Clear Sky Science · en
RO4938581, a GABAA-α5 negative allosteric modulator rescued behavioral and EEG phenotypes of a mouse model of Dup15q syndrome
Why this brain research matters
Dup15q syndrome is a rare genetic condition that often causes autism, seizures, and serious learning difficulties. Families have little beyond supportive care, and there is no treatment that tackles the root biology. This study used mice carrying the same extra DNA segment as people with Dup15q to probe what goes wrong in the brain—and to test whether a highly targeted experimental drug can dial that problem back. The work highlights a specific type of brain receptor as a promising, testable target for future precision medicines.
Too much braking in the brain
Our brains rely on a delicate balance between "go" signals that excite neurons and "stop" signals that quiet them down. In Dup15q syndrome, a stretch of chromosome 15 is duplicated. That region includes three genes that build parts of a key "stop" receptor in the brain, called the GABAA receptor, and especially a version containing a component named alpha-5. The researchers engineered mice with the same kind of duplication and found that these animals made about 50% more of the alpha-5–containing receptors in brain regions important for memory, emotion, and movement, including the hippocampus and cortex. Multiple methods—from gene expression to receptor imaging—converged on the same conclusion: this particular brake system is overbuilt in the Dup15q model.
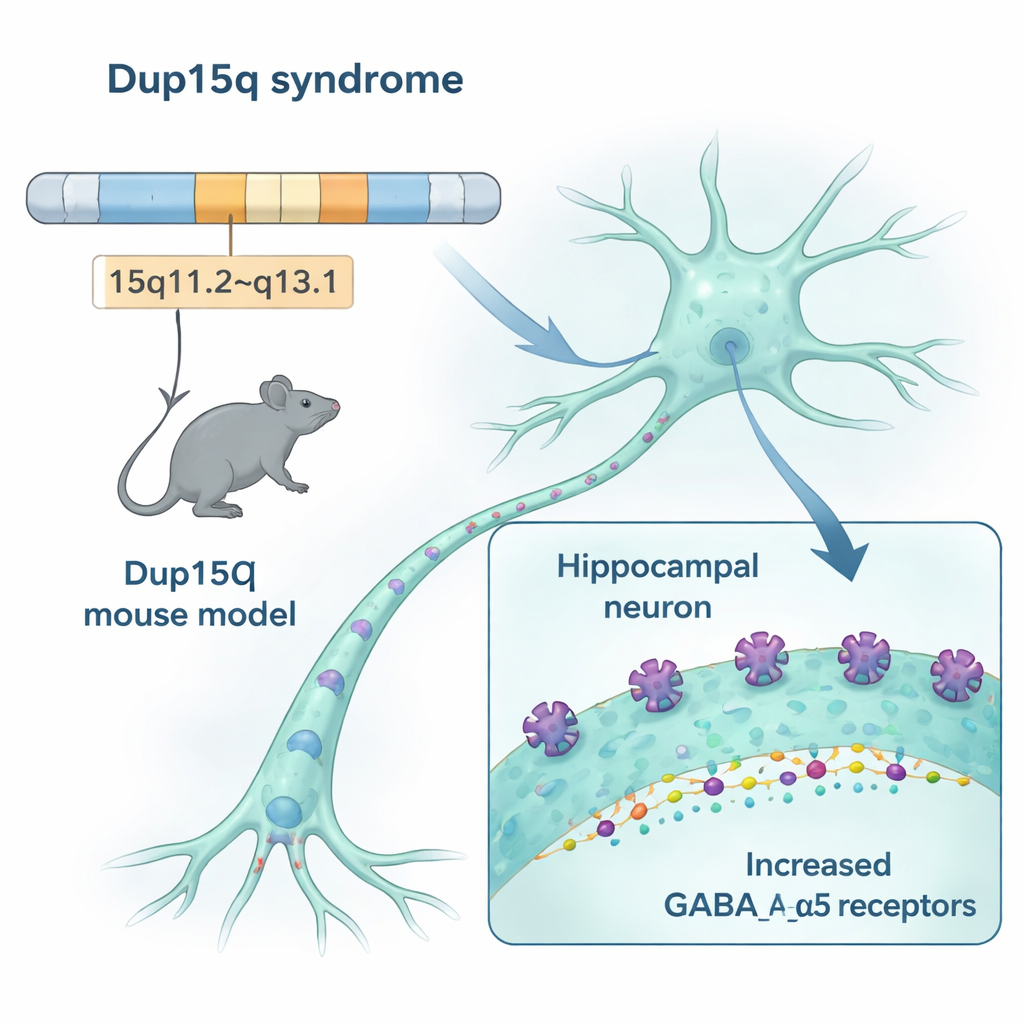
How extra receptors change brain signals
Having more braking receptors is only important if it actually changes how circuits fire. In thin brain slices from the hippocampus, the team measured the tiny electrical currents that inhibitory neurons send onto other cells. In the Dup15q mice, these inhibitory currents came more often, suggesting that neurons experienced a stronger ongoing brake, even though the size of each individual signal was unchanged. A separate test of circuit behavior, called paired-pulse inhibition, also showed that networks in these mice were more strongly damped than those of their healthy littermates. Together, the data indicate that the extra receptors make inhibitory circuits more dominant, shifting the balance away from flexible information processing.
