Clear Sky Science · en
Fatty acid uptake mediated by FABP4 promotes the formation of CD8+T cell senescence through lipid peroxidation in the adipocyte-rich microenvironment of Ovarian Cancer
Why Fat and Immune Cells Matter in Ovarian Cancer
Ovarian cancer often spreads into the abdominal cavity, where it encounters large deposits of body fat. This fatty environment is not just a passive backdrop—it actively shapes how immune cells behave. In particular, it can weaken the very CD8 “killer” T cells that are supposed to attack tumors. This study explores how fat-derived substances push these T cells into an aged, worn-out state, and how blocking a key fat-transport protein might help restore their cancer-fighting power.
Fat-Rich Surroundings That Age Immune Defenders
Doctors have long noticed that many women with advanced ovarian cancer accumulate fluid in the abdomen and have tumors nestled in fat-rich tissues called the omentum. By analyzing single-cell RNA data from patients and examining tumor samples under the microscope, the researchers found that CD8 T cells near these fat deposits were more likely to show hallmarks of cellular aging, or senescence. These “aged” T cells build up waste pigments, stop dividing, and produce inflammatory signals instead of killing cancer cells. In both human samples and mouse models, CD8 T cells taken from fat- and fluid-rich tumor areas were noticeably more senescent than those from lymph nodes or spleen, linking the adipocyte-rich tumor microenvironment directly to T cell aging.
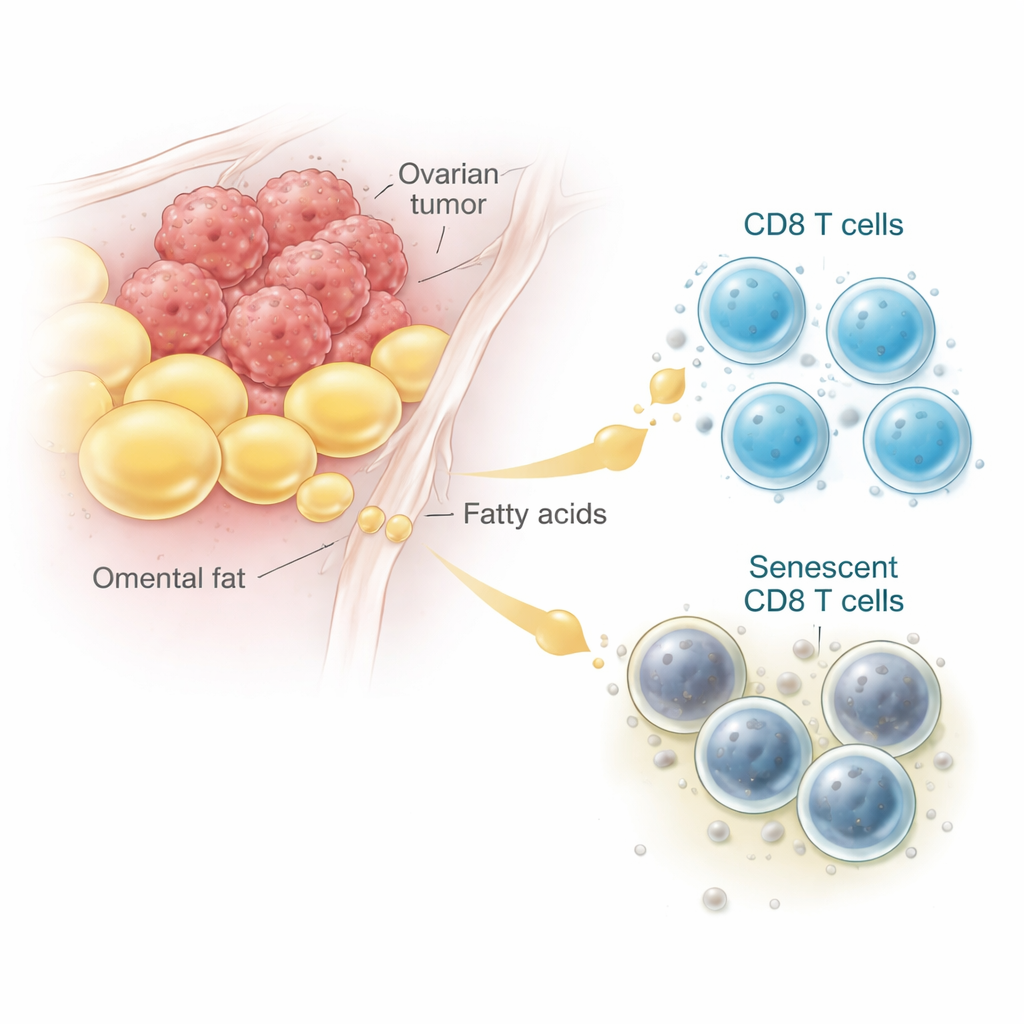
How a Common Fatty Acid Turns Helpful Fuel into Harm
To understand what fat is doing to T cells, the team recreated the tumor setting in the lab. They grew ovarian cancer cells together with mouse CD8 T cells, then added either extracts from fat tissue or a specific fatty acid called oleic acid, which is abundant in ovarian cancer fluid. At moderate to high doses, oleic acid made more CD8 T cells become senescent—but only when tumor cells were present. Instead of burning the incoming fat for energy, the T cells accumulated lipids that became chemically damaged, a process known as lipid peroxidation. Gene and lipid analyses showed increased markers of oxidized fats and stress, while energy production from fat stayed flat. In short, in the presence of tumor, fat that should be fuel was diverted into damaging reactions that age and disable T cells.
