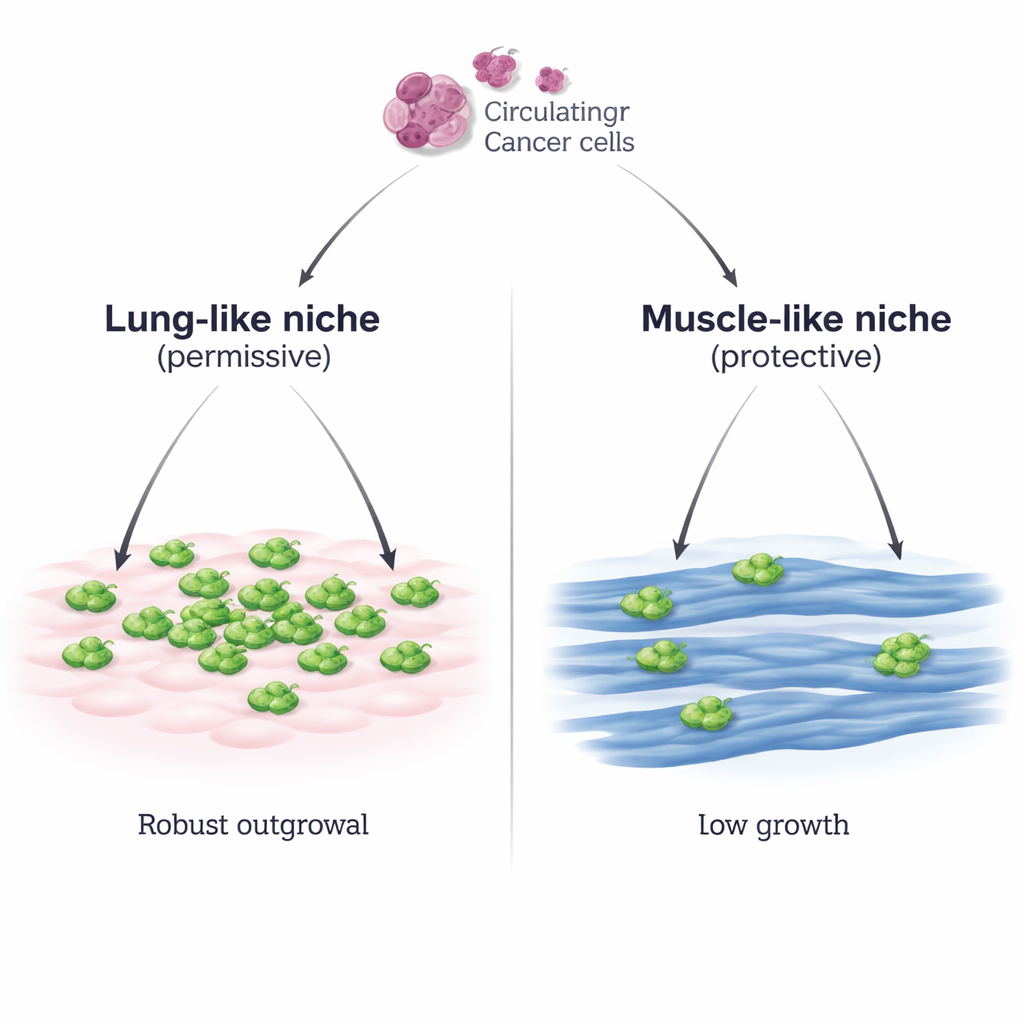
Two neighborhoods for traveling tumor cells
The researchers built a controlled laboratory system that mimics two different neighborhoods a breast cancer cell might encounter: a lung-like environment made of lung connective tissue cells (MLg), and a muscle-like environment made of mature skeletal muscle fibers (C2C12 myotubes). They placed mouse breast cancer cells on top of each layer and watched what happened. On the lung-like cells, cancer cells attached well and then multiplied rapidly, forming dense patches. On the muscle cells, they attached just as well, but their growth remained sparse and slow, even when the starting number of cancer cells was greatly increased or when a second breast cancer cell line was used. A separate skeletal muscle line (Sol8) showed the same ability to hold cancer growth back, underscoring that this is a general property of muscle cells rather than a quirk of one cell type.
How cancer and host cells rewire their activity
To understand what was happening inside the cells, the team isolated cancer cells and host cells from these mixed cultures and sequenced their RNA, a readout of which genes are turned on or off. Cancer cells sitting on muscle fibers underwent far more extensive gene activity changes than those growing on lung cells, suggesting that the muscle environment forces them to adapt strongly. In the muscle setting, cancer cells switched on programs linked to muscle-like features, energy processing, and, surprisingly, low-oxygen (hypoxia) responses. At the same time, the usual gene programs that drive rapid cell division were dialed down. By contrast, cancer cells on lung cells kept a strongly growth-focused profile and needed much less reprogramming, consistent with the idea that the lung-like niche is permissive and easy for tumor cells to exploit.
