Clear Sky Science · en
The multifunctional RNA helicase DDX39A drives glioblastoma progression by modulating WISP1 alternative splicing that induces an immunosuppressive macrophage polarization
Why this brain cancer study matters
Glioblastoma is one of the deadliest brain cancers, notorious for returning after treatment and resisting drugs. This study uncovers how a single molecular “switch” in tumor cells not only helps the cancer grow, but also persuades nearby immune cells to stop fighting and start protecting the tumor instead. By tracing this chain of events, the researchers also identify an existing antipsychotic drug that could be repurposed to disrupt this harmful network.
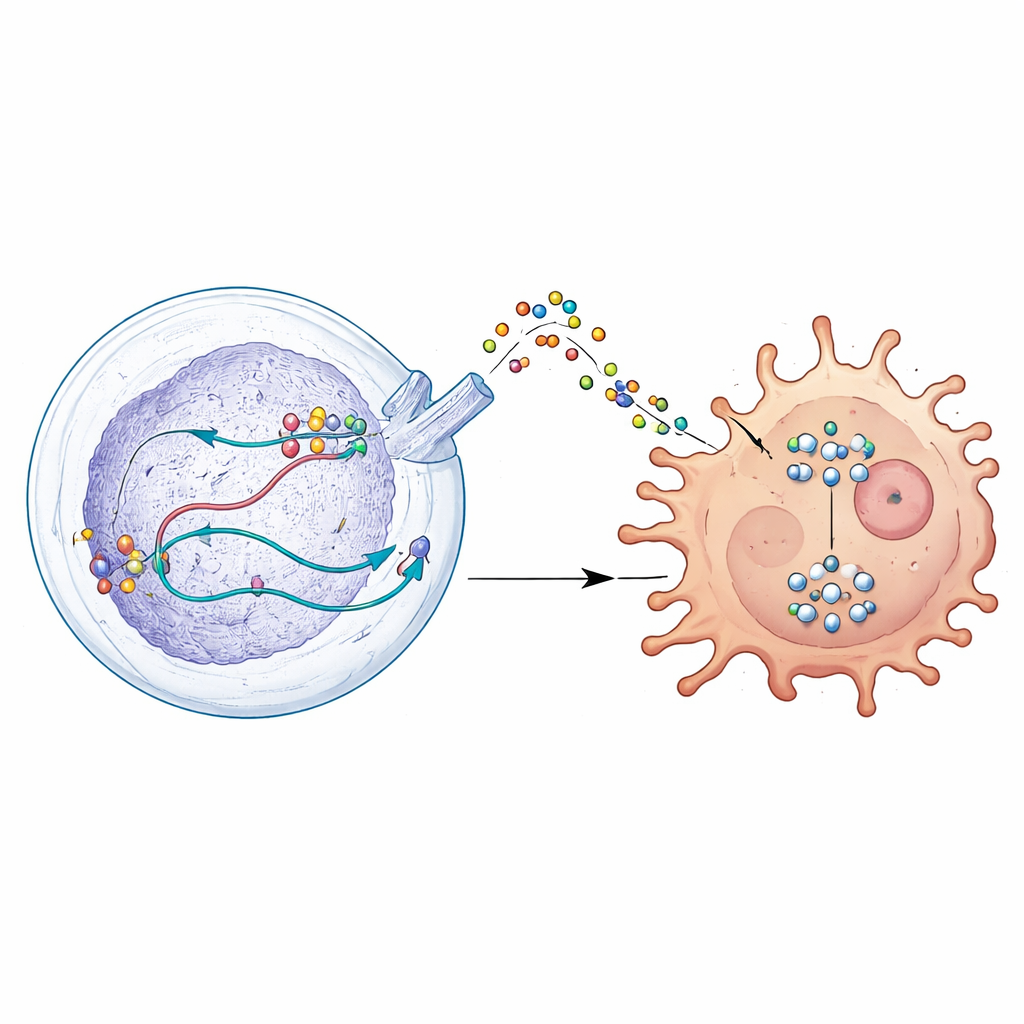
A key helper inside brain tumor cells
The team focused on a protein called DDX39A, a molecular machine that helps cells process RNA, the working copy of genetic information. By analyzing large patient datasets and tumor samples, they found that DDX39A is much more active in glioblastoma than in normal brain tissue, and its levels rise with tumor grade. Single-cell analyses showed that DDX39A is especially abundant in fast-dividing tumor cells and in certain immune cells called macrophages inside the tumor mass. Patients whose tumors had higher DDX39A tended to have worse outcomes, suggesting that this protein is closely tied to aggressive disease.
How this helper fuels growth and spread
To test what DDX39A actually does, the researchers used genetic tools to dial it down in glioblastoma stem-like cells grown in the lab and in mouse brain tumor models. When DDX39A was reduced, tumor cells grew more slowly, invaded surrounding tissue less effectively, and were more likely to undergo programmed cell death. The cancer stem-like cells also lost much of their ability to form new tumor spheres, a sign that their capacity for self-renewal was impaired. In mice, tumors with lowered DDX39A were smaller and the animals lived longer, confirming that this protein actively drives tumor progression, not just passively marks it.
