Clear Sky Science · en
POM121 O-GlcNAcylation facilitates bone metastasis in non-small cell lung cancer through enhanced c-MYC nuclear import and ECM reprogramming
Why this research matters
When lung cancer spreads to the bones, it causes severe pain, fractures, and sharply shorter survival, yet doctors still have few tools to stop this deadly step. This study digs into the inner workings of lung cancer cells to uncover how some of them gain a special advantage that lets them travel from the lung to bone and thrive there. By tracing this process down to tiny changes on a single gatekeeping protein at the edge of the cell nucleus, the authors identify a new chain of events that could be targeted to better predict, prevent, or treat bone metastases in patients with non-small cell lung cancer.
A sugar tag that changes cancer behavior
The researchers focused on post-translational modifications, tiny chemical changes that proteins acquire after they are made. One such change, called O-GlcNAcylation, is the attachment of a small sugar to specific spots on proteins. Using lung cancer cell lines and mouse models, the team compared ordinary tumor cells with subpopulations that had been repeatedly selected for their ability to colonize bone. They found that these bone-seeking cells had much higher overall levels of O-GlcNAcylation, driven by increased activity of the enzyme OGT, which installs the sugar tag. Among hundreds of modified proteins, one stood out: POM121, a core component of the nuclear pore, the structure that controls traffic in and out of the cell nucleus.
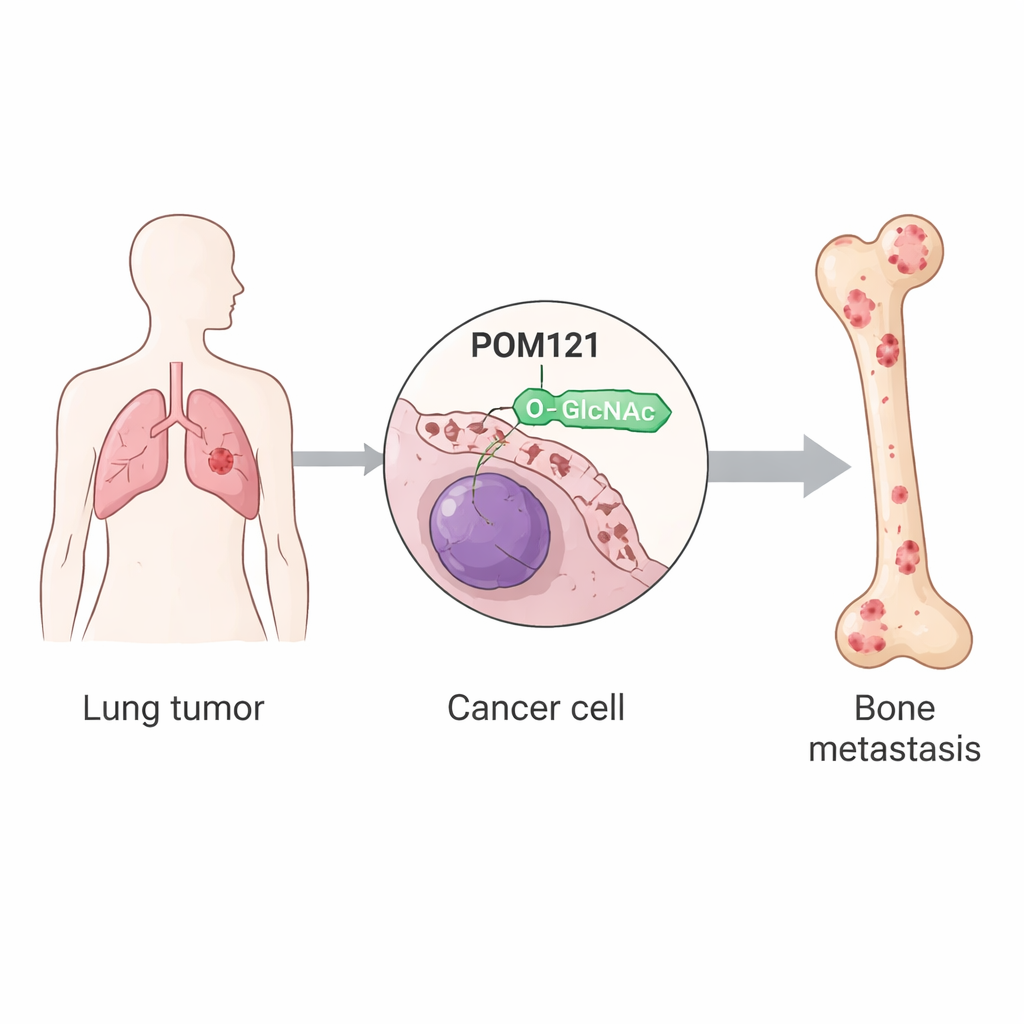
The nuclear gatekeeper POM121 is stabilized
POM121 sits in the membrane that surrounds the nucleus, forming part of the channel through which many signaling proteins must pass. In bone-metastatic lung cancer cells, POM121 carried a heavy load of O-GlcNAc at a single amino acid position called serine 199. When this site was changed so it could no longer be modified, the protein became unstable and was rapidly broken down by the cell’s disposal machinery. The study shows that the sugar tag protects POM121 from being marked for destruction by an enzyme called TRIM21, which normally attaches ubiquitin “disposal” labels. With O-GlcNAc in place, TRIM21 cannot bind well, ubiquitination falls, and POM121 accumulates at the nuclear pore without changing its location.
