Clear Sky Science · en
Cereblon (CRBN) inhibits prostate cancer metastasis by negatively regulating 6-phosphogluconate dehydrogenase (6PGD)
Why this research matters
When prostate cancer spreads from the prostate to other organs, it becomes far harder to treat and is often deadly. This study reveals how a little-known cellular “quality control” protein, cereblon (CRBN), helps keep prostate cancer from spreading by dismantling a metabolic enzyme that fuels aggressive behavior. Understanding this built‑in brake system suggests new ways to slow or stop metastasis, especially in hard‑to-treat forms of the disease.
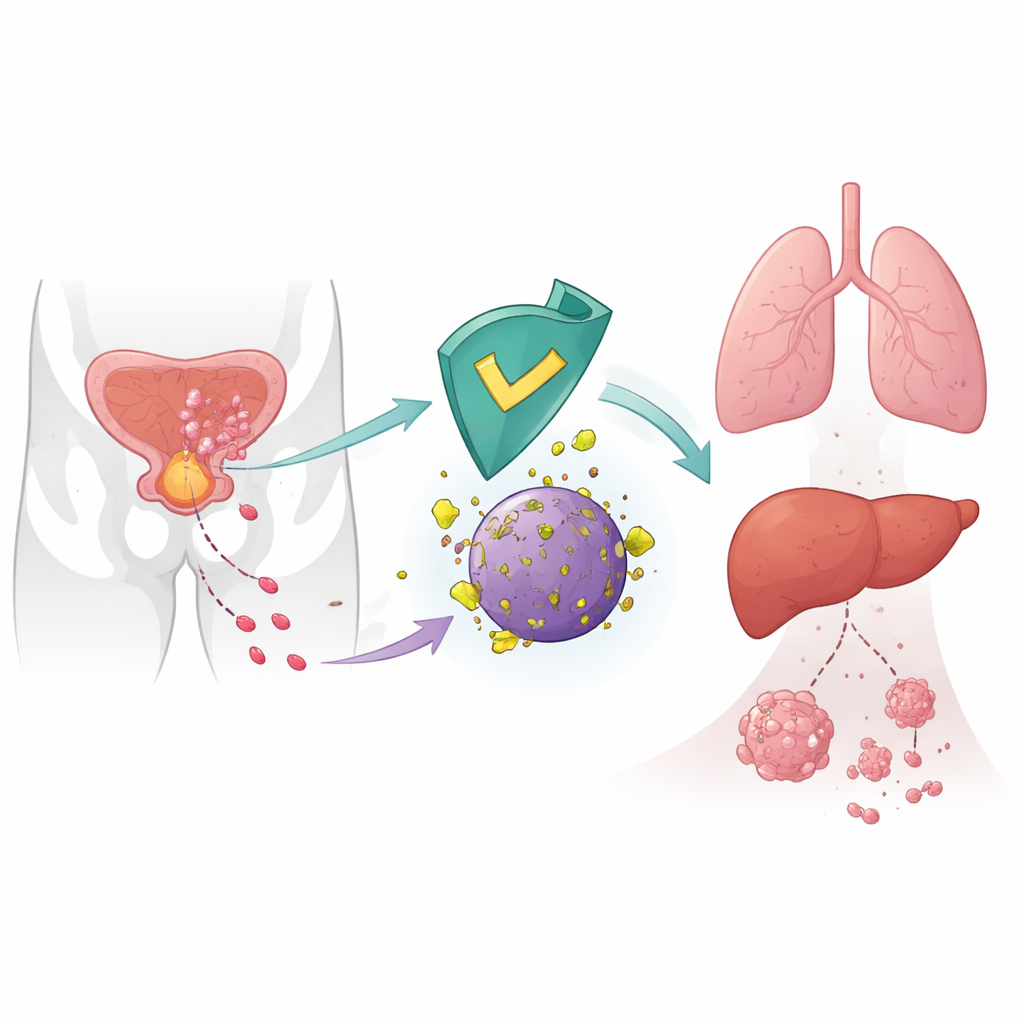
An internal brake on cancer spread
Cancer cells do not spread at random; they rewire their inner machinery to move, invade, and colonize distant organs. The authors focused on CRBN, a protein best known as the target of drugs like thalidomide used in blood cancers. In healthy cells, CRBN is part of a molecular disposal unit that tags certain proteins for destruction. Earlier work hinted that low CRBN levels in tumors were linked to worse outcomes, but its direct role in metastasis was unclear. Here, the researchers show that CRBN acts as a natural suppressor of prostate cancer spread by targeting a metabolic enzyme called 6-phosphogluconate dehydrogenase (6PGD) for breakdown.
