Clear Sky Science · en
YEATS2 promotes DNA repair and induces anoikis resistance by enhancing chromatin accessibility to drive prostate cancer metastasis
Why Cancer Cells That Refuse to Let Go Matter
When cancer spreads from its original site to distant organs, it becomes far harder to treat. Prostate cancer is no exception: once it has seeded new tumors in lymph nodes or bone, survival drops sharply. To spread, tumor cells must first detach from their home tissue and enter blood or lymph vessels. Most such wanderers die, but a dangerous few gain the ability to resist this detachment-triggered cell death, a process called anoikis. This study uncovers how a protein named YEATS2 helps prostate cancer cells survive that perilous journey by supercharging their ability to repair DNA damage.
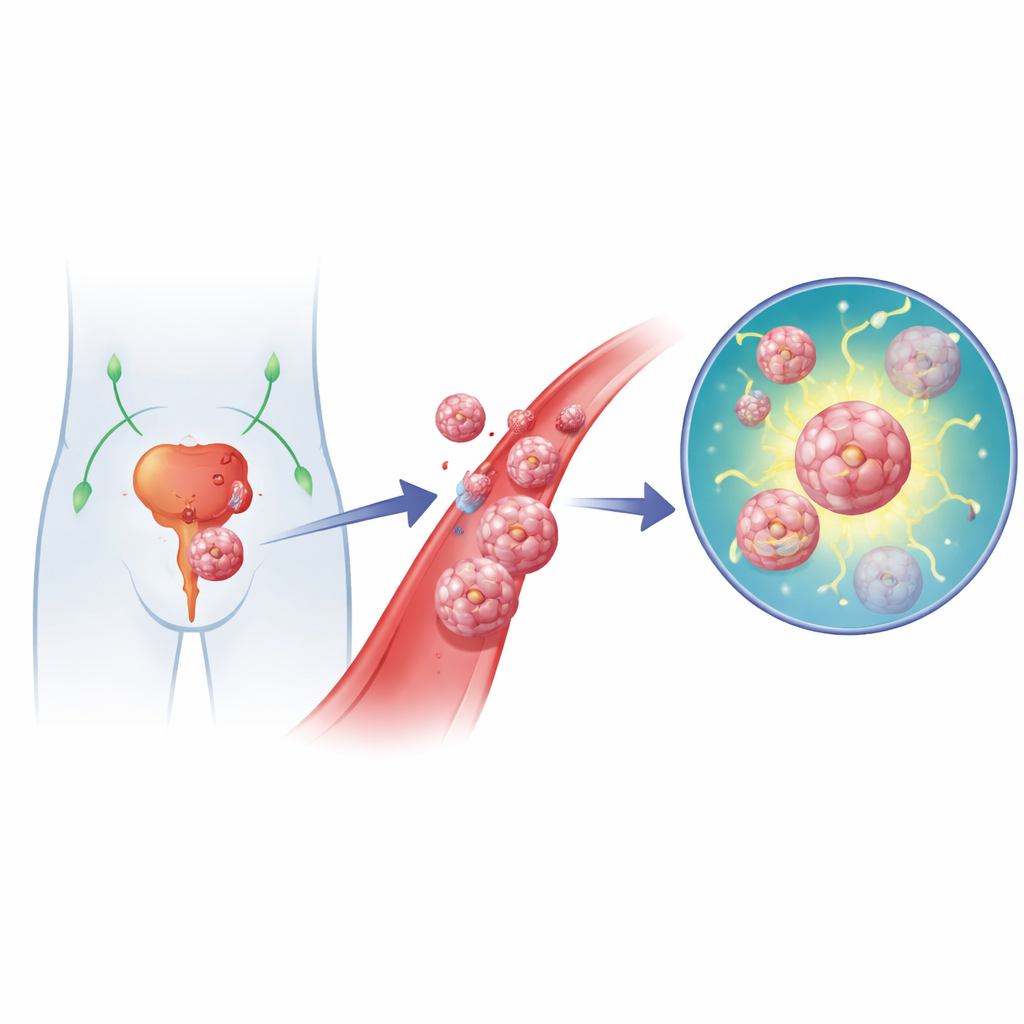
Cells on the Move
The researchers began by asking what makes metastatic prostate tumors different from their primary counterparts. They compared genetic data from public cancer databases with their own experiments on prostate cancer cells that had been pushed to survive without attachment, mimicking conditions in the bloodstream. Across patient samples and lab-grown cells, a small set of genes consistently stood out as altered in metastatic and anoikis-resistant cells. Among them, YEATS2 showed both strong upregulation and a tight link to poor outcomes: men whose tumors had more YEATS2 tended to live for a shorter time and experienced earlier disease progression.