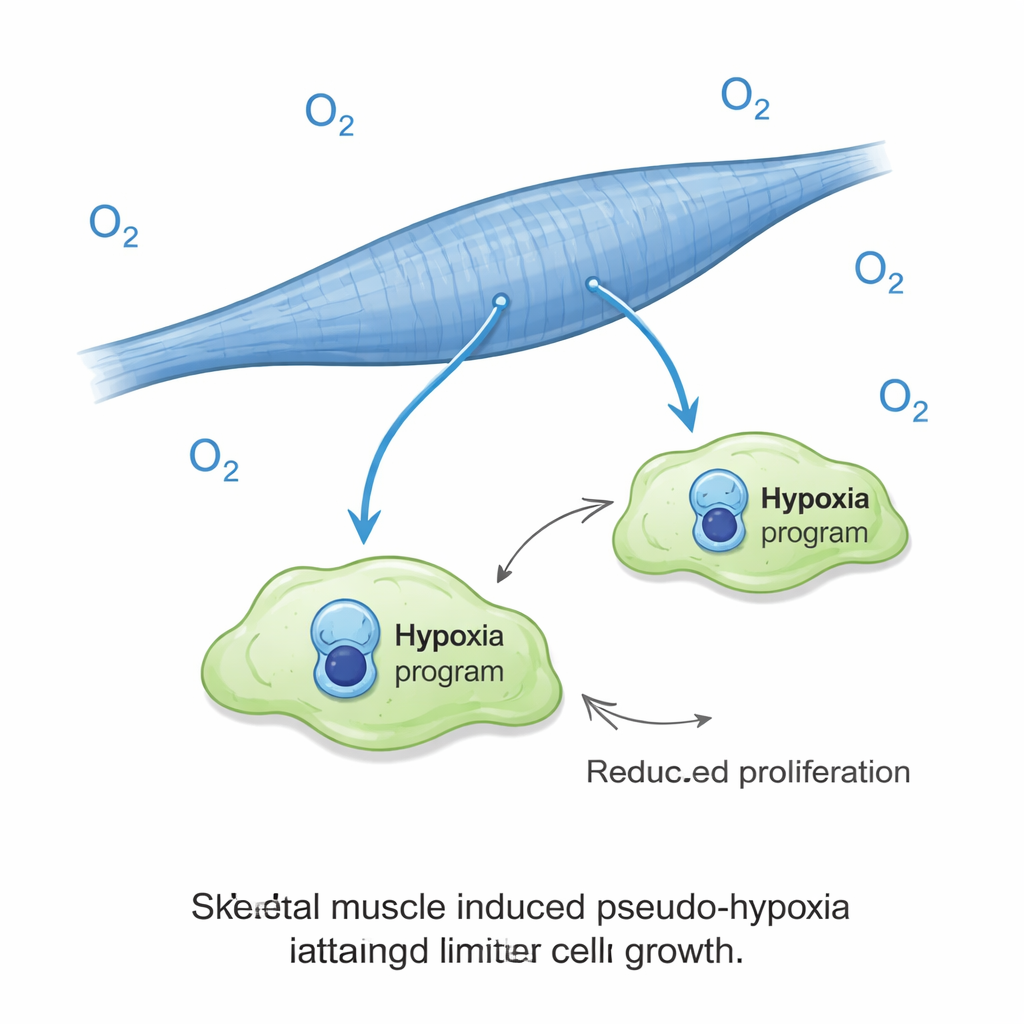
A surprising role for low-oxygen signals
One of the most striking findings was that cancer cells on muscle fibers turned on a hypoxia-like gene signature even though they were grown under normal oxygen levels. In most tumors, hypoxia is a bad sign, usually linked to aggressive behavior and poor outcomes. Here, it was linked to the opposite: poor growth. The scientists then exposed the cultures to truly low oxygen and found that this nearly wiped out cancer cell expansion on muscle, while actually boosting cancer growth in the lung-like cultures. In other words, the same cue that usually helps tumors thrive became a brake when cancer cells were in a muscle environment. This oxygen effect did not appear to be explained by simple changes in lactate, acidity, or sugar availability in the culture medium.
More than just secreted factors
The team also tested whether simple soluble substances released by muscle cells could explain their protective effect. Adding extra lactate, altering the acidity, changing glucose levels, or growing cancer cells in medium that had previously bathed muscle cells did not reproduce the strong suppression seen in direct contact co-cultures. This suggests that close physical contact, or very short-range signals at the cell surface, are key to how muscle cells push cancer cells into a low-growth, possibly dormant, state. Interestingly, muscle cells themselves remained relatively stable in their gene activity when facing cancer, whereas lung cells were more easily reprogrammed, highlighting muscle as a robust, “metastasis-resistant” tissue.
What this means for patients and therapies
Overall, the study reveals that skeletal muscle acts as a hostile habitat that forces cancer cells into a hypoxia-like, low-proliferation mode, even when plenty of oxygen is available. This helps explain why muscle is such a rare site of metastasis. It also offers a cautionary message for drug development: therapies designed to block hypoxia signaling across the body might unintentionally weaken this natural muscle-based defense, making it easier for cancer cells to grow where they normally would not. Understanding and preserving, or even mimicking, the protective features of muscle tissue could open new avenues for preventing or controlling metastatic disease.
Citation: Aunan, A., Claeyssen, C., Abdelhalim, M. et al. Transcriptomic profiling of co-cultured cancer-host cells identifies hypoxia as a driver of the skeletal muscle cell’s anti-proliferative effect on cancer cells. Oncogenesis 15, 7 (2026). https://doi.org/10.1038/s41389-026-00601-9
Keywords: cancer metastasis, skeletal muscle, hypoxia, tumor microenvironment, cancer dormancy