Clear Sky Science · en
L1TD1 promotes colorectal mucinous adenocarcinoma progression by enhancing ABCC3 mRNA stability
Why this mucus-heavy cancer matters
Colorectal cancer is a leading cause of cancer death worldwide. Within it, a special subtype called mucinous adenocarcinoma stands out because its tumors are filled with thick mucus that makes up more than half of the tumor mass. That slimy barrier may help cancer cells hide from chemotherapy and the body’s defenses. This study asks a basic but crucial question: what controls all that mucus, and can we shut it down to slow the cancer and make treatments work better?
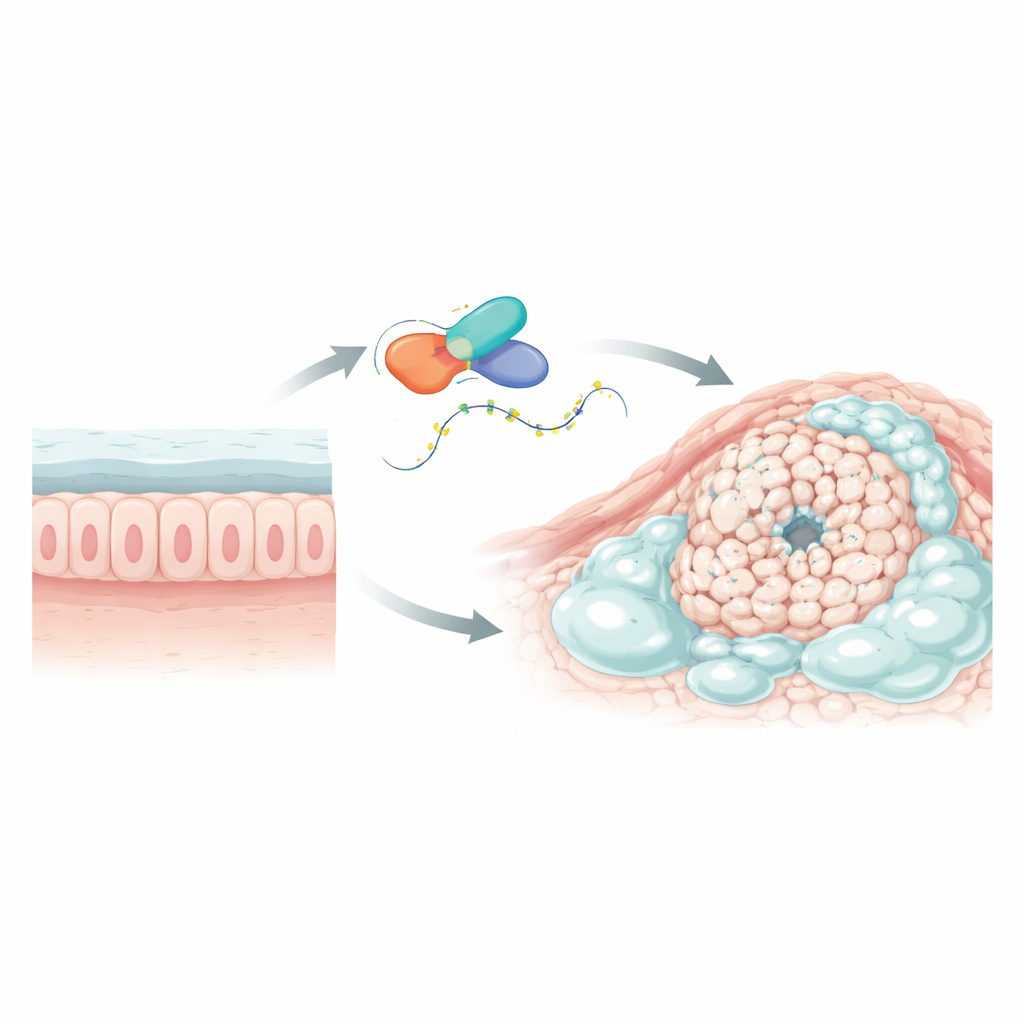
Finding a key troublemaking gene
The researchers first searched large cancer databases and single-cell sequencing data to pinpoint genes that behave differently in mucus-rich colorectal tumors compared with ordinary colorectal cancers and nearby healthy tissue. One gene, called L1TD1, clearly stood out. It was sharply increased in mucinous tumors but not in regular tumors and was higher in tumor tissue than in surrounding normal colon. L1TD1 encodes a protein that binds to RNA, the molecule that carries genetic messages from DNA to the cell’s protein-making machinery. Because RNA-binding proteins can fine-tune how long messages last and how strongly they are read, L1TD1 was a strong candidate to act as a master regulator in these mucus-laden cancers.
How L1TD1 pushes cancer growth and spread
To test what L1TD1 actually does, the team turned to colon cancer cell lines and mouse models. When they boosted L1TD1 levels in cancer cells, the cells multiplied faster, moved more readily, and invaded through tissue-like barriers more aggressively. Dialing L1TD1 down had the opposite effect, slowing growth and reducing invasion. In mice injected with L1TD1-high cells, tumors grew larger and spread more often to the liver and abdominal cavity. Tumors with more L1TD1 also tended to be bigger, less well differentiated, and at more advanced stages in patients, linking this gene to worse disease behavior in the clinic.
Turning on mucus and drug resistance
Mucus itself turned out to be central to L1TD1’s impact. Cells with more L1TD1 produced higher amounts of key mucus-forming proteins, especially MUC2 and MUC5AC, and showed features of goblet cells, the normal mucus-secreting cells of the gut. Three-dimensional cultures and tissue staining from both patients and mice confirmed that higher L1TD1 went hand in hand with thicker, more abundant mucus. Importantly, this mucus was not just a bystander. When the researchers selectively reduced MUC2, the extra growth, invasion, and chemotherapy resistance driven by L1TD1 largely disappeared. Cells rich in L1TD1 were harder to kill with the common drug oxaliplatin, and tumors carrying these cells shrank less in treated mice, underscoring mucus as a shield that L1TD1 helps build.
