Clear Sky Science · en
Cascading inertial microfluidics for high-throughput, multi-scale enrichment of tumor cells and intact clusters towards enhanced malignancy diagnosis
Why Sorting Cancer Cells in Fluid Matters
When cancer spreads, tumor cells often escape into the fluids that bathe our organs. In people with lung cancer, this can include the liquid that builds up around the lungs, called pleural effusion. Hidden within this fluid are rare single tumor cells and tight clumps of cells. These clusters are thought to be especially dangerous because they are strongly linked to aggressive disease and poor outcomes. Yet, current lab methods struggle to gently and efficiently pull both single cells and clusters out of large volumes of patient fluid. This study introduces a new chip-based technology that can rapidly and carefully sort these cells from pleural effusions, opening the door to more reliable cancer diagnosis and better insight into how tumors spread.
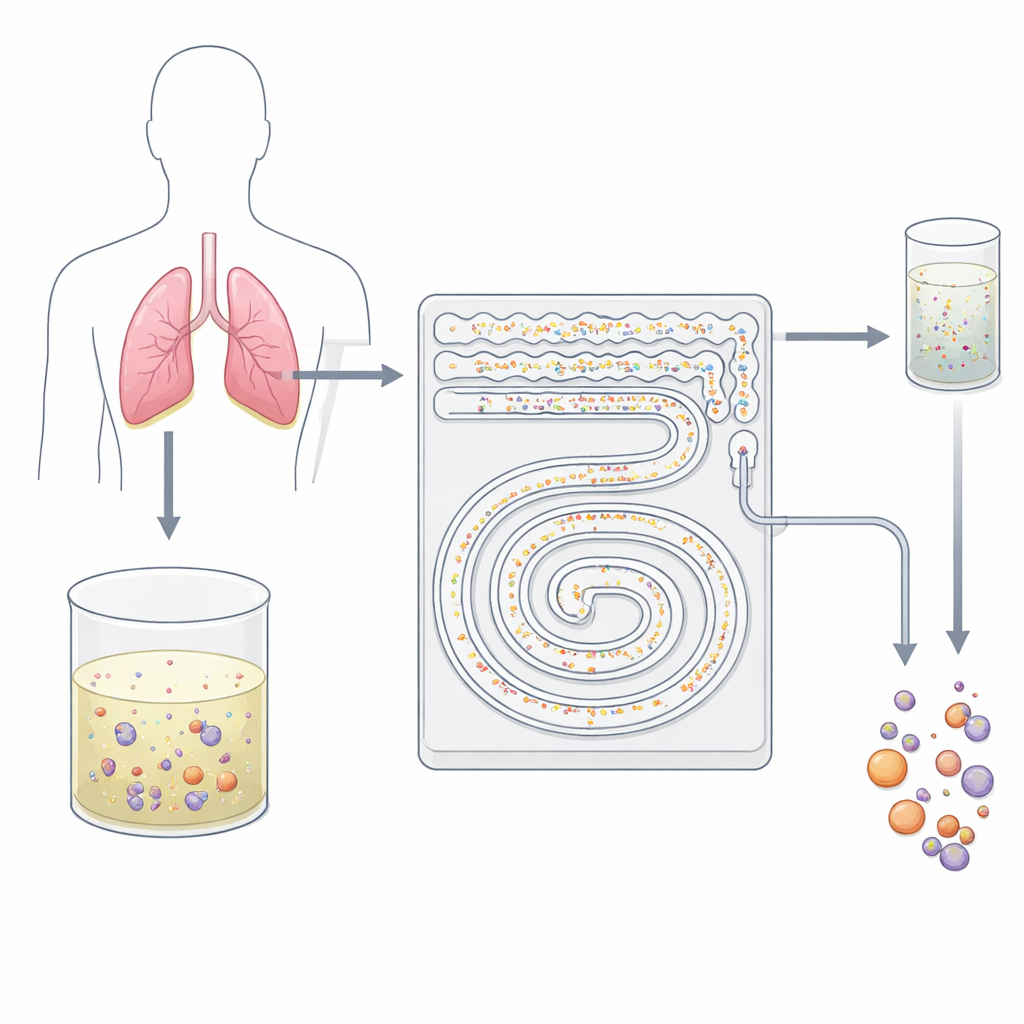
A Gentle Cell Strainer on a Chip
The researchers designed a palm-sized plastic device carved with tiny channels that act like a smart, two-step filter. Instead of using sticky antibodies or harsh sieving, the chip relies on how cells behave in fast-flowing liquid. As fluid rushes through curved microchannels, invisible forces push particles of different sizes into different streamlines. The team takes advantage of this effect, known broadly as inertial microfluidics, to separate small background blood cells from larger tumor cells and their clusters without touching or labeling them. Because everything happens in a continuous, closed flow, the device can handle tens of milliliters of fluid in just minutes—something traditional slide-based cytology cannot easily achieve.
First Step: Clearing the Crowd
In the first stage of the device, the pleural effusion—after removing excess liquid and resuspending the cells—is driven through an array of parallel serpentine channels. Here, the swirling, curved flow pushes small white blood cells toward the channel walls, while larger tumor cells and clusters stay near the center of the stream. The small cells are guided into waste outlets, dramatically thinning out the background. This high-throughput step can run at about 8 milliliters per minute, meaning a typical 50-milliliter clinical sample can be processed in roughly six and a half minutes without clogging. What emerges from this first stage is a much cleaner mixture, enriched in potentially malignant cells and ready for finer separation.