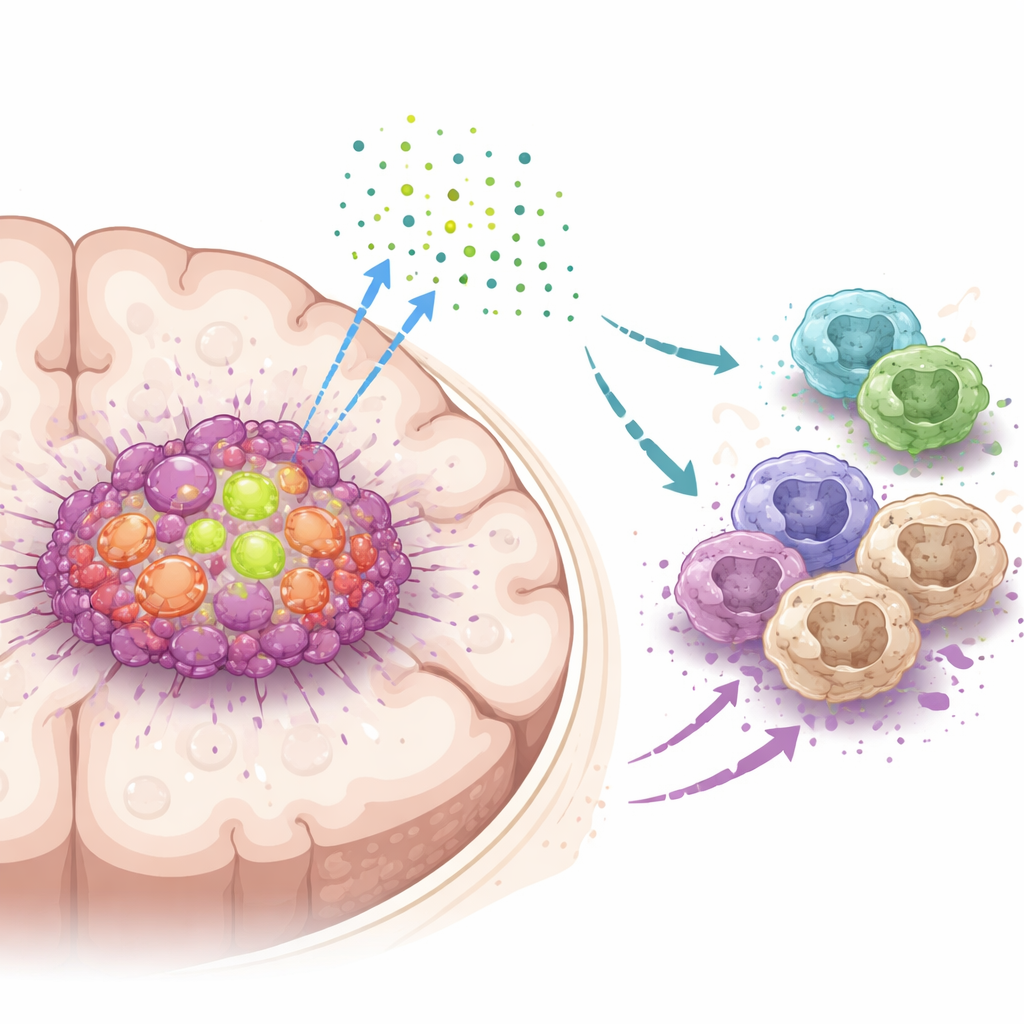
A secret signal that disarms immune defenders
Digging deeper, the scientists discovered that DDX39A changes how a gene called WISP1 is processed. Instead of simply turning WISP1 on or off, DDX39A helps shape its RNA in a way that stabilizes the message and boosts production of WISP1 protein. Tumor cells then release WISP1 into their surroundings, where it acts like a distressingly deceptive beacon. Nearby macrophages, which can either attack tumors or support them, respond to this signal by shifting into an immunosuppressive, tumor-helping state. This shift is driven through activation of a growth-control pathway inside cells known as AKT. When DDX39A or WISP1 was reduced, this pathway quieted down, tumor cells became less aggressive, and macrophages moved back toward a more tumor-fighting profile.
Turning a psychiatric drug into a cancer weapon
Armed with this pathway map, the researchers searched drug databases for molecules that might interfere with DDX39A’s activity. They homed in on fluphenazine, a long-used antipsychotic medication known to reach the brain. Computer modeling and binding tests showed that fluphenazine can attach directly to DDX39A. In tumor cells, the drug weakened DDX39A’s grip on WISP1 RNA, lowered WISP1 levels, and reduced AKT signaling. Treated cells grew less, invaded less, formed fewer tumor spheres, and were more prone to die. In mouse models, fluphenazine slowed brain tumor growth, reduced the number of tumor-supporting macrophages, and extended survival, all while being delivered at doses similar to those already used in humans for other conditions.
What this means for future treatment
In everyday terms, this study shows that glioblastoma cells exploit DDX39A to fine-tune a message (WISP1) that both boosts their own stamina and reprograms nearby immune cells into bodyguards instead of soldiers. Blocking this helper, either genetically or with a drug like fluphenazine, weakens the cancer on two fronts: the tumor cells themselves and the protective shield of immunosuppressive macrophages. While much more testing is needed before this strategy can reach patients, the work opens a promising route to repurpose a familiar medicine to hit a newly revealed weak spot in one of the most stubborn brain cancers.
Citation: Zhang, Y., Xue, Z., Zhang, N. et al. The multifunctional RNA helicase DDX39A drives glioblastoma progression by modulating WISP1 alternative splicing that induces an immunosuppressive macrophage polarization. Oncogene 45, 1026–1041 (2026). https://doi.org/10.1038/s41388-026-03715-x
Keywords: glioblastoma, RNA splicing, tumor microenvironment, macrophage polarization, drug repurposing