Clear Sky Science · en
Disruption of androgen receptor-cofactor interactions by the RNA-binding protein FUS/TLS alters androgen signalling in prostate cancer
Why this research matters for men's health
Prostate cancer depends heavily on male hormones, called androgens, and on a protein that senses them, the androgen receptor. Most modern treatments work by cutting off these hormones or blocking this receptor. Yet many tumors eventually escape control and return in a harder‑to‑treat form. This study uncovers how another protein, FUS/TLS, can act as a powerful brake on androgen signaling in early prostate cancer—and how that brake seems to fail as the disease becomes more aggressive.
A traffic controller for hormone signals
The androgen receptor acts like a molecular switch. When androgens bind to it, the receptor moves into the cell’s nucleus, latches onto DNA, and turns on genes that help prostate cells grow. It does not work alone: helper proteins can boost or dampen its activity. The authors focused on FUS/TLS (called FUS), a protein known for roles in RNA handling and gene control, and previously linked to slower tumor growth. Using prostate cancer cells grown in the lab, they showed that many of the genes controlled by FUS overlap with genes controlled by the androgen receptor, hinting that FUS directly shapes how hormone signals are translated into growth instructions.
How FUS puts the brakes on tumor‑driving genes
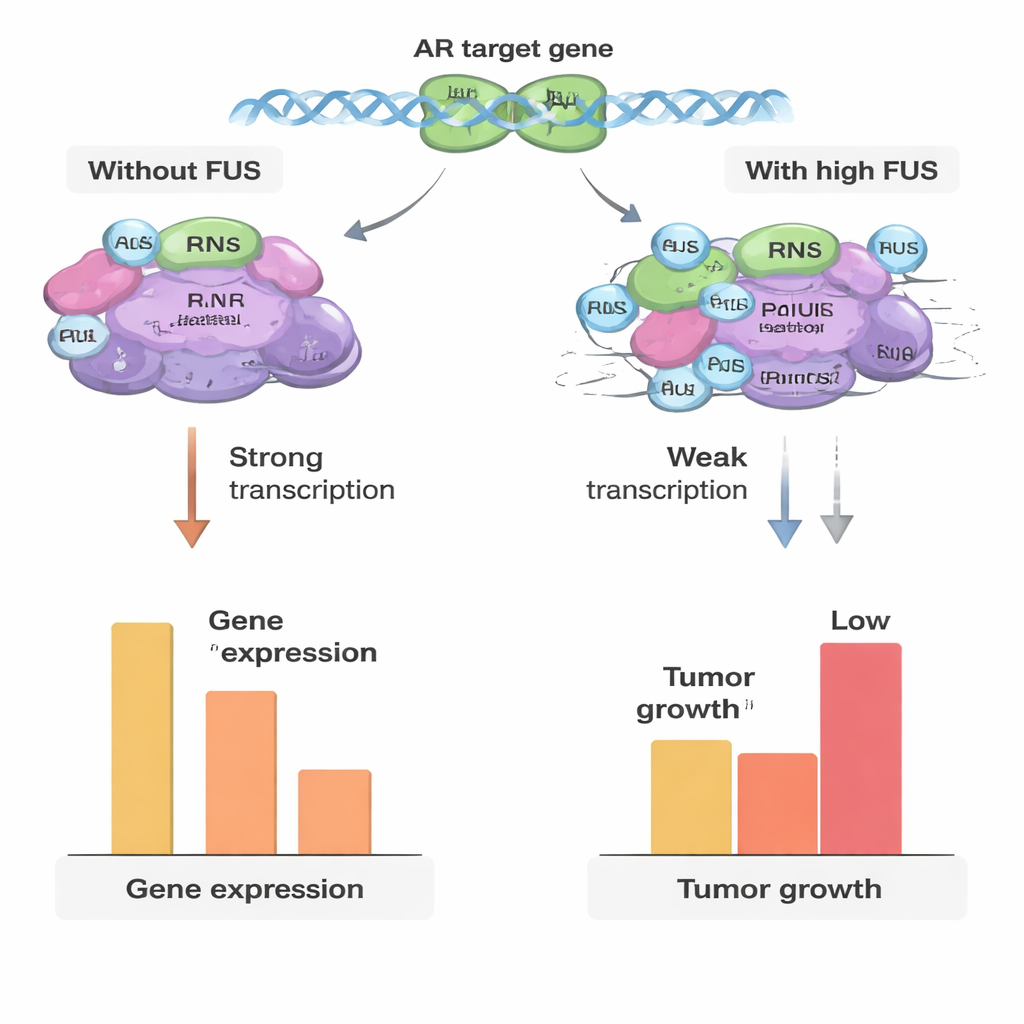
To see what FUS does, the team artificially raised or lowered its levels in prostate cancer cells and tracked which genes changed in response to a potent androgen‑like drug. They found that FUS can push androgen‑controlled genes in both directions, sometimes enhancing and sometimes weakening their response. However, the dominant effect was repression: classic androgen‑stimulated genes such as PSA (KLK3) and TMPRSS2 were turned down when FUS was high and turned up when FUS was removed. Reporter tests—simple readouts of androgen receptor activity—confirmed that increasing FUS cut receptor activity by roughly half to two‑thirds across several cell types.