A metabolic engine that cancer hijacks
6PGD sits in a metabolic route known as the oxidative pentose phosphate pathway, which helps cells generate NADPH, a molecule that supplies both building blocks and antioxidant power. Many tumors ramp up this pathway to grow faster and tolerate stress. The team found that prostate cancer samples from patients tend to have less CRBN and more 6PGD than normal prostate tissue, and that this imbalance is especially pronounced in aggressive disease. In prostate cancer cell lines, knocking down CRBN or degrading it with a designer compound caused 6PGD protein levels and enzyme activity to rise, boosting NADPH. Restoring CRBN had the opposite effect, lowering 6PGD and NADPH without changing the underlying 6PGD gene message, pointing to a protein-level control.
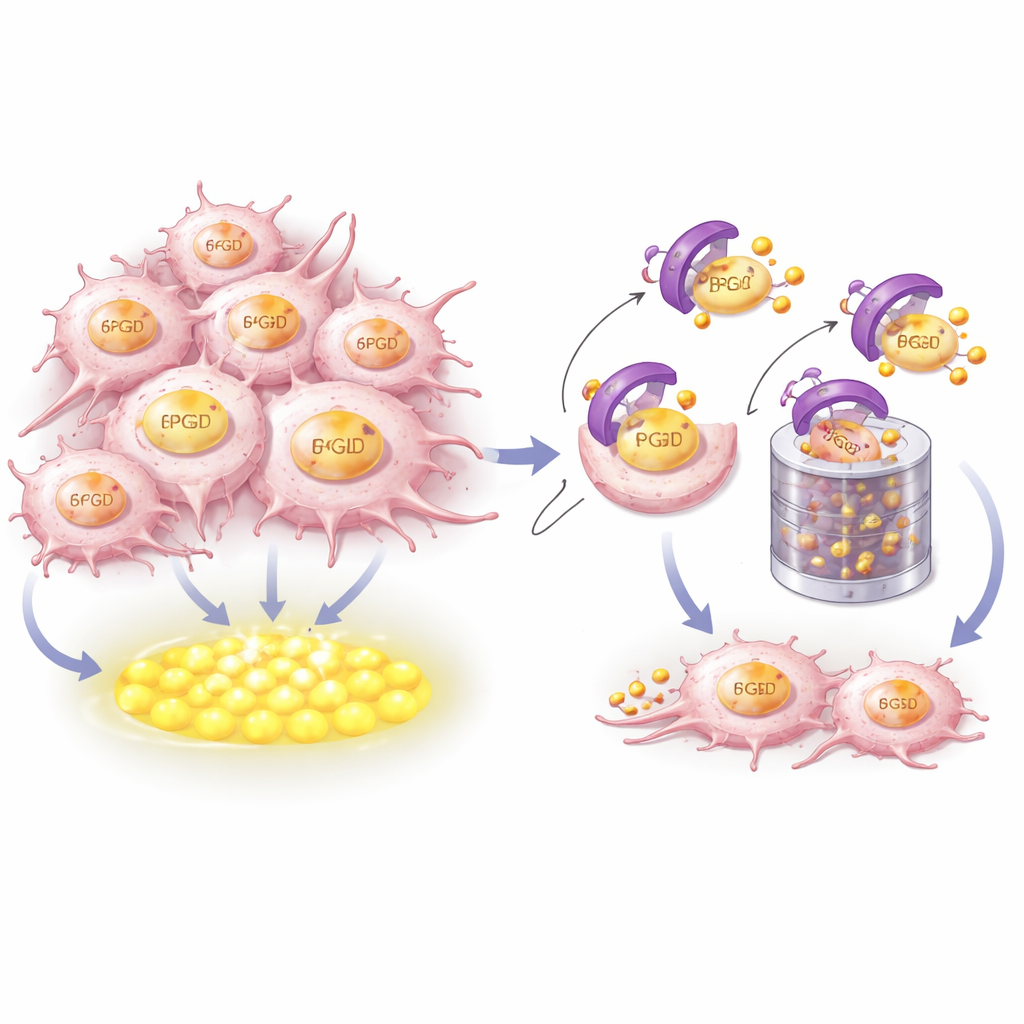
How CRBN recognizes and destroys the fuel source
To understand how CRBN controls 6PGD, the researchers combined structural modeling with biochemical experiments. They showed that CRBN physically grabs a short, highly conserved helical segment near the tail end of 6PGD. Key amino acids on both proteins form a snug docking interface. When these contact points were altered, CRBN could no longer bind 6PGD, attach the small ubiquitin tags that mark it for disposal, or send it to the cellular shredder known as the proteasome. Notably, this recognition surface is separate from the pocket where immune-modulating drugs bind CRBN, meaning CRBN’s control over 6PGD is part of its native housekeeping role rather than a drug-induced effect.
From altered metabolism to wandering cells
The consequences of this molecular handshake play out at the level of cell behavior. In prostate cancer cells lacking CRBN, elevated 6PGD and NADPH went hand in hand with increased movement and invasion through artificial barriers, hallmarks of metastatic potential. When CRBN was overproduced, cells became less mobile and invasive, even though their growth rate did not change. Gene activity profiling showed that