Second Step: Splitting Singles from Clusters
The partially enriched sample then enters a slanted spiral channel that includes a repeating pattern of narrow and wide sections. In this stage, several flow forces balance differently for objects of different sizes, steering them into distinct lanes near the inner or outer wall of the spiral. Single tumor-like cells, which are smaller, line up at one lateral position, whereas bulkier clusters migrate to a different position closer to the inner wall. By placing outlets at the right spots, the chip physically divides these streams: one outlet collects mainly single tumor cells, another collects intact clusters, and other outlets remove the remaining small cells. Importantly, the clusters stay in one piece, avoiding the damage and break-up often seen with traditional filtration methods.
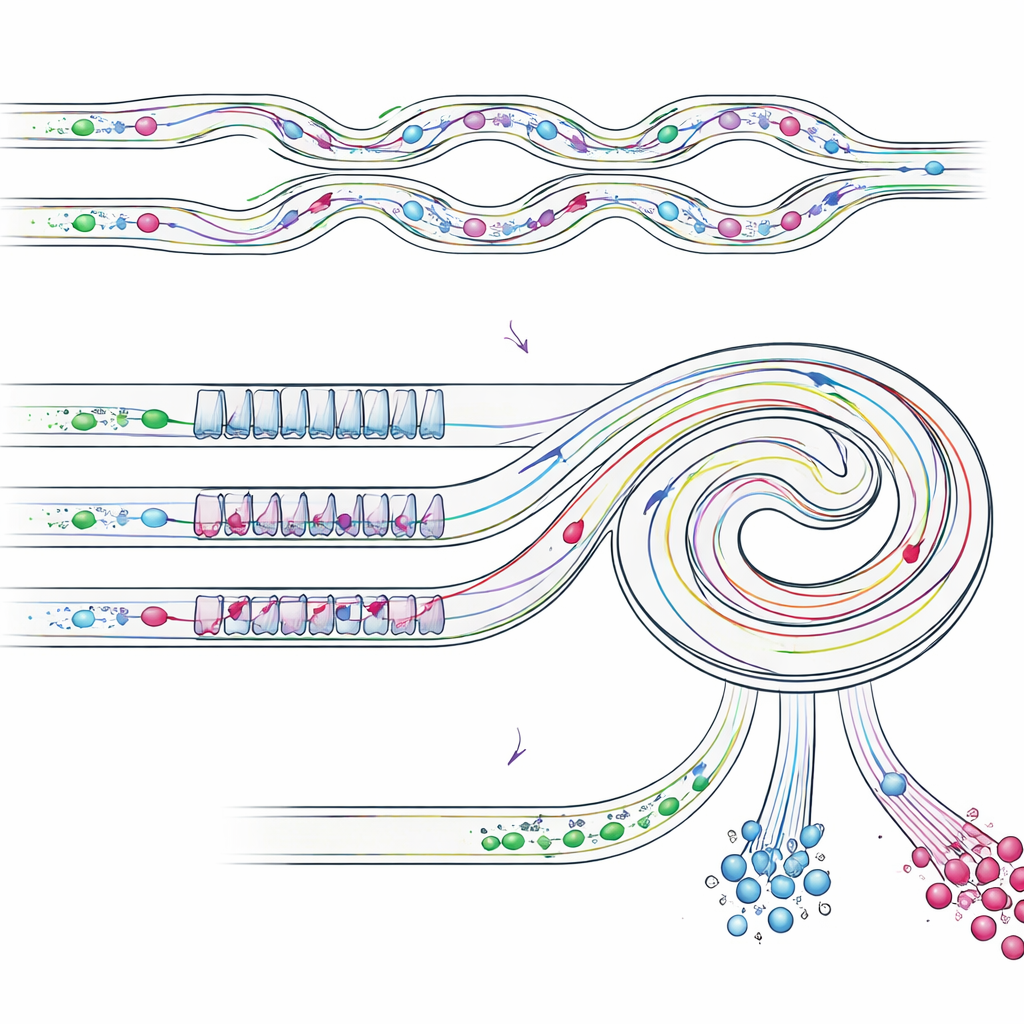
Putting the Device to the Test
To check how well their design worked, the team first used plastic beads of known sizes to mimic blood cells, single tumor cells, and clusters. They showed that the cascaded chip recovered more than 87% of the “single-cell” beads and about 92% of the “cluster” beads, while removing most smaller “blood cell” beads. They then moved on to real lung cancer cells (A549 cells) mixed with white blood cells. In these tests, the device recovered about 82% of the cancer cells overall, with around 76% purity in the enriched fraction and nearly 80% purity for cell clusters. Finally, they processed actual pleural effusion samples from three lung cancer patients. Starting from fluid in which tumor cells and clusters made up less than 1% of all cells, the chip produced fractions in which single malignant cells reached about 68% of all cells, and clusters made up about 35% of cells in a dedicated outlet stream.
What This Could Mean for Patients
By using only fluid flow and channel geometry, this device can quickly and gently pull out both single tumor cells and intact clusters from large volumes of patient fluid without chemical labels or complex instruments. The enriched cells remain suitable for standard tests like immunofluorescence staining and microscopy, fitting smoothly into existing hospital workflows. While the current study involved a small number of patients and will need larger trials to prove its impact on diagnosis and prognosis, the work demonstrates a powerful concept: a fast, label-free “cell concentrator” that reveals both the number and the clustered state of tumor cells in pleural effusions. In the long run, such tools could sharpen malignancy assessment, guide personalized treatment choices, and offer a clearer window into how cancers metastasize.
Citation: Zhu, Z., Ren, H., Zhang, Z. et al. Cascading inertial microfluidics for high-throughput, multi-scale enrichment of tumor cells and intact clusters towards enhanced malignancy diagnosis. Microsyst Nanoeng 12, 101 (2026). https://doi.org/10.1038/s41378-026-01235-y
Keywords: liquid biopsy, microfluidic cell sorting, tumor cell clusters, pleural effusion, cancer diagnosis