Behavior and brain waves in affected mice
The scientists next asked whether this altered wiring translated into behaviors that echo human symptoms. In a series of tests, the Dup15q mice showed hints of social and communication differences and, most clearly, trouble with cognitive flexibility. In a water maze task, both normal and Dup15q mice learned where a hidden platform was. But when the platform was moved, the Dup15q animals were slow to abandon the old location and adapt to the new one—a laboratory parallel of the rigid routines often seen in autism. The team also recorded electroencephalography (EEG) signals, the summed rhythms of many neurons. As in people with Dup15q, the mice showed unusually strong beta-band activity, a fast rhythm often boosted by drugs that enhance GABAA receptors, reinforcing the idea that their inhibitory system was overactive.
A targeted drug that loosens the brakes
To see if they could normalize this over-braking, the researchers turned to RO4938581, an experimental compound that selectively weakens alpha-5–type GABAA receptors without affecting other forms. In hippocampal slices, the drug reduced the amount of inhibitory charge transfer in Dup15q neurons, countering the excess braking. Given daily by mouth for several weeks, the same compound improved the animals’ performance in the reversal phase of the water maze, allowing them to adjust to the moved platform more like healthy mice. It also brought their social interactions closer to normal. In EEG recordings, an acute dose partially reduced the exaggerated beta power in Dup15q mice, nudging their brain rhythms toward the typical pattern.
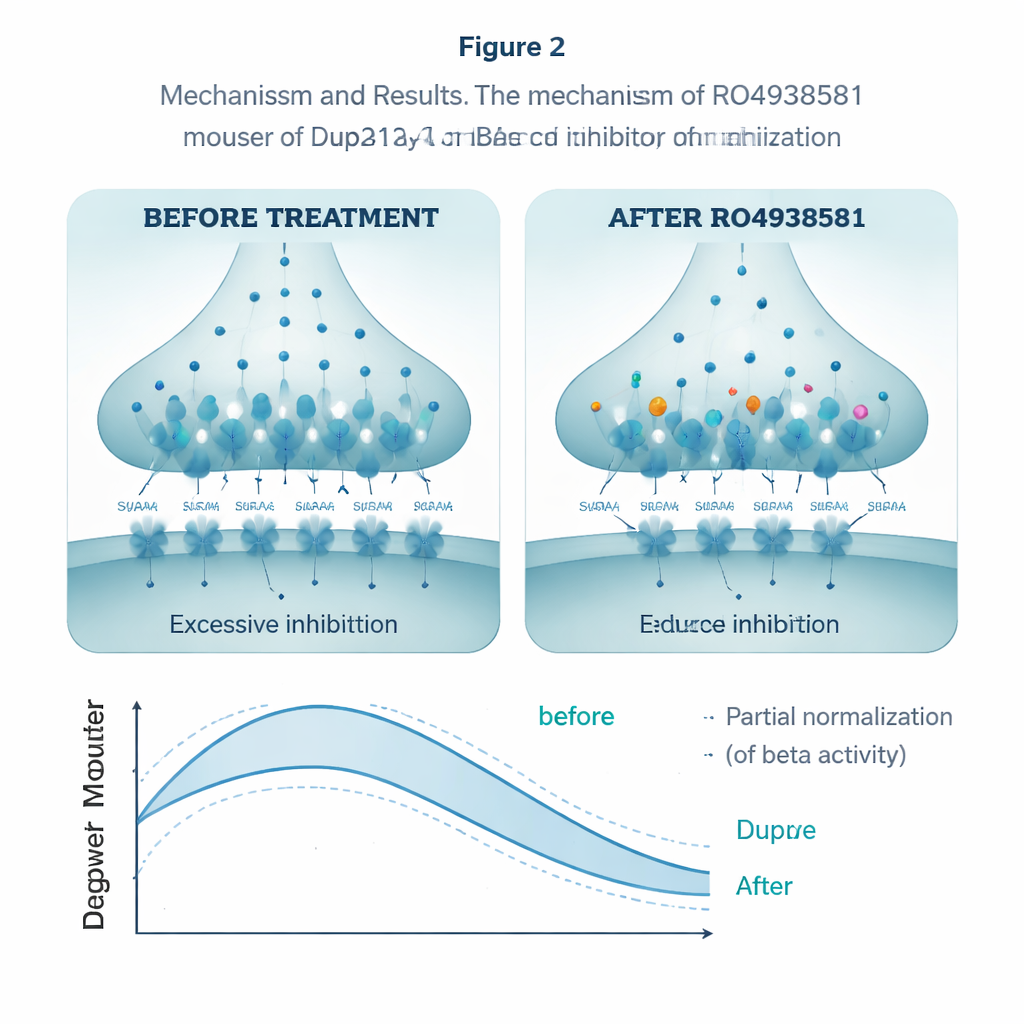
What this could mean for future treatments
For families living with Dup15q syndrome, these findings do not represent an immediate cure, but they do offer a concrete lead. The work pinpoints one particular receptor subtype—GABAA receptors containing the alpha-5 unit—as both overproduced and functionally important in a relevant animal model. Modestly turning down this receptor with a negative modulator like RO4938581 did not fix everything, but it did improve flexible learning, social behavior, and a key EEG signature. That combination of brain, behavior, and biomarker changes strengthens the case for testing safer, human-ready alpha-5–targeting drugs as precision treatments for Dup15q and potentially related neurodevelopmental conditions.
Citation: Nakagawa, R., Nani, F., Hipp, J.F. et al. RO4938581, a GABAA-α5 negative allosteric modulator rescued behavioral and EEG phenotypes of a mouse model of Dup15q syndrome. Mol Psychiatry 31, 1351–1360 (2026). https://doi.org/10.1038/s41380-025-03247-y
Keywords: Dup15q syndrome, GABAA alpha-5 receptor, neurodevelopmental disorder, EEG beta power, mouse model