Clear Sky Science · en
Association of retinoids, retinoic acid receptors and epigenetics in breast cancer
Why Vitamin A Matters for Breast Cancer
Most people know vitamin A as a nutrient important for vision, but its active form in the body, retinoic acid, also helps cells decide whether to grow, specialize, or self‑destruct when damaged. This review article looks at how retinoic acid and its cellular “docking stations” are altered in breast cancer, why promising lab results have not yet translated into successful drugs for patients, and how new insights into the cancer cell’s “software” — its epigenetic code — may revive this treatment strategy.
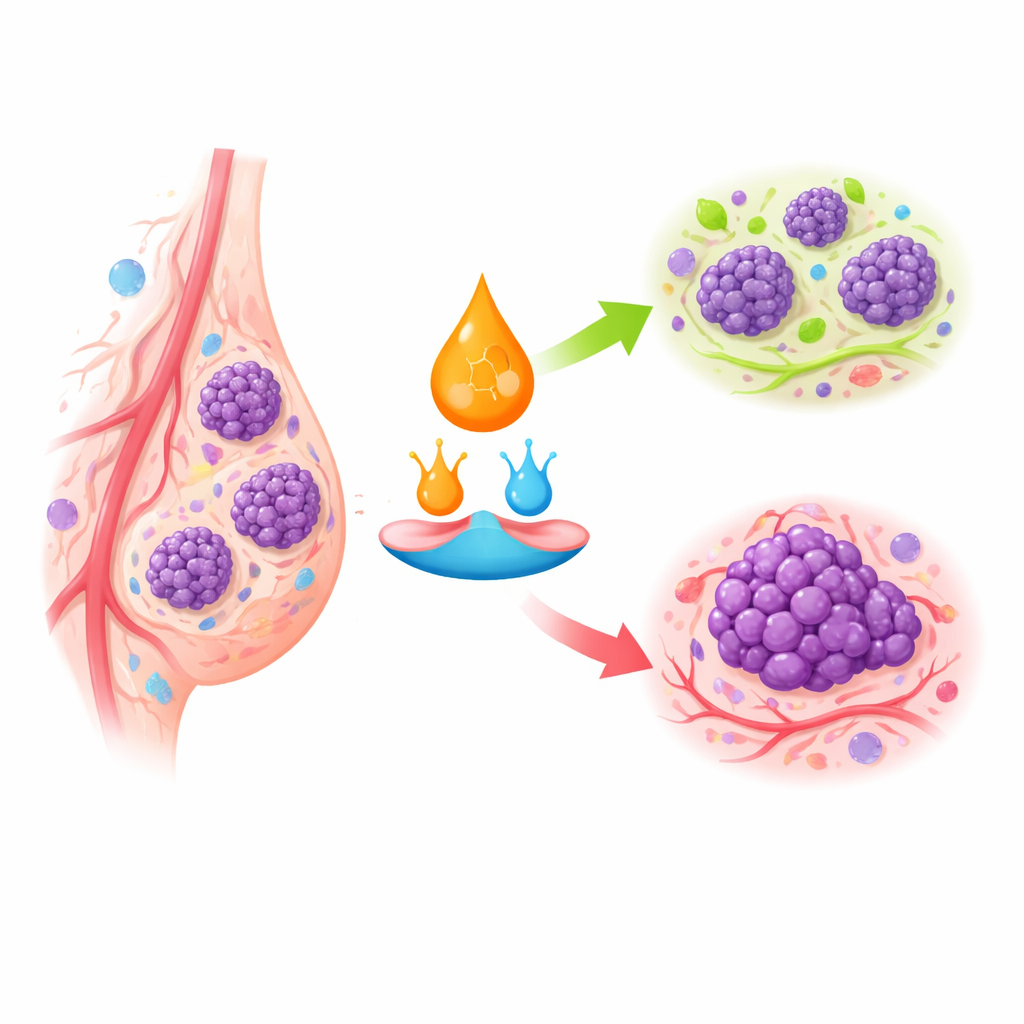
Cellular Traffic Lights for Growth and Self‑Destruction
Retinoic acid works by binding to specific receptors inside cells, which then switch whole groups of genes on or off. In healthy breast tissue, this system encourages cells to stop dividing, mature into stable forms, or die if they become abnormal. In breast cancer, especially in the more aggressive subtypes, key receptors are missing or silenced, and the internal transport of retinoic acid is rerouted. Instead of being delivered to receptors that slow growth, it can be shunted toward pathways that encourage survival and proliferation. This helps explain why some breast tumors barely respond to retinoid drugs even though the same drugs are highly effective in certain blood cancers.
Hidden Switches in the Cancer Cell’s Software
The authors focus on epigenetics — chemical tags on DNA and its packaging proteins that act like software settings for the genome. In many breast cancers, these tags lock important retinoic acid receptors, especially one called RARβ2, into an “off” position without changing the underlying DNA sequence. Extra methyl groups on DNA and tightening of the surrounding protein spool shut down this receptor, while other enzymes reshape the chromatin landscape to favor growth and migration. Non‑coding RNAs, which do not make proteins but influence gene activity, further skew signaling away from cell death and differentiation. Together, these changes give cancer cells flexibility to resist treatment and adapt to stress.
