Clear Sky Science · en
Modelling adipose tissue-cancer crosstalk: a three-dimensional perspective
Why Fat and Cancer Are Closer Than You Think
Most of us think of body fat as extra padding or stored energy, but research now reveals it as a busy, talkative organ that can shape our health in surprising ways. This review article explores how fat tissue and cancer cells communicate, and how new three-dimensional (3D) lab models are helping scientists mimic that dialogue more realistically than ever before. Understanding this hidden conversation could improve cancer prevention, drug testing, and treatments, especially in an increasingly overweight world. 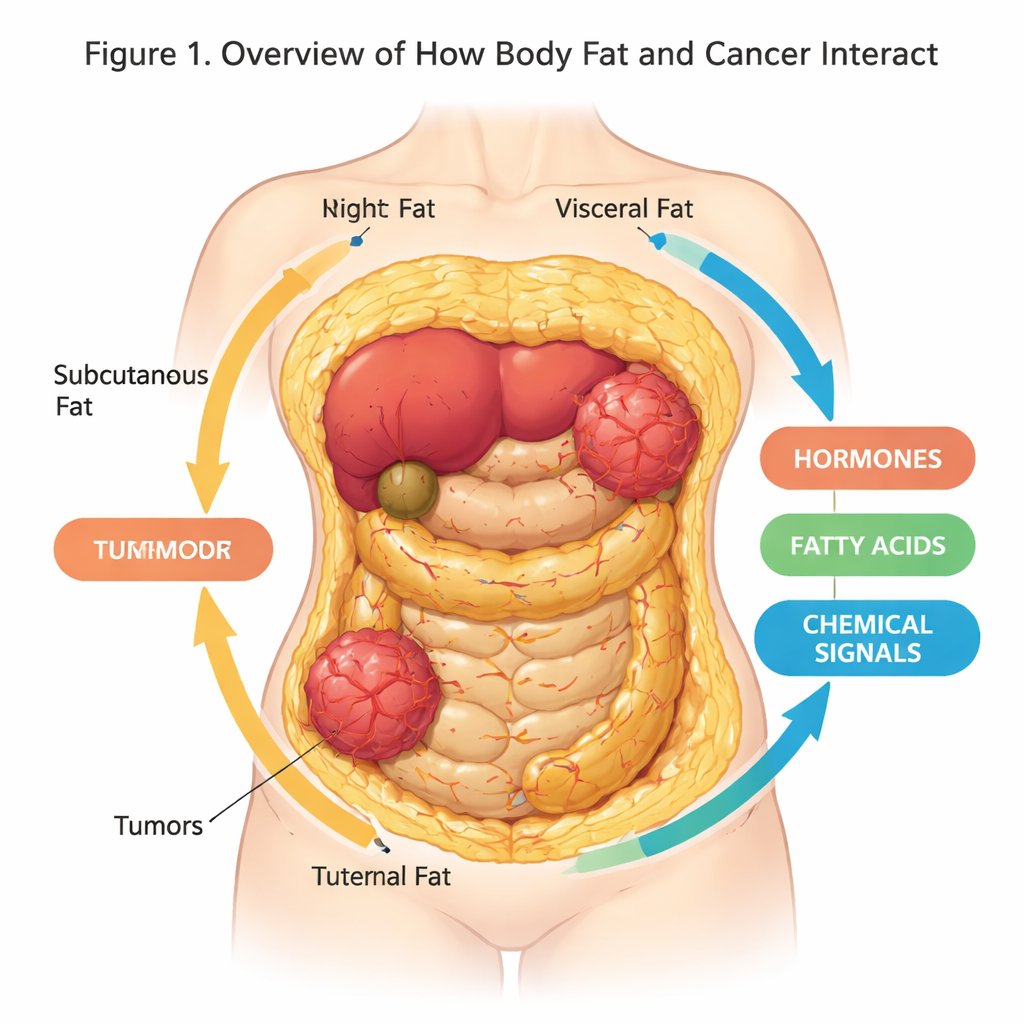
Fat as a Living, Talking Organ
Adipose tissue—our body fat—is not just a passive storage site. It is a complex organ made up of fat cells, immune cells, blood vessels, and support cells, all embedded in a structural mesh known as the extracellular matrix. Different kinds of fat exist in different places: subcutaneous fat under the skin, visceral fat around internal organs, and white, brown, and beige fat cells that vary in how they store energy or burn it as heat. These cells constantly release a cocktail of hormones, fatty acids, and signaling proteins that influence metabolism, inflammation, and, as we now know, cancer risk and behavior.
Obesity, Inflammation, and Cancer Risk
When we gain too much weight, white fat cells grow in size and number and the tissue becomes stressed and inflamed. This “unhealthy” fat releases more inflammatory molecules and fatty acids into the bloodstream, while handling sugar and fat less efficiently. Large epidemiological studies have linked obesity to a higher risk and worse outcomes for many cancers, including breast, colon, pancreatic, and uterine cancers. The review explains how dysfunctional fat can feed tumors with extra fuel, stiffen the surrounding tissue, and recruit other cell types that together create a more cancer-friendly environment.
