Clear Sky Science · en
Synaptic effects of interleukin-6 on human iPSC-derived dopaminergic neurons
Why Inflammation Matters for Mood
Many people with depression also show signs of low-grade inflammation in their blood, and these immune changes are strongly linked to symptoms like loss of pleasure, fatigue, and slowed movement. This study asks a focused question: how does one key inflammatory signal, a molecule called interleukin‑6, directly affect human brain cells that use dopamine, the chemical that helps drive motivation and reward? By growing human dopamine‑producing neurons from stem cells in the lab, the researchers could watch how these cells respond to inflammation and whether that response differs between females and males.
From Blood Signals to Brain Motivation
Depression is common, disabling, and often stubbornly resistant to standard antidepressants, especially in people whose blood tests show high inflammation. Previous work in humans and animals has shown that inflammatory triggers can dampen activity in the brain’s reward circuitry, particularly in a region called the ventral striatum that relies heavily on dopamine. When inflammatory molecules are experimentally increased, people and animals typically show less drive to work for rewards. Interleukin‑6 is one such molecule, and it can reach dopamine neurons in the brain under stress. Yet the precise ways it alters dopamine‑releasing cells have remained unclear, especially in human neurons and across sexes.
Building Human Dopamine Neurons in a Dish
To get at these mechanisms, the team reprogrammed skin or blood cells from healthy men and women into induced pluripotent stem cells and then guided them to become midbrain dopamine neurons, the type involved in motivation. After about eight weeks of maturation, these neurons had the expected markers and electrical activity. The scientists then exposed the cells to interleukin‑6 for 24 hours and measured three key features: how much dopamine they released, how often they fired electrical impulses, and how efficiently tiny dopamine‑filled packages called synaptic vesicles moved and docked at the nerve ending to release their cargo.
Different Stories in Female and Male Cells
The results revealed a striking split between female‑ and male‑derived neurons. In cells from women, interleukin‑6 clearly weakened the dopamine system: dopamine release dropped, electrical firing slowed, and synaptic vesicles moved more sluggishly and were less often parked ready to release at the nerve ending. In cells from men, those same inflammatory signals caused only minor slowdowns but triggered what looked like built‑in compensation. Vesicles moved faster and became more numerous at the terminals, and the number of presynaptic contact points increased, changes that could help preserve dopamine output despite inflammation. Gene‑expression analyses supported these functional differences, showing that interleukin‑6 switched on stronger inflammatory programs in female neurons than in male neurons.
A Long Noncoding RNA as a Hidden Switch
Digging deeper, the researchers focused on a regulatory molecule called MIAT, a long noncoding RNA that does not make protein but can fine‑tune gene activity. Male dopamine neurons started out with higher MIAT levels than female neurons, and interleukin‑6 pushed MIAT in opposite directions in the two sexes. When the team used gene editing to delete MIAT in male neurons, the cells lost their protective, compensatory response. After interleukin‑6 exposure, these MIAT‑deficient male neurons now showed reduced dopamine release, slower firing, and fewer docked vesicles—much like female neurons. MIAT also influenced genes that control dopamine recycling and the sensitivity of cells to interleukin‑6, suggesting it acts as a molecular hub that shapes how dopamine neurons weather inflammatory stress.