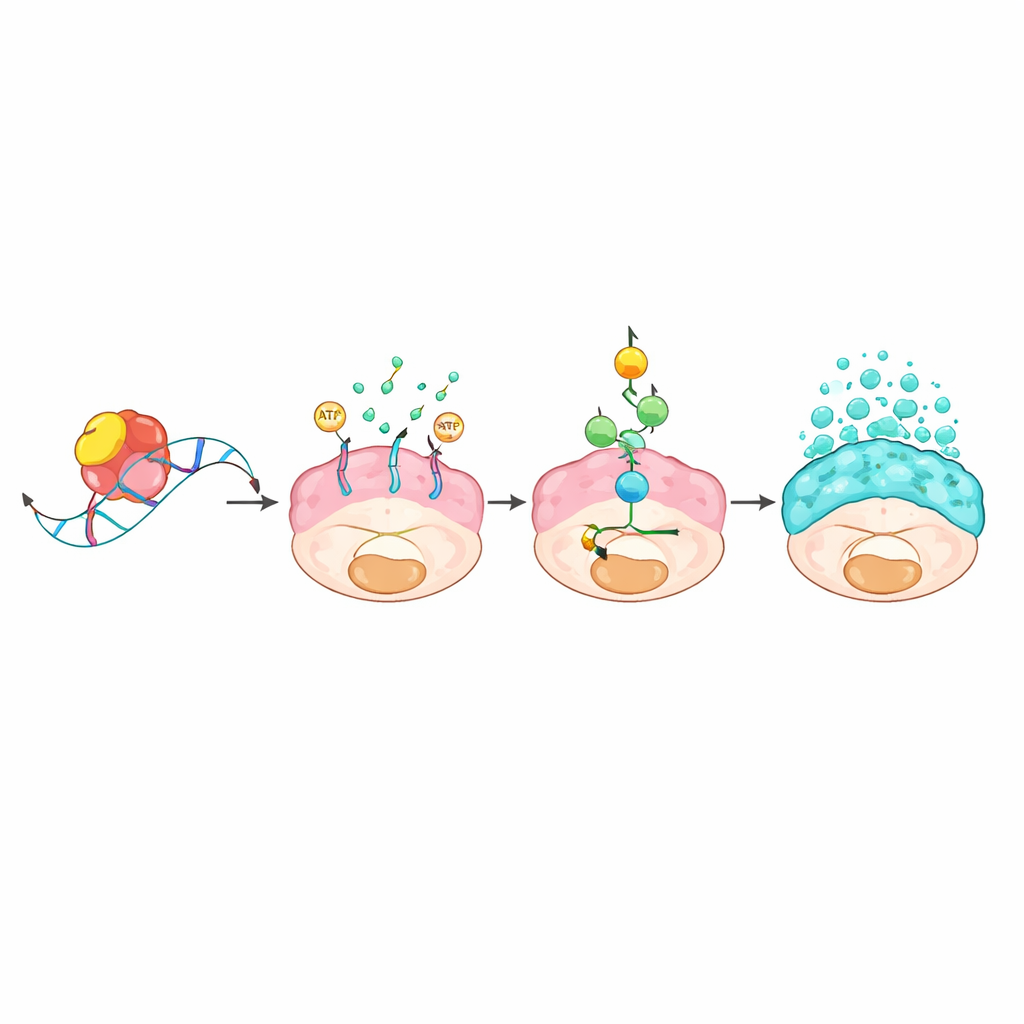
A molecular chain linking RNA control to mucus floods
The team next asked how an RNA-binding protein could cause such dramatic changes. By fishing out RNA molecules physically attached to L1TD1 and comparing gene activity with and without L1TD1, they identified a transporter gene called ABCC3 as a prime target. L1TD1 clamped onto a short “GUGU” motif in the tail end of ABCC3’s RNA message, stabilizing it so the cell made more of the ABCC3 protein, known as MRP3. This transporter sits in the cell membrane and consumes energy to pump substances out of the cell. The resulting energy drain activated a metabolic alarm system centered on the sensor AMPK, which in turn switched on a classic growth and stress pathway known as MAPK. Once activated, this cascade boosted the production and release of mucus proteins and reinforced the cancer cells’ ability to grow, migrate, and resist drugs. Blocking ABCC3 genetically or with an experimental compound, or inhibiting the MAPK pathway, all dampened mucus production and weakened the tumors.
What this means for patients
Taken together, the work reveals a stepwise control system for mucus-heavy colorectal cancer: L1TD1 stabilizes ABCC3’s RNA, ABCC3 drains cellular energy, the AMPK–MAPK pathway switches on, and the tumor responds by overproducing mucus and becoming more aggressive and drug-resistant. For patients, this suggests several promising angles. L1TD1 and ABCC3 levels could help flag tumors likely to behave badly or resist standard chemotherapy. More importantly, drugs that disrupt the L1TD1–ABCC3 interaction, block MRP3’s pump activity, or target the downstream signaling pathway might thin the mucus barrier, slow tumor growth, and make existing treatments more effective for this challenging form of colorectal cancer.
Citation: He, H., Yuan, J., Wang, H. et al. L1TD1 promotes colorectal mucinous adenocarcinoma progression by enhancing ABCC3 mRNA stability. Oncogene 45, 1071–1086 (2026). https://doi.org/10.1038/s41388-026-03716-w
Keywords: mucinous colorectal cancer, L1TD1, ABCC3 MRP3, mucin production, chemoresistance