Clear Sky Science · en
The role of histone demethylase PHF2 as a tumour suppressor in hepatocellular carcinoma by regulating SRXN1
Why this liver cancer study matters
Liver cancer is one of the deadliest cancers worldwide, and current drugs extend life only modestly for many patients. This study asks a different question: instead of targeting the usual cancer-driving genes, can we exploit the way DNA is packaged and chemically marked inside liver cells to find new, more precise treatment options? The researchers home in on a little-known protein called PHF2 and uncover how it may help keep liver cells from turning cancerous.
Looking for weaknesses in cancer’s control system
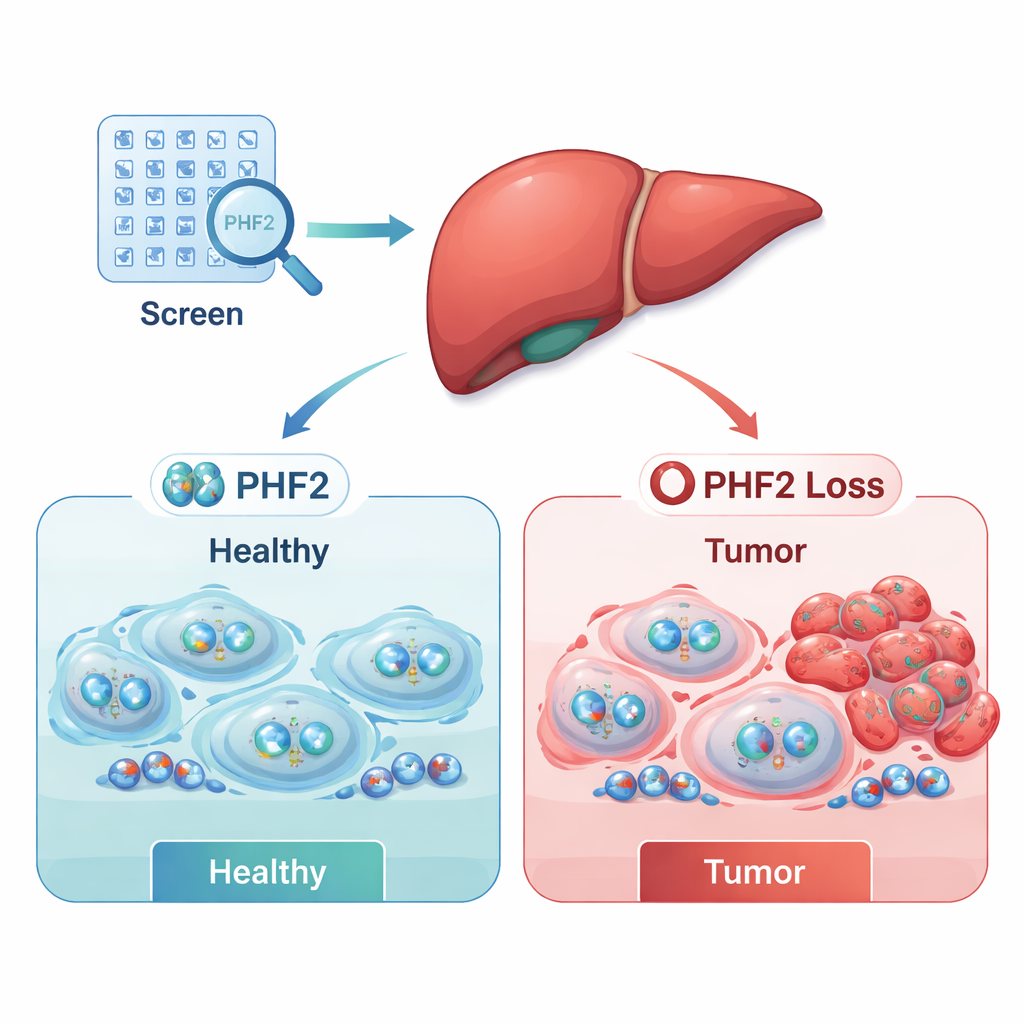
Cancer is not driven only by broken genes; it is also fueled by faulty "epigenetic" controls—the chemical tags and protein complexes that decide which genes are turned on or off. In hepatocellular carcinoma, the most common form of liver cancer, many such regulators are disturbed. The team systematically tested 497 epigenetic regulators in six liver cancer cell lines, using RNA tools to temporarily reduce each regulator one by one and then measuring how well the cancer cells survived. This large screen was designed to reveal which regulators cancers rely on to grow, and which normally act as brakes on tumor formation.
