Clear Sky Science · en
Sortilin exhibits tumor suppressor-like activity by limiting EGFR transduction function
Why this matters for cancer treatment
Lung cancer is the leading cause of cancer deaths worldwide, and many modern drugs target a molecule called EGFR that drives tumor growth. These drugs often work well at first, but tumors commonly become resistant. This study uncovers a natural "brake" inside lung cancer cells—an unexpected role for a protein called sortilin—that can tone down EGFR’s cancer‑promoting signals. Understanding this relationship could help explain why some patients respond better to EGFR‑targeted therapies and suggest new ways to keep those treatments working longer. 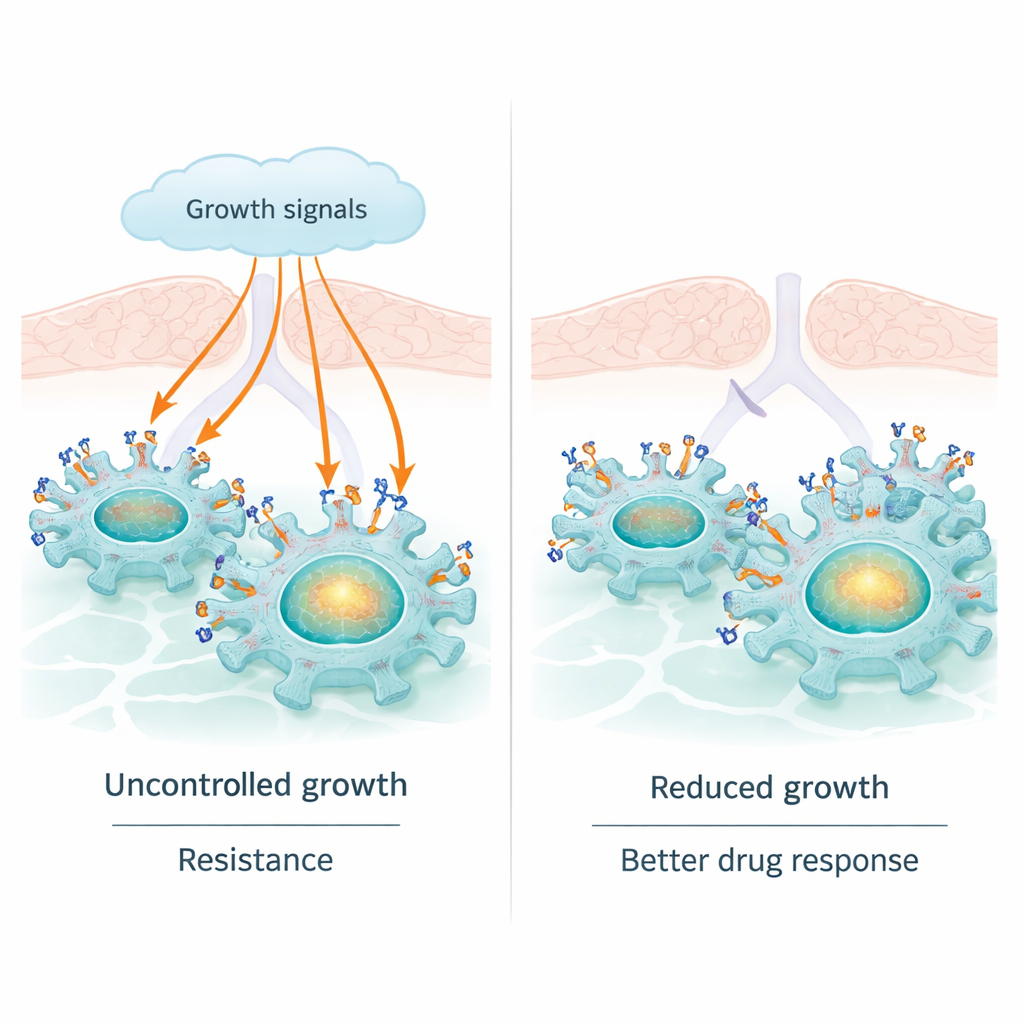
A growth switch gone into overdrive
EGFR is a receptor sitting on the surface of many cells, where it senses growth signals from the environment. In a large fraction of lung adenocarcinomas, EGFR is altered so that it is permanently switched on. This fuels constant growth and division, and it is why EGFR‑blocking pills, called tyrosine kinase inhibitors, can shrink tumors. But EGFR has another, less appreciated side: it can travel into the cell nucleus and act directly on DNA, turning on genes such as cMYC and CCND1 that further boost growth, survival, and treatment resistance. This nuclear role of EGFR may help tumors escape even when its surface activity is chemically blocked.
Sortilin steps into the nucleus
Sortilin was previously known mainly as a sorting protein that helps internalize EGFR from the cell surface and send it toward degradation, thereby weakening its growth signals. In this work, the authors show that sortilin also follows EGFR into the nucleus when lung cancer cells are stimulated. Using high‑resolution microscopy and biochemical fractionation, they observed complexes of EGFR and sortilin forming inside nuclei soon after activation. Importantly, when EGFR could not enter the nucleus, sortilin stayed out as well, indicating that EGFR acts as sortilin’s “ticket” into this compartment. Once there, EGFR–sortilin complexes were found bound to stretches of DNA close to the start sites of key growth‑control genes, including cMYC and CCND1.