A physical block on the hormone machinery
The scientists then asked how FUS exerts this control. Microscopy and interaction tests showed that FUS and the androgen receptor meet each other inside the nucleus and physically bind. FUS attaches to a specific region of the receptor through its RNA‑recognition domain. It also binds to several key helper proteins that would normally boost receptor activity. Detailed experiments at a well‑known androgen‑responsive gene revealed that FUS does not stop the receptor from grabbing DNA; instead, it prevents the full team of helpers and the basic transcription machinery from assembling there. In other words, FUS leaves the “switch” on the DNA but strips away the parts needed to fully turn up the volume of gene expression.
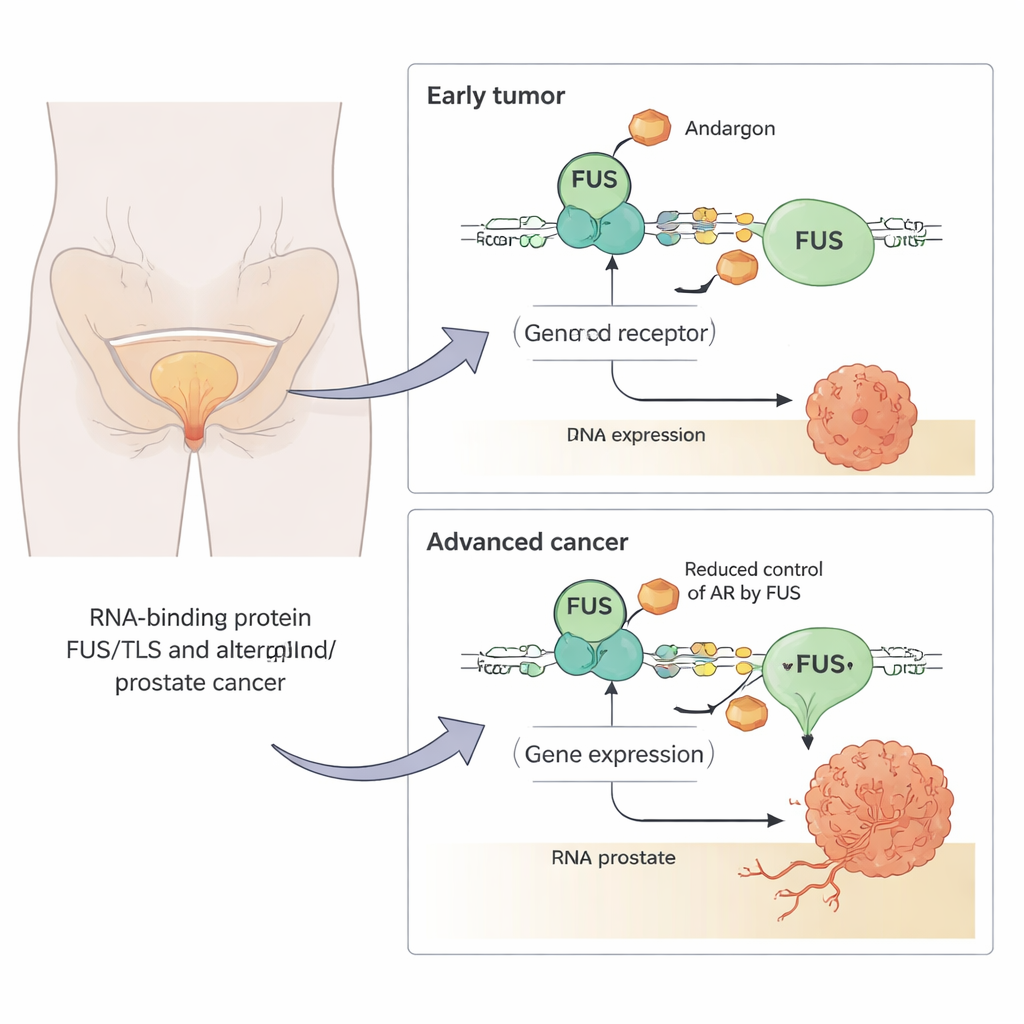
A shifting role as cancer advances
Because treatment resistance in prostate cancer often involves changes in these helper proteins, the authors examined FUS levels in cell lines and in patient samples spanning benign tissue, primary tumors, and advanced castration‑resistant prostate cancer (CRPC). In early tumors, FUS protein levels were generally lower than in benign samples, consistent with the idea that tumors down‑regulate this natural brake to grow more easily. Surprisingly, in advanced CRPC, FUS levels rose again and were higher than in primary tumors. Yet in a CRPC‑like cell model, boosting FUS no longer strongly altered androgen‑dependent genes, even though it still slowed overall cell growth. This suggests that as the cancer evolves, the androgen network is rewired so that FUS loses much of its grip on the receptor, while possibly acting through other growth‑related pathways.
What this means for future treatments
For non‑specialists, the key message is that FUS behaves like a built‑in safety device that normally keeps male‑hormone signals from over‑driving prostate cells. In early cancer, lowering FUS helps tumors grow; in advanced disease, the hormone system changes enough that FUS can be abundant but no longer effectively restrains the androgen receptor. Understanding exactly how this brake works—and why it fails—could inspire new strategies that either restore its control or mimic its ability to disarm the hormone‑driven growth machinery, potentially improving therapies for men with advanced prostate cancer.
Citation: Brooke, G.N., Leach, D.A., Culley, R.L. et al. Disruption of androgen receptor-cofactor interactions by the RNA-binding protein FUS/TLS alters androgen signalling in prostate cancer. Oncogene 45, 757–773 (2026). https://doi.org/10.1038/s41388-026-03682-3
Keywords: prostate cancer, androgen receptor, hormone therapy resistance, RNA-binding proteins, FUS TLS