Clear Sky Science · en
Therapeutic optimization of LIPA targeting to induce endoplasmic reticulum stress and cell death in ovarian cancer
Why stressing cancer cells could save lives
Ovarian cancer is one of the deadliest cancers affecting women, in part because it is often diagnosed late and comes back after treatment. This study explores a surprising weakness shared by many ovarian tumors: they live under constant internal “stress” as they churn out proteins to fuel rapid growth. The researchers report a new drug-like compound, ERX-208, designed to push this stress over the edge—selectively killing ovarian cancer cells while largely sparing healthy tissue.
Turning a hidden weakness into a target
Inside every cell, a structure called the endoplasmic reticulum acts like a quality-control factory for newly made proteins. Cancer cells, which divide quickly and harbor many mutations, overload this factory and operate close to their stress limit. Normally, cells cope by switching on a protective program known as the unfolded protein response. But if stress becomes too intense or too long-lasting, this same system can flip into a self-destruct mode and trigger cell death. The authors reasoned that a drug that further increases this built‑in stress specifically in ovarian tumor cells could become a powerful and broadly useful treatment.
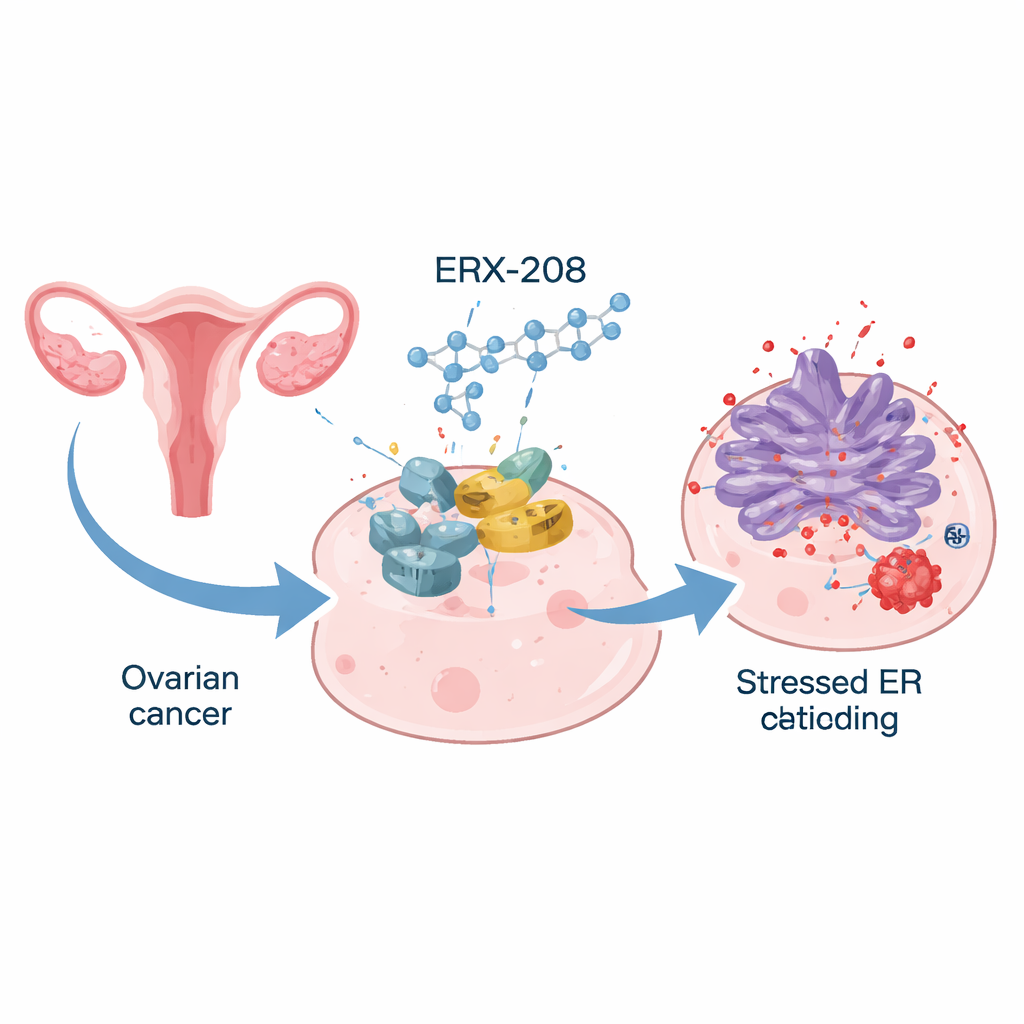
Designing a sharper anti-cancer tool
The team had previously discovered a small molecule called ERX-41 that binds to a protein named LIPA and, in doing so, increases stress inside tumor cells. However, ERX-41 was not potent enough for optimal clinical use. In the new work, chemists systematically redesigned parts of the ERX-41 molecule, creating a library of related compounds and testing how small structural tweaks influenced their ability to halt cancer cell growth. This effort led to ERX-208, which keeps the original three-ring “spine” of ERX-41 but adds a larger, more complex chemical group that grips LIPA more effectively. Laboratory tests showed that ERX-208 was about five times more powerful than its predecessor at stopping the growth of ovarian cancer cells.
Forcing cancer cells into fatal overload
When ovarian cancer cells from all major subtypes were treated with ERX-208, they not only stopped dividing, they also underwent orderly cell death through a process called apoptosis. The compound had strong effects across 23 different cancer cell models, including cells freshly obtained from patients’ tumors and abdominal fluid, while showing little harm to normal ovarian surface cells. Detailed molecular analyses revealed a consistent pattern: ERX-208 switched on multiple branches of the cell’s stress-response network and dialed down genes that drive the cell cycle. Under the microscope, the protein-folding factory inside treated cells became swollen and distorted—visual evidence that the internal machinery was being overwhelmed.