Finding new cancer helpers and protectors
The screen uncovered two broad groups: potential cancer “accelerators” (oncogenes) whose loss harmed cancer cell survival, and potential “brakes” (tumor suppressors) whose loss made cancer cells grow better. By combining their lab data with large public patient datasets, the researchers narrowed the list to a few clinically relevant candidates. Two regulators, SUPT7L and SMARCC1, tended to be more active in tumors and were linked with poorer survival. In contrast, one regulator—PHF2—was often less active in liver tumors, and patients whose tumors kept higher PHF2 levels generally lived longer. These patterns suggested that PHF2 might act as a tumor suppressor in human liver cancer.
How losing PHF2 changes liver cancer cells
To test PHF2 more deeply, the team engineered liver cancer cells to stably produce less of this protein and followed their behavior over time. Cells with reduced PHF2 grew faster and showed higher levels of Ki-67, a marker of active cell division, indicating that PHF2 normally helps restrain proliferation. These effects were most pronounced in already aggressive liver cancer cell lines, implying that simply losing PHF2 may not start cancer on its own, but can speed up the growth of malignant cells once tumors have formed.
A surprising link to the cell’s stress defenses
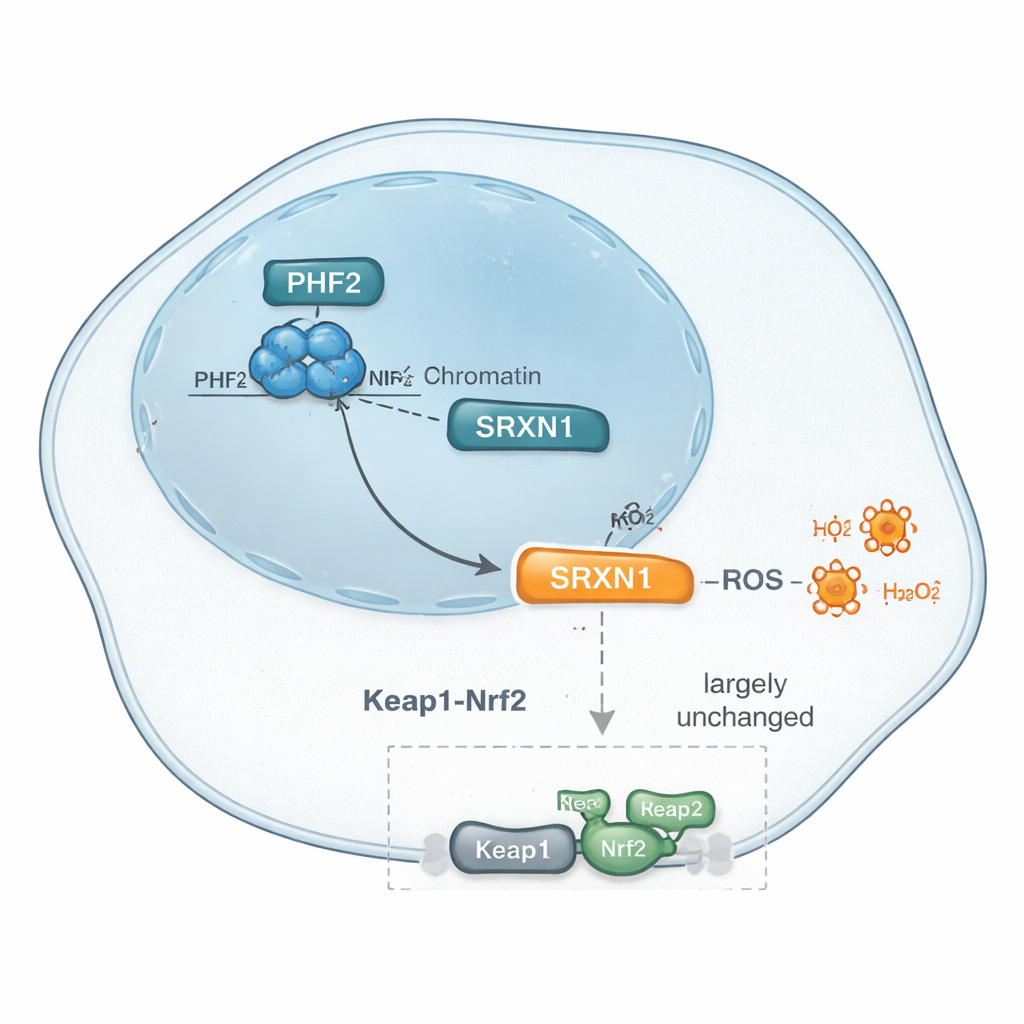
Next, the researchers used modern protein-profiling methods to catalog thousands of proteins whose levels changed when PHF2 was reduced. Among many subtle shifts, one pattern stood out: levels of a stress-protective protein called SRXN1 consistently dropped when PHF2 was lost. SRXN1 helps cells manage reactive oxygen species—chemically reactive by-products that can damage DNA and proteins but can also push cancer cells to adapt. The data suggested that PHF2 helps maintain SRXN1 levels, and that altering this balance might influence how liver cancer cells respond to internal stress and, ultimately, how quickly they grow.
Acting outside a well-known stress pathway