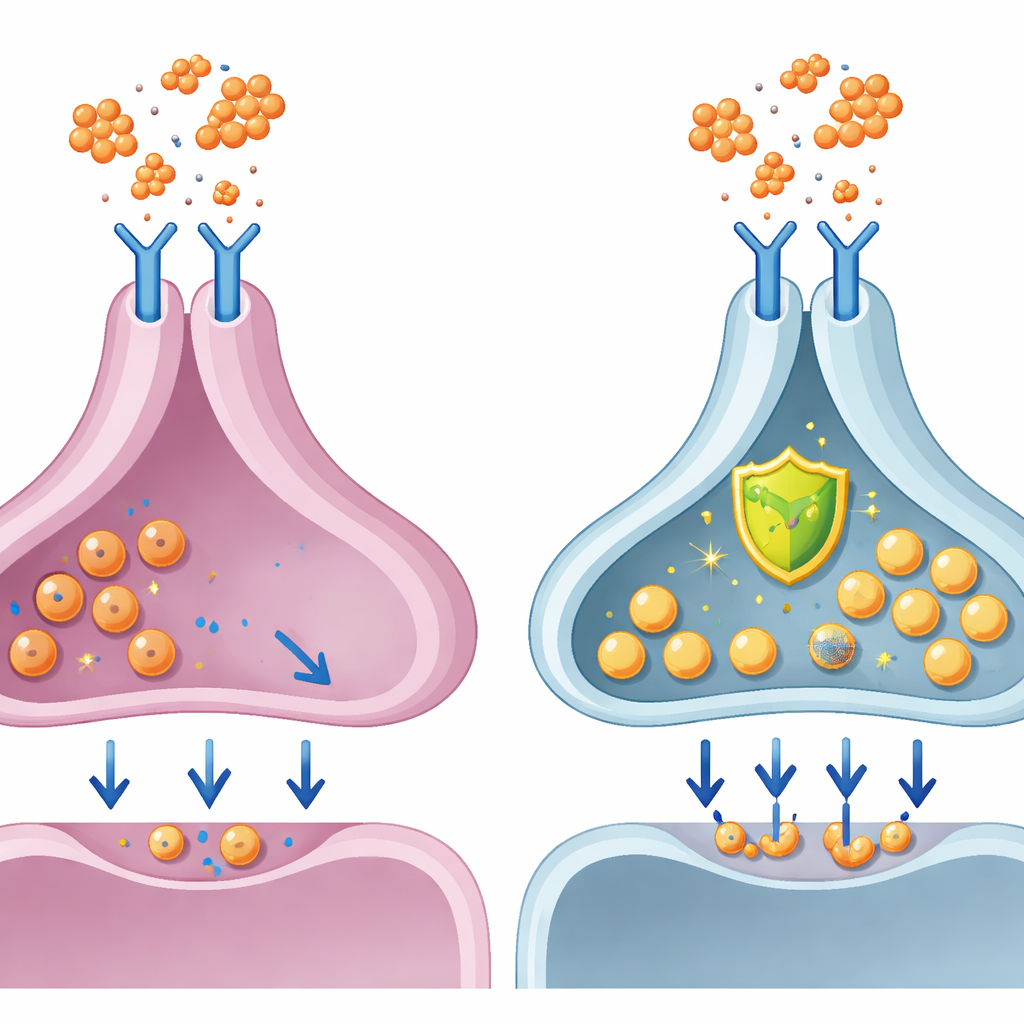
Blocking the Harmful Pathway
Because interleukin‑6 signals through a well‑known chain of proteins called the JAK‑STAT pathway, the scientists tested whether an existing drug that blocks this route could protect dopamine neurons. They added baricitinib, a JAK inhibitor already approved for inflammatory conditions, together with interleukin‑6. In female‑derived dopamine neurons, baricitinib largely reversed the drop in dopamine release, the slowing of electrical activity, and the synaptic vesicle problems. This suggests that targeting this pathway can directly shield human dopamine cells from inflammation‑induced dysfunction, at least in the dish, and may help explain why such drugs have been linked to improvements in mood and motivation in some patients.
What This Means for People with Depression
Taken together, this work shows that inflammatory signals can weaken human dopamine neurons in a sex‑specific way: female neurons tend to show outright damage to dopamine release machinery, while male neurons lean toward adaptive changes that help maintain function. A noncoding RNA, MIAT, appears to be a key part of this difference, and blocking downstream signaling with baricitinib can rescue vulnerable cells. For people, these findings support the idea that inflammation‑targeted treatments could be especially helpful for depressed patients with high inflammatory markers and that sex and individual molecular profiles may matter when choosing such therapies.
Citation: Huang, Y., Michalski, C., Zhou, Y. et al. Synaptic effects of interleukin-6 on human iPSC-derived dopaminergic neurons. Neuropsychopharmacol. 51, 934–945 (2026). https://doi.org/10.1038/s41386-025-02320-y
Keywords: inflammation and depression, dopamine neurons, sex differences, interleukin-6, anti-inflammatory treatments