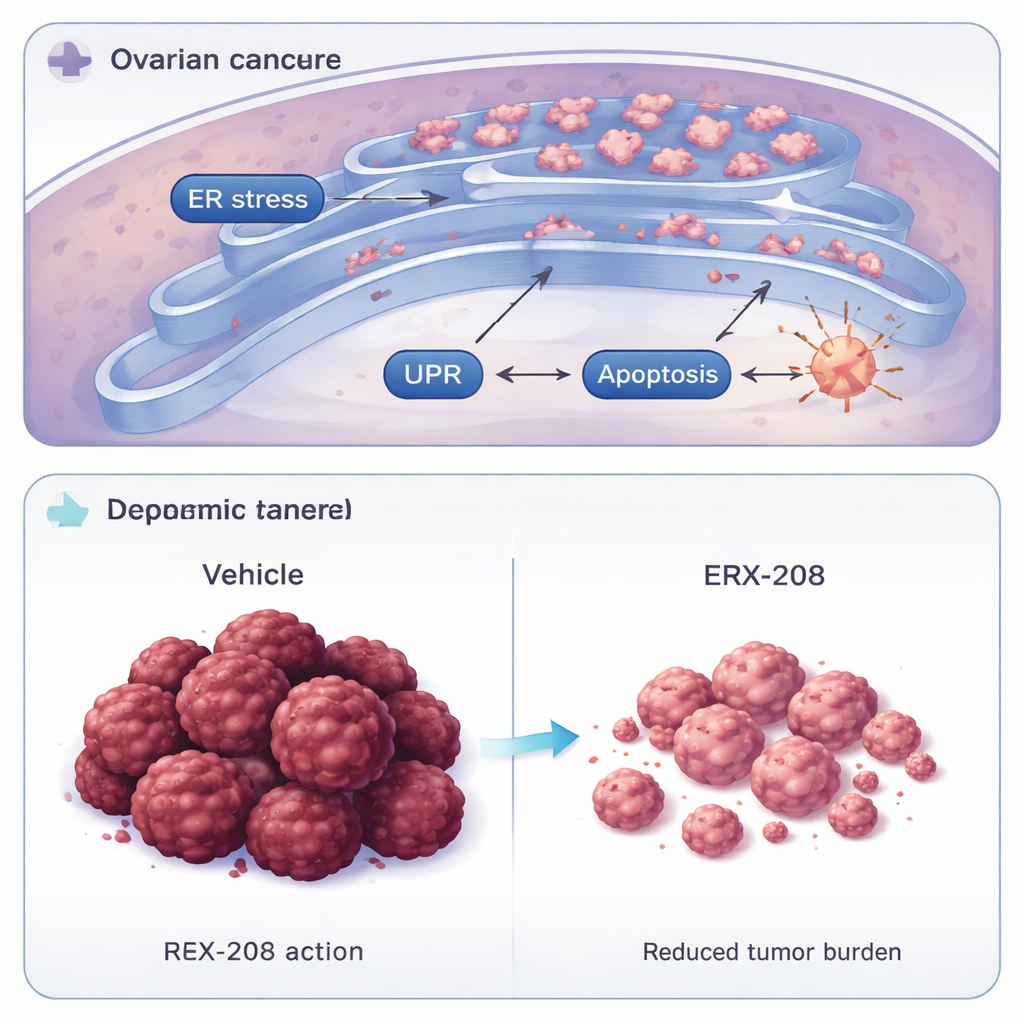
Zeroing in on the LIPA switch
To confirm how ERX-208 works, the researchers removed the LIPA protein from cancer cells and found that these cells became largely resistant to the drug and failed to turn on stress markers. By comparing computer models of how ERX-41 and ERX-208 dock onto LIPA, then altering individual amino acids in the protein, they mapped out a broader contact surface for ERX-208. Certain mutations in LIPA disrupted the action of ERX-208 but not ERX-41, suggesting that the new compound achieves its superior potency by engaging LIPA more extensively and precisely. This tight, selective interaction helps explain why ERX-208 strongly stresses cancer cells that depend on LIPA while sparing many normal cells.
Promising results in realistic tumor models
The scientists then moved beyond cells in a dish to more realistic models that mimic human disease. In mice carrying tumors grown from ovarian cancer cell lines or directly from patient tumors, ERX-208 shrank established tumors, slowed the spread of cancer within the abdomen, and reduced the number of metastatic nodules. These benefits were seen at doses that did not cause measurable weight loss, organ damage, or disruption of normal antibody‑producing immune cells. Drug measurements showed that ERX-208 reached especially high levels in the ovary and liver—organs central to the disease and drug processing—while remaining low in the brain, an encouraging safety feature.
What this could mean for future treatment
Overall, the study introduces ERX-208 as a next‑generation compound that exploits a common pressure point in ovarian cancer: dependence on a stressed protein-folding system controlled through LIPA. By pushing this system past its tipping point, ERX-208 triggers self‑destruction in tumors drawn from diverse patients and subtypes, yet appears well tolerated in animal tests. While much work remains before it can be tried in people—including larger safety studies and eventual clinical trials—the findings suggest that deliberately “stressing” cancer cells through LIPA could become a new class of therapy for ovarian cancer and potentially other tumors that share this hidden vulnerability.
Citation: Viswanadhapalli, S., Lee, TK., Elmore, S. et al. Therapeutic optimization of LIPA targeting to induce endoplasmic reticulum stress and cell death in ovarian cancer. Oncogene 45, 790–804 (2026). https://doi.org/10.1038/s41388-026-03689-w
Keywords: ovarian cancer, endoplasmic reticulum stress, LIPA, ERX-208, targeted therapy