The Fat Transporter FABP4 as a Key Gatekeeper
Digging deeper, the researchers asked which molecules controlled this harmful fat influx. They found that a transporter protein called FABP4 was strongly switched on in CD8 T cells exposed to fat extracts or oleic acid. FABP4 acts like a shuttle, carrying long-chain fatty acids into cells. When they blocked FABP4 using either a small-molecule drug (BMS309403) or genetic silencing, T cells took up less fat, showed less lipid peroxidation, and were less likely to become senescent. These protected T cells divided better, produced more cancer-killing molecules such as interferon-gamma and granzyme B, and released fewer suppressive and inflammatory signals. Restoring FABP4 levels reversed these benefits, confirming that this transporter is a central switch linking fat uptake to T cell aging.
From Mouse Experiments to a Potential Treatment Strategy
The team then tested FABP4 blockade in living mice with ovarian cancer that had spread inside the abdomen. Treating mice with the FABP4 inhibitor reduced fat uptake and lipid damage in CD8 T cells drawn from the abdominal fluid, lowered the proportion of senescent T cells, and boosted levels of key effector molecules. When the FABP4 inhibitor was combined with standard chemotherapy, mice had fewer visible tumor nodules, stronger T cell responses in spleen, lymph nodes, and ascites, and lived longer than those given chemotherapy alone. These findings suggest that targeting fat metabolism can make existing treatments more effective by reviving the immune system’s frontline soldiers.
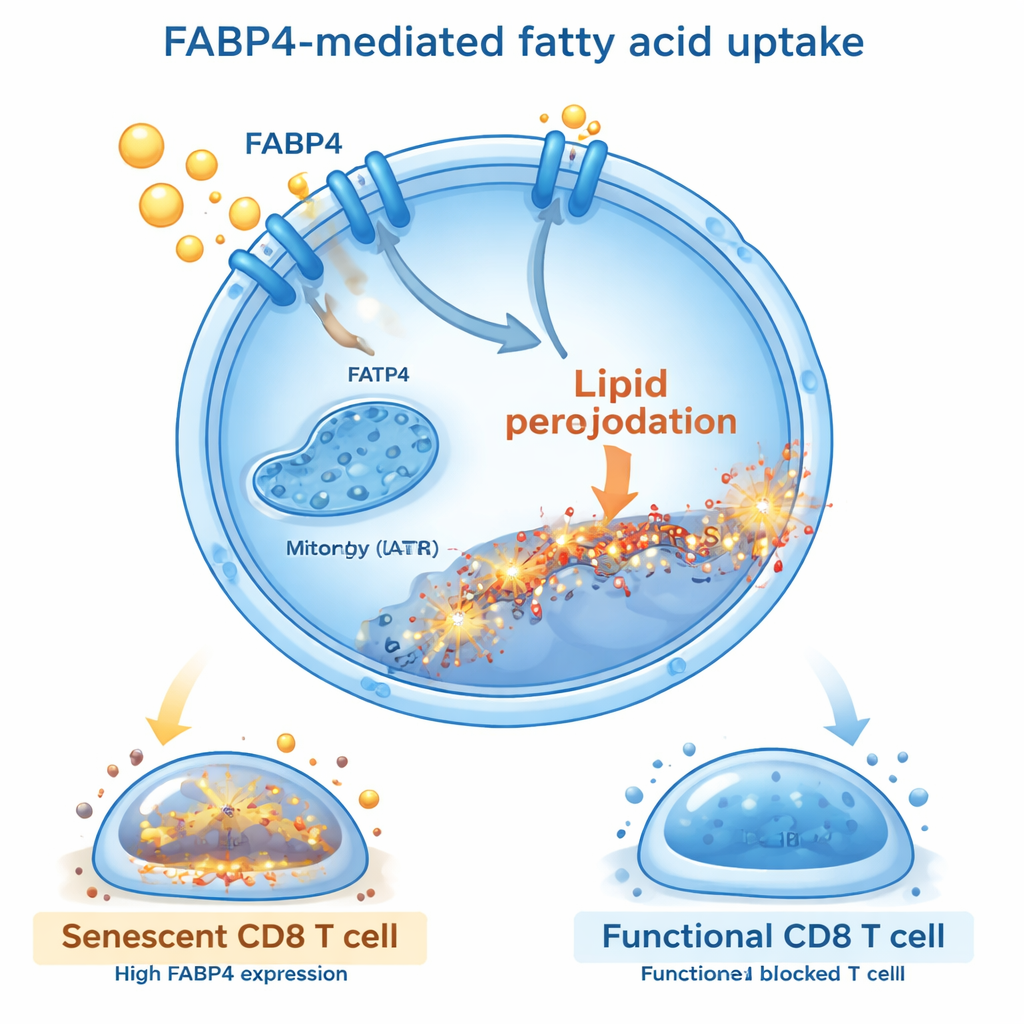
What This Means for Patients
For lay readers, the takeaway is that in ovarian cancer, it is not just the tumor that matters, but also the fatty neighborhood it lives in. Fat-derived molecules, especially certain fatty acids, can prematurely age killer T cells, robbing them of their ability to attack cancer. This work identifies the fat transporter FABP4 as a key culprit in that process. By blocking FABP4, at least in mice, researchers can reduce T cell aging, restore their killing function, and improve the impact of chemotherapy. While more research is needed before this becomes a therapy for patients, the study opens a new avenue: treating not only the cancer itself, but also the metabolic traps in its environment that disarm the immune system.
Citation: Yu, C., Li, X., Qian, X. et al. Fatty acid uptake mediated by FABP4 promotes the formation of CD8+T cell senescence through lipid peroxidation in the adipocyte-rich microenvironment of Ovarian Cancer. Oncogenesis 15, 9 (2026). https://doi.org/10.1038/s41389-026-00600-w
Keywords: ovarian cancer, T cell senescence, fatty acid metabolism, tumor microenvironment, FABP4