CRBN raised levels of CDH1, associated with tightly packed epithelial cells, and lowered levels of MMP1, linked to tissue breakdown and migration. Importantly, these shifts required direct CRBN–6PGD binding and could be reversed by blocking 6PGD’s enzyme activity, tying changes in metabolism to changes in metastatic behavior.
Evidence from animal models and other cancers
The team tested these ideas in mice by injecting prostate cancer cells into the bloodstream or spleen. Cells engineered to produce extra CRBN formed far fewer metastatic nodules in the liver and lungs than control cells, confirming an anti-metastatic role in living animals. Mice engineered to lack CRBN showed higher 6PGD protein levels in several organs. Similar patterns appeared in glioblastoma, lung, and kidney cancer cell lines: removing CRBN increased 6PGD and boosted cell movement and invasion, while genetic or drug-mediated 6PGD inhibition curbed this aggressive behavior. These observations suggest that the CRBN–6PGD brake on metastasis is not unique to prostate cancer.
What this could mean for future treatments
Overall, the study proposes that CRBN serves as a metabolic gatekeeper that restrains the energy supply needed for cancer cells to loosen, migrate, and seed new tumors. When CRBN is lost or low, 6PGD accumulates, NADPH production surges, and cells are more likely to undergo changes that favor metastasis. For patients, this points to two possibilities: measuring CRBN and 6PGD levels could help flag tumors with high metastatic risk, and therapies that boost CRBN function—or selectively dampen 6PGD in CRBN-poor tumors—might offer new ways to slow or prevent the spread of prostate and other cancers.
Citation: Guchhait, K., Yoon, HS., An, HS. et al. Cereblon (CRBN) inhibits prostate cancer metastasis by negatively regulating 6-phosphogluconate dehydrogenase (6PGD). Oncogene 45, 1234–1246 (2026). https://doi.org/10.1038/s41388-026-03717-9
Keywords: prostate cancer metastasis, cancer metabolism, cereblon, 6PGD enzyme, NADPH pathway