Testing a Suspect
To probe YEATS2’s role, the team built mouse models of lymph node spread by injecting prostate cancer cells into the animals’ footpads and later harvesting both the primary tumors and the nearby popliteal lymph nodes. Cells taken from lymph node metastases were better at surviving in suspension, moving, and invading than their counterparts from the original tumor. These metastatic cells also carried more YEATS2. When the scientists artificially boosted YEATS2 levels in prostate cancer cells, the cells became harder to kill after detachment and more able to migrate and invade in lab tests. Knocking down YEATS2 had the opposite effect and sharply reduced lymph node metastases in mice.
DNA Repair as a Survival Trick
Detaching from the surrounding tissue stresses cancer cells, in part by ramping up harmful molecules that damage DNA. Using markers of DNA breaks, the authors showed that cells floating in suspension accumulate more DNA injuries, especially if they come from primary tumors rather than metastases. Overexpressing YEATS2 lessened this damage, while silencing YEATS2 made it worse and weakened survival in suspension. Further analyses of large patient datasets revealed that genes tied to DNA damage response and repair were strongly associated with YEATS2 activity. One repair gene, RAD50, emerged as a key partner: it was highly expressed in metastatic and anoikis-resistant cells, rose when YEATS2 was increased, and helped restore survival when reintroduced into YEATS2-deficient cells.
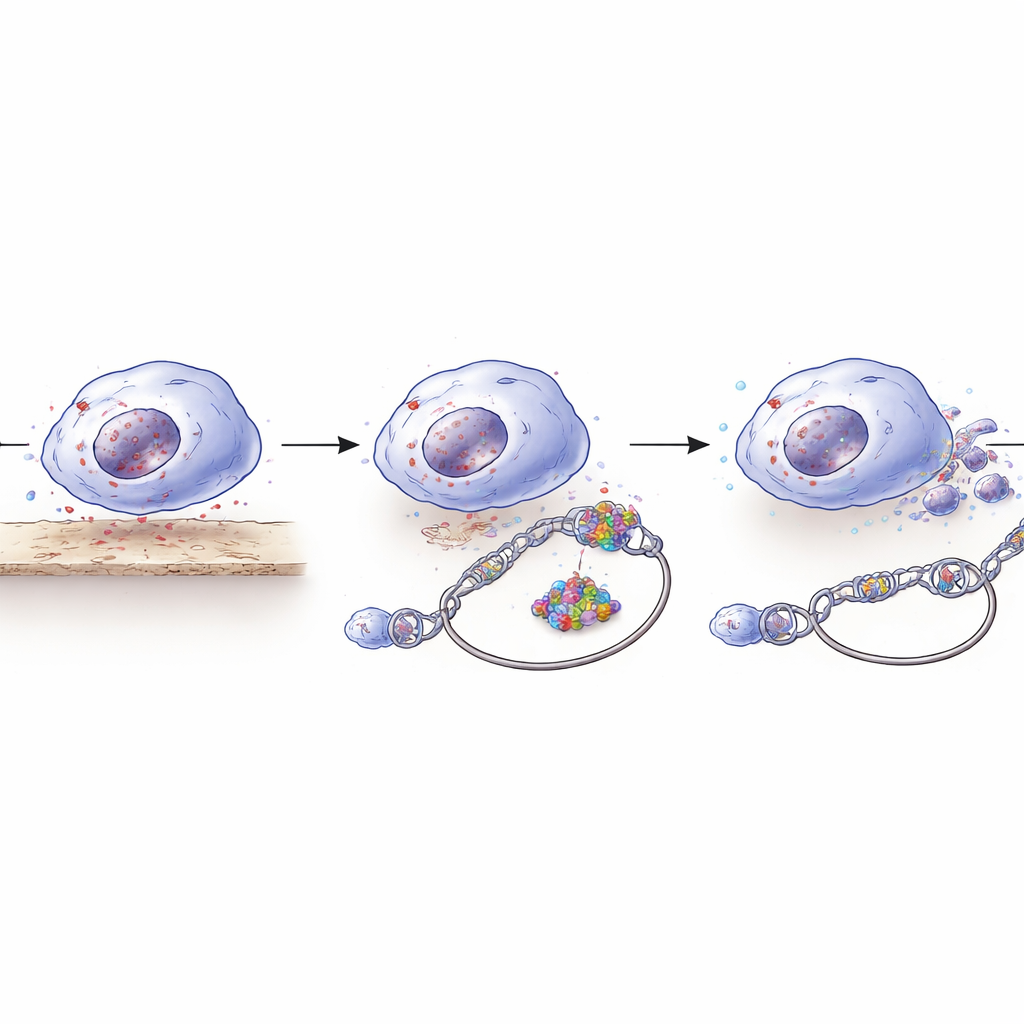
Opening DNA’s Bookmarks
How does YEATS2 turn up RAD50? YEATS2 is part of a protein complex that alters how tightly DNA is packaged, making certain genes easier or harder to read. The team used a technique that maps “open” versus “closed” stretches of DNA and found that YEATS2 made the RAD50 promoter region more accessible. At this loosened segment of DNA, YEATS2 recognized a chemical tag on histone proteins (H3K27 acetylation) and, through its specialized YEATS domain, helped attract additional acetyl marks at