A Two-Way Conversation Between Fat and Tumors
Cancer is now seen as a disease of an entire ecosystem, not just of rogue cells. Tumor cells tap into nearby fat for support, reprogramming fat cells, immune cells, and fibroblasts to help them grow and spread. In return, fat cells respond by breaking down stored lipids and releasing fatty acids and hormones that cancer cells can readily use. This cross-talk can happen at a distance through circulating signals, or locally where tumors grow next to fat, as in the breast or abdomen. Tumor cells can even drive fat cells to lose their original identity and become more like scar-forming or immune-like cells that reshape the tissue and promote invasion. 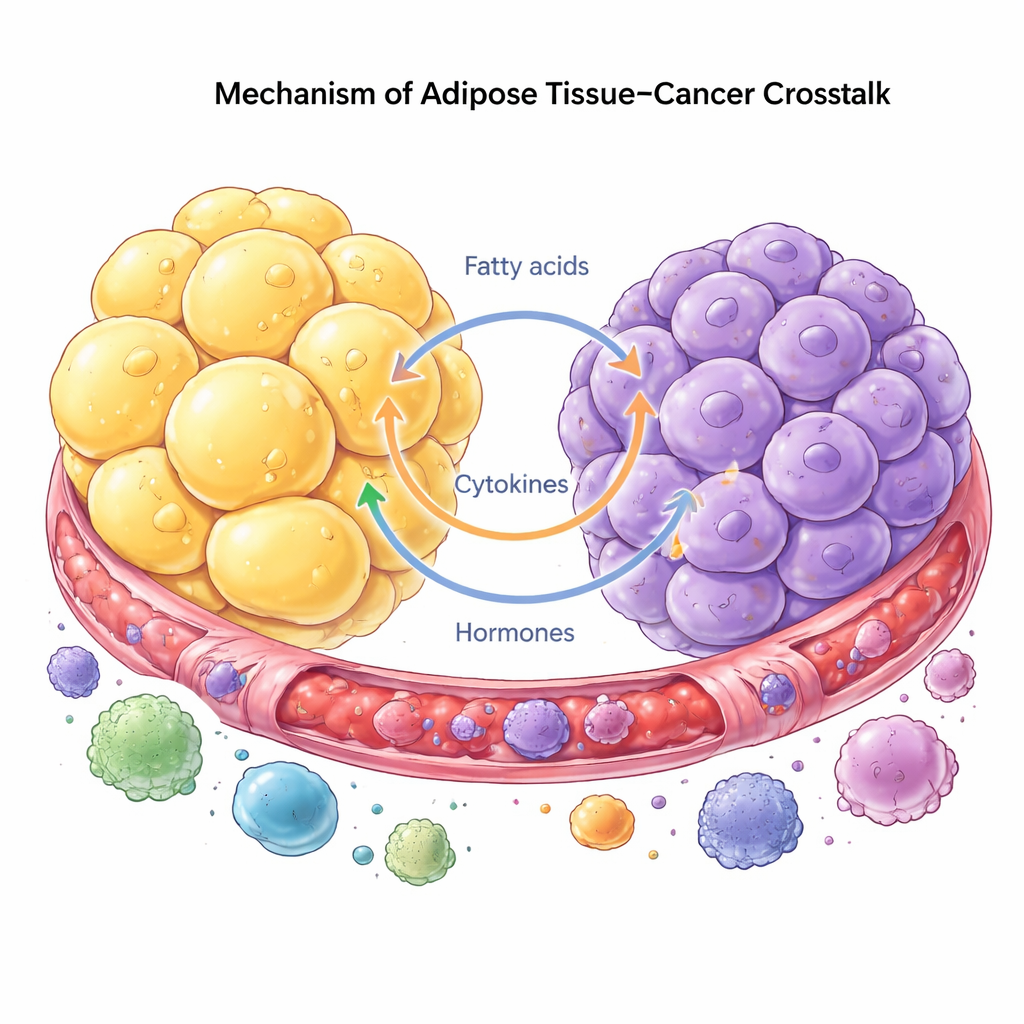
Building Mini Tissues in 3D
Traditional flat (2D) cell cultures and animal models often fail to capture these complex interactions. The authors describe a new generation of 3D systems that better resemble real tissue. Spheroids are simple cell clusters that self-assemble into tiny spheres; scaffolds are engineered materials that provide a 3D support for cells to grow on; organoids are miniature, self-organizing versions of tissues; and assembloids combine multiple organoids or spheroids into composite “mini-organs.” Researchers are now using these 3D approaches to co-culture fat cells and cancer cells, study how obesity-like conditions change their behavior, and test how drugs might disrupt the harmful dialogue between them.
Promise and Current Gaps
Each model offers trade-offs. Spheroids are cheap and high-throughput but relatively simple. Scaffolds allow careful control of physical properties such as stiffness, which can influence cancer spread, but are more technically demanding. Organoids and assembloids capture the highest level of realism, with multiple interacting cell types, yet they are costly, hard to standardize, and often lack full blood vessel networks or immune components. The review emphasizes that today’s systems still fall short of perfectly reproducing human fat and tumor tissue, particularly in replicating the full extracellular matrix, mature fat cells, and immune cell participation.
Where This Research Is Heading
The authors conclude that improving 3D models of fat–cancer interactions is essential for more accurate drug testing and for uncovering how obesity promotes cancer. As these “mini-tissues” become more lifelike—combining realistic fat, tumor cells, blood vessels, and immune cells—they could help researchers predict which treatments will work in specific patients, design better strategies to block cancer’s use of body fat as fuel, and ultimately guide therapies that target not just tumors, but the unhealthy environments that help them thrive.
Citation: Strusi, G., Suelzu, C.M. & Stebbing, J. Modelling adipose tissue-cancer crosstalk: a three-dimensional perspective. Oncogene 45, 905–913 (2026). https://doi.org/10.1038/s41388-026-03697-w
Keywords: adipose tissue, obesity, cancer microenvironment, 3D cell culture, tumor metabolism