A built‑in brake on cancer genes
To understand what these complexes do, the researchers mapped their binding across the genome and measured changes in gene activity. They found that EGFR alone tends to promote the activation of growth‑related genes, while the presence of sortilin at the same DNA regions dampens this effect. When sortilin levels were artificially reduced, EGFR binding at target genes increased, and cancer‑driving genes such as cMYC and CCND1 became more active. In contrast, boosting sortilin levels had the opposite effect: EGFR’s grip on DNA loosened, recruitment of the transcription machinery dropped, and the expression of these oncogenic genes fell. In mouse models, lung tumors engineered to overproduce sortilin grew more slowly and showed reduced levels of cMYC and CCND1. 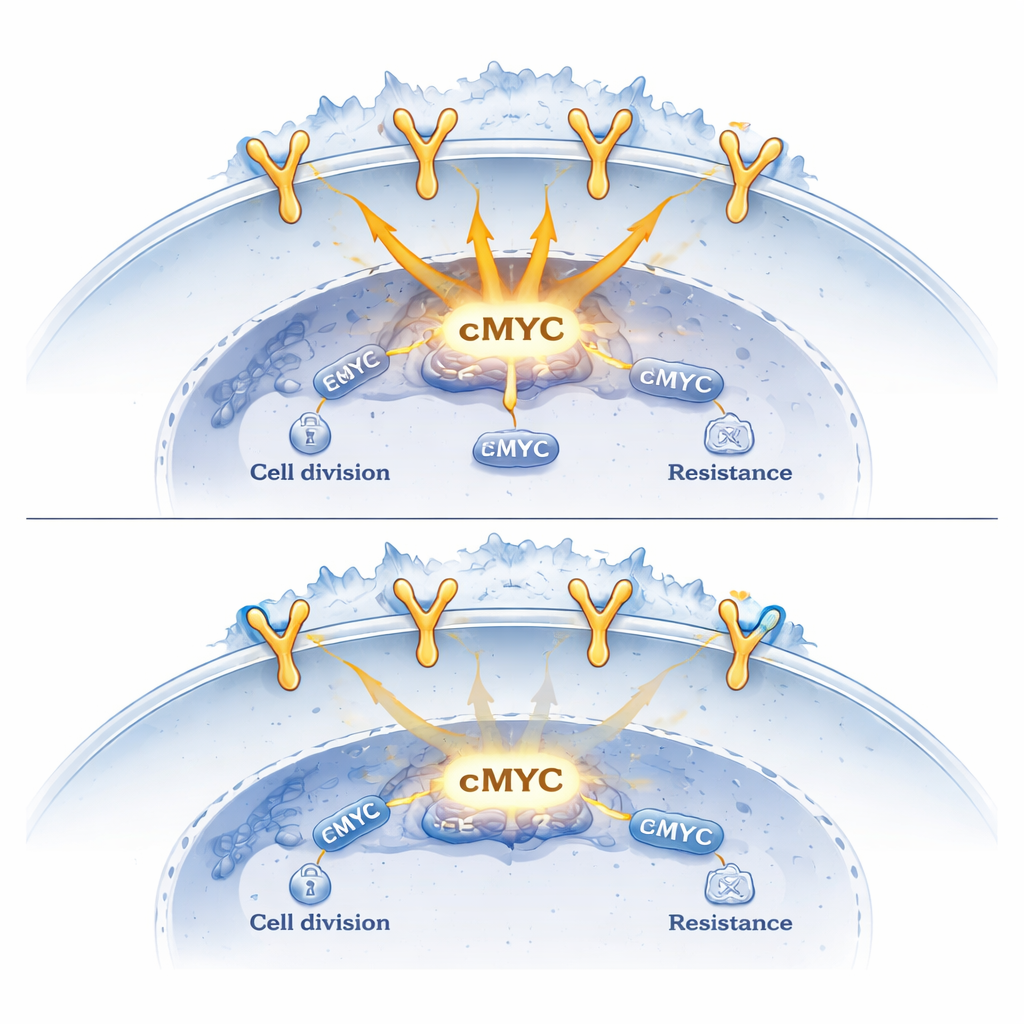
Interplay with EGFR drugs
The study also explored how this mechanism interacts with osimertinib, a widely used EGFR‑targeted drug designed to block troublesome EGFR mutations. Surprisingly, even when osimertinib shut down EGFR’s enzyme activity, the receptor still moved into the nucleus. There, it continued to bind DNA, especially near the cMYC gene. Sortilin, however, also accumulated in the nucleus under these conditions. When sortilin was abundant, cMYC levels dropped more strongly in response to osimertinib, and cancer cells became less viable. Analysis of patient tumors and large public cancer datasets showed that sortilin levels are often lower in lung adenocarcinomas than in normal lung tissue, and that higher sortilin expression is consistently linked to lower cMYC expression.
What this means for patients
Viewed together, the findings paint sortilin as a kind of tumor‑suppressor‑like partner that rides with EGFR into the nucleus and then restrains it. By limiting EGFR’s ability to switch on powerful growth genes such as cMYC, sortilin can slow tumor progression and enhance the impact of EGFR‑blocking drugs. Because many lung tumors have reduced sortilin, they may lack this internal brake, helping explain why some cancers are particularly aggressive or quickly become drug‑resistant. Measuring sortilin levels in tumors could help predict how well a patient will respond to EGFR‑targeted therapies, and strategies that raise sortilin activity might offer a new way to keep EGFR‑driven cancers under better long‑term control.
Citation: Lapeyronnie, E., Granet, C., Tricard, J. et al. Sortilin exhibits tumor suppressor-like activity by limiting EGFR transduction function. Oncogene 45, 875–888 (2026). https://doi.org/10.1038/s41388-026-03680-5
Keywords: lung cancer, EGFR, sortilin, cMYC, targeted therapy resistance