nearby positions. These changes acted like opening a bookmarked page and highlighting a paragraph, allowing the transcription factor NR2C2 to bind more efficiently and drive RAD50 production. Together, YEATS2 and NR2C2 formed a pro-survival circuit that boosted repair capacity just when detached cancer cells needed it most.
Blocking the Escape Route
Because RAD50 is part of the MRN complex, a core machine for fixing dangerous DNA double-strand breaks, the scientists explored whether disrupting this machinery could blunt metastasis. They used Mirin, a small molecule that inhibits the MRN complex, and also lowered levels of one of RAD50’s partners, MRE11. Both strategies increased DNA damage and reduced the ability of prostate cancer cells to colonize the lungs or lymph nodes in mice, even when RAD50 was abundant. These findings suggest that metastatic prostate cancer relies heavily on reinforced DNA repair to survive detachment and travel.
What This Means for Patients
In everyday terms, this work shows that some prostate cancer cells evade death by learning to patch up their DNA more effectively during the most dangerous phase of their journey: when they have broken away from their original home. YEATS2 acts as a molecular facilitator, opening the right pages of the genetic instruction book so that RAD50 and its repair partners can be produced in high amounts. This enhanced repair allows rogue cells to ride the bloodstream or lymphatic system without dying, increasing the chances they will seed new tumors. Targeting the YEATS2–NR2C2–RAD50 pathway, or the MRN repair complex it feeds, could therefore offer new ways to stop prostate cancer from spreading, potentially improving outcomes for men with advanced disease.
Citation: Li, H., Song, Y., Cong, Y. et al. YEATS2 promotes DNA repair and induces anoikis resistance by enhancing chromatin accessibility to drive prostate cancer metastasis. Oncogene 45, 971–988 (2026). https://doi.org/10.1038/s41388-026-03696-x
Keywords: prostate cancer metastasis, DNA repair, anoikis resistance, chromatin accessibility, RAD50