Opening the door for a powerful cancer driver
What difference does extra-stable POM121 make? The key lies in a famous cancer-promoting protein, the transcription factor c-MYC. c-MYC must enter the nucleus to switch genes on, but its passage depends on nuclear pore components like POM121. The authors showed that when POM121 was abundant and O-GlcNAcylated, more c-MYC was found inside the nucleus; when POM121 levels were reduced or its sugar tag was removed, c-MYC nuclear import dropped sharply. In mice, lung cancer cells carrying the non-modifiable POM121 mutant formed far fewer and smaller bone metastases, while restoring c-MYC in POM121-deficient cells rescued much of their bone-colonizing power. This places POM121 upstream of c-MYC in a critical metastatic pathway.
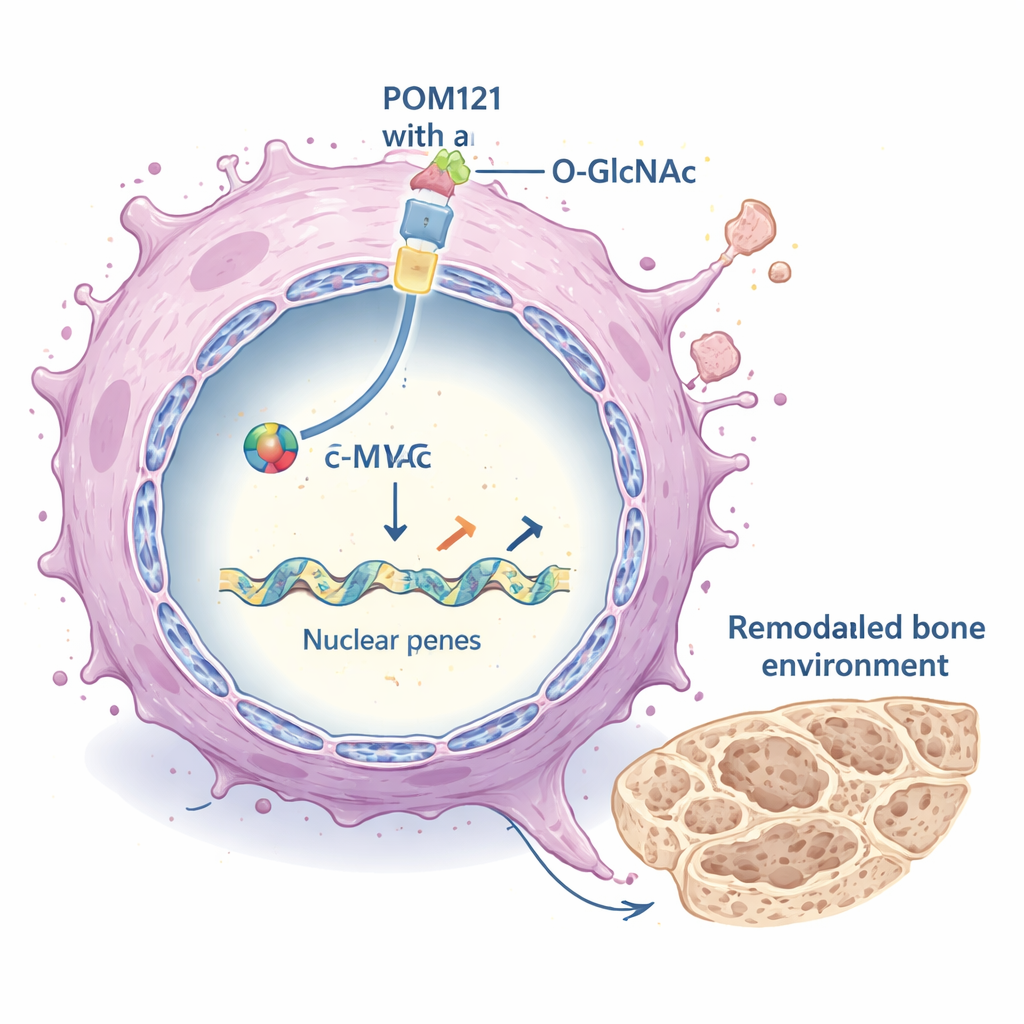
Rewiring the tumor’s surroundings
Once inside the nucleus, c-MYC acts as a master switch for many genes. Here, it was found to strongly activate a set of genes that shape the extracellular matrix (ECM) – the scaffold of proteins surrounding cells. These ECM genes, which include various collagens and enzymes that remodel tissue structure, were dialed down when POM121 or c-MYC were blocked, and dialed up when O-GlcNAcylation and POM121 were high. The altered ECM in turn boosted major growth pathways inside the cancer cells, notably the PI3K–AKT–mTOR signaling cascade, which supports survival, growth, and spread. Similar patterns appeared across several different lung cancer models, suggesting this mechanism is not limited to a single cell line.
From lab discovery to patient impact
Looking at patient data, the researchers found that high levels of OGT, POM121, and c-MYC, as well as the ECM genes they regulate, were linked with worse outcomes and with lung tumors that had already spread to bone. Taken together, the work outlines an OGT–POM121–c-MYC–ECM axis: extra O-GlcNAcylation stabilizes the nuclear gatekeeper POM121, which ferries more c-MYC into the nucleus, which then rewires the tissue scaffold and growth signals to favor bone metastasis. For patients, this suggests new types of biomarkers to flag those at risk for bone spread, and new therapeutic angles – from blocking OGT or POM121 function to dampening c-MYC or ECM remodeling – that could one day help keep lung cancer from reaching the skeleton.
Citation: Ren, YZ., Zhao, MN., Du, FL. et al. POM121 O-GlcNAcylation facilitates bone metastasis in non-small cell lung cancer through enhanced c-MYC nuclear import and ECM reprogramming. Oncogene 45, 728–743 (2026). https://doi.org/10.1038/s41388-026-03687-y
Keywords: lung cancer bone metastasis, POM121, O-GlcNAcylation, c-MYC, extracellular matrix