SRXN1 is usually controlled by a master stress-response switch called Nrf2, which turns on many antioxidant genes when cells face oxidative stress. Because SRXN1 fell when PHF2 was suppressed, the team asked whether PHF2 might simply be acting through this well-known Nrf2 pathway. They examined key Nrf2-related genes and proteins, treated cells with drugs that activate Nrf2 by blocking its inhibitor Keap1, and used genetic tools to reduce Keap1 directly. Across these tests, the response of PHF2-deficient cells looked much like that of normal cells, and broader pathway and transcription-factor analyses did not highlight Nrf2 as a main player. Instead, other factors, including ZNF384 and Nrf1—both previously linked to liver cancer—emerged as candidates. Together, the evidence suggests that PHF2’s control over SRXN1 and antioxidant responses may bypass classic Nrf2 signaling, hinting at a distinct stress-regulation route in these tumors.
What this could mean for future treatments
To a non-specialist, the key takeaway is that this study identifies PHF2 as a promising protective factor in liver cancer: when PHF2 is present and active, tumor cells grow more slowly; when it is diminished, they gain a growth advantage and alter how they handle cellular stress. Because PHF2 itself may be difficult to block or boost with conventional small-molecule drugs, the authors point to emerging strategies such as RNA-based therapies or drugs that enhance PHF2’s activity through upstream signaling partners. They also emphasize that PHF2’s effects are context-dependent—its impact may vary with other mutations like p53 status—so any future therapy would likely need careful patient selection. Overall, the work refines our understanding of how epigenetic regulators shape liver cancer behavior and opens new avenues for designing treatments that restore or mimic PHF2’s tumor-suppressive influence.
Citation: Thng, D.K.H., Hooi, L., Yong, W.K. et al. The role of histone demethylase PHF2 as a tumour suppressor in hepatocellular carcinoma by regulating SRXN1. Oncogenesis 15, 6 (2026). https://doi.org/10.1038/s41389-026-00597-2
Keywords: hepatocellular carcinoma, epigenetic regulation, PHF2, SRXN1, liver cancer therapy