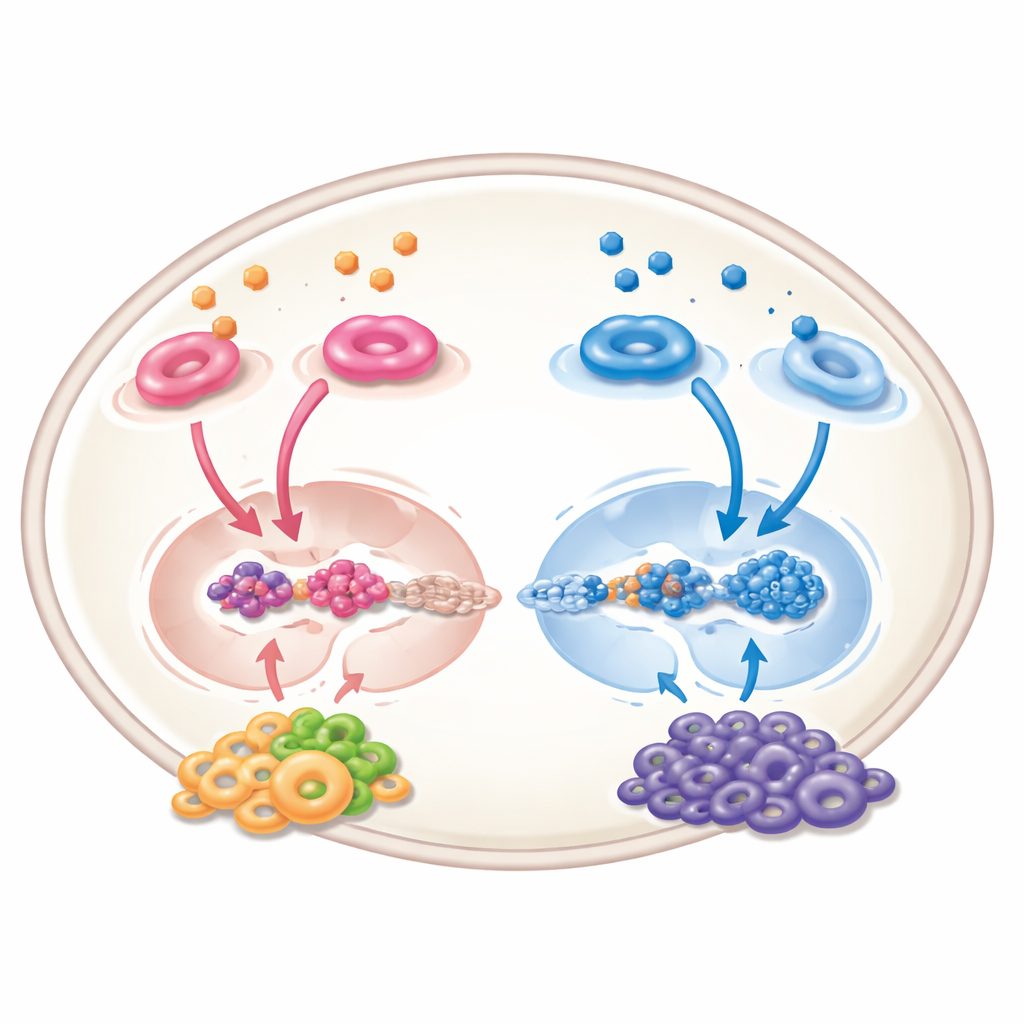
Rewiring the Signal Pathways
Importantly, these epigenetic locks are reversible. In laboratory models, drugs that remove DNA methylation or loosen chromatin can restore RARβ2 and other components of the retinoic acid pathway. When combined with retinoic acid itself and standard chemotherapy, these “priming” agents shrink tumors more effectively and reduce the pool of stem‑like cells thought to fuel relapse. The way retinoic acid is shuttled inside the cell also matters: one carrier protein guides it to growth‑suppressing receptors, while another delivers it to pro‑growth pathways. Adjusting the balance between these carriers can flip retinoic acid from acting like a fertilizer to acting like a brake.
Beyond the Tumor: The Neighborhood Effect
Retinoic acid does not act only on cancer cells; it also reshapes the tumor’s surrounding “neighborhood” of blood vessels, immune cells, and support cells. In experimental systems, it can reduce new vessel growth, push immune cells toward a more active, tumor‑fighting state, and limit suppressive cells that normally blunt the body’s defenses. Yet the story is not entirely positive: in certain stromal cells that surround the tumor, activation of retinoic acid signaling can actually promote cancer growth by strengthening chemical communication channels that attract and support malignant cells. These opposing effects mean that any future therapy must be carefully tuned to hit the right cell types in the right way.
Designing Smarter Trials for the Future
Early clinical trials of retinoid drugs in breast cancer largely failed, but they were conducted before today’s understanding of subtypes, biomarkers, and complex pharmacology. The authors argue that new trials should select patients whose tumors show the molecular hallmarks of retinoid sensitivity, such as preserved receptor expression or specific DNA‑methylation patterns, and should pair retinoids with drugs that unlock epigenetic blocks, target growth pathways, or boost immune attack. Improved formulations that keep drug levels steady in the body, along with close monitoring of both tumor and surrounding tissue, will be crucial. If these hurdles can be overcome, retinoic acid–based treatments may yet find a place as precision tools against selected forms of breast cancer.
Citation: Szymański, Ł., Schenk, T., Ławiński, M. et al. Association of retinoids, retinoic acid receptors and epigenetics in breast cancer. Oncogene 45, 961–970 (2026). https://doi.org/10.1038/s41388-026-03699-8
Keywords: retinoic acid, breast cancer, epigenetics, differentiation therapy, tumor microenvironment