Clear Sky Science · en
Design of an automated cell batch microinjection system based on magnetic tweezers for zebrafish embryos
Why tiny fish eggs matter
Before new medicines or gene therapies ever reach people, scientists often test them in small, fast-growing animals. Zebrafish embryos are favorites because they are transparent and develop quickly, letting researchers watch organs form in real time. But to change their genes or deliver drugs, scientists must inject each delicate egg with a microscopic needle, a task that is usually slow, tiring, and highly dependent on human skill. This paper presents a new robotic system that can automatically inject batches of zebrafish embryos faster, more gently, and more reliably than a human, opening the door to larger and more consistent experiments.
A new way to handle fragile cells
Traditional microinjection requires a trained operator to steer a glass needle under a microscope, find each embryo, twist it into the right orientation, and pierce it without damaging vital structures. That is especially tricky because embryos are soft spheres with important regions that must be avoided to keep them alive. Existing robotic systems have helped automate transport and piercing, but they typically cannot control how each embryo is rotated in three dimensions. As a result, the needle may enter at a risky angle, lowering survival rates and limiting how many embryos can be processed reliably. The team behind this study set out to build a fully integrated platform that can transport, rotate, inject, and release embryos in a smooth sequence, without constant lens changes or manual adjustments.
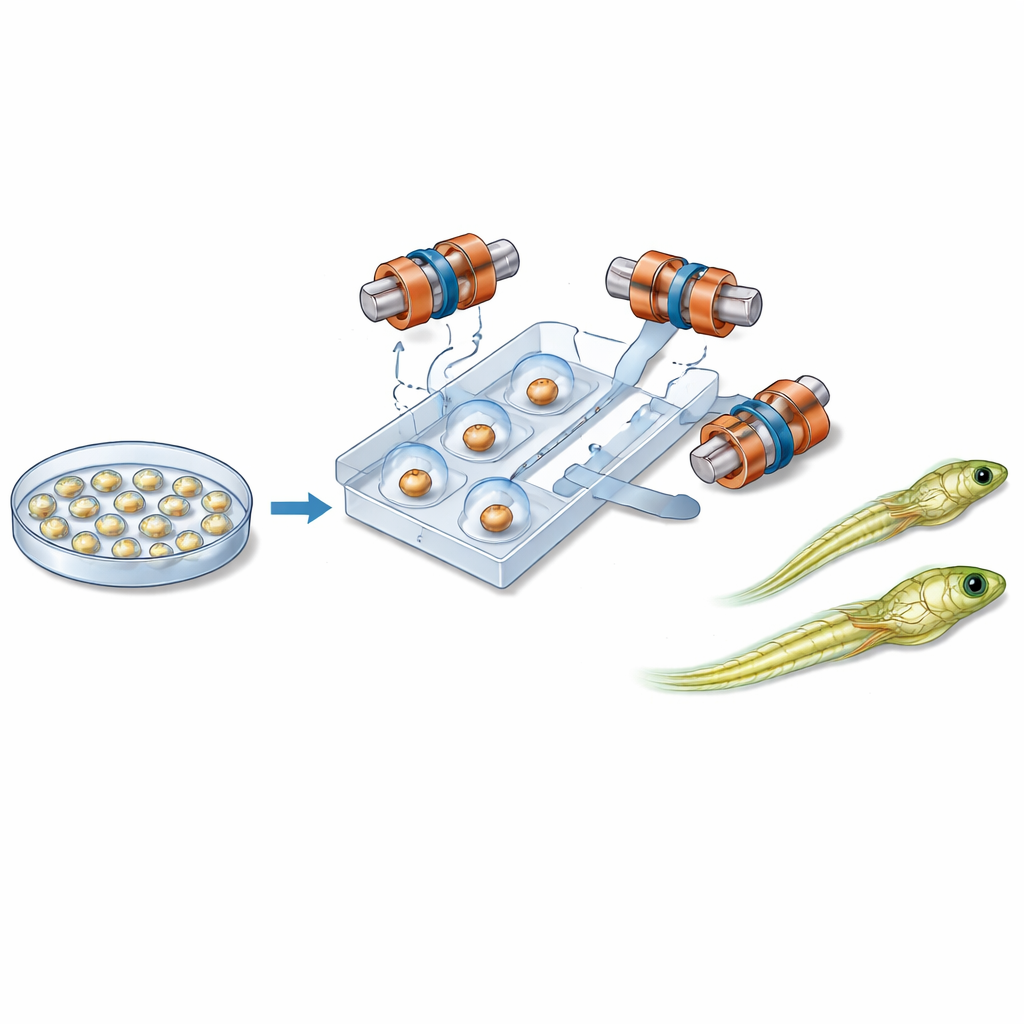
Magnets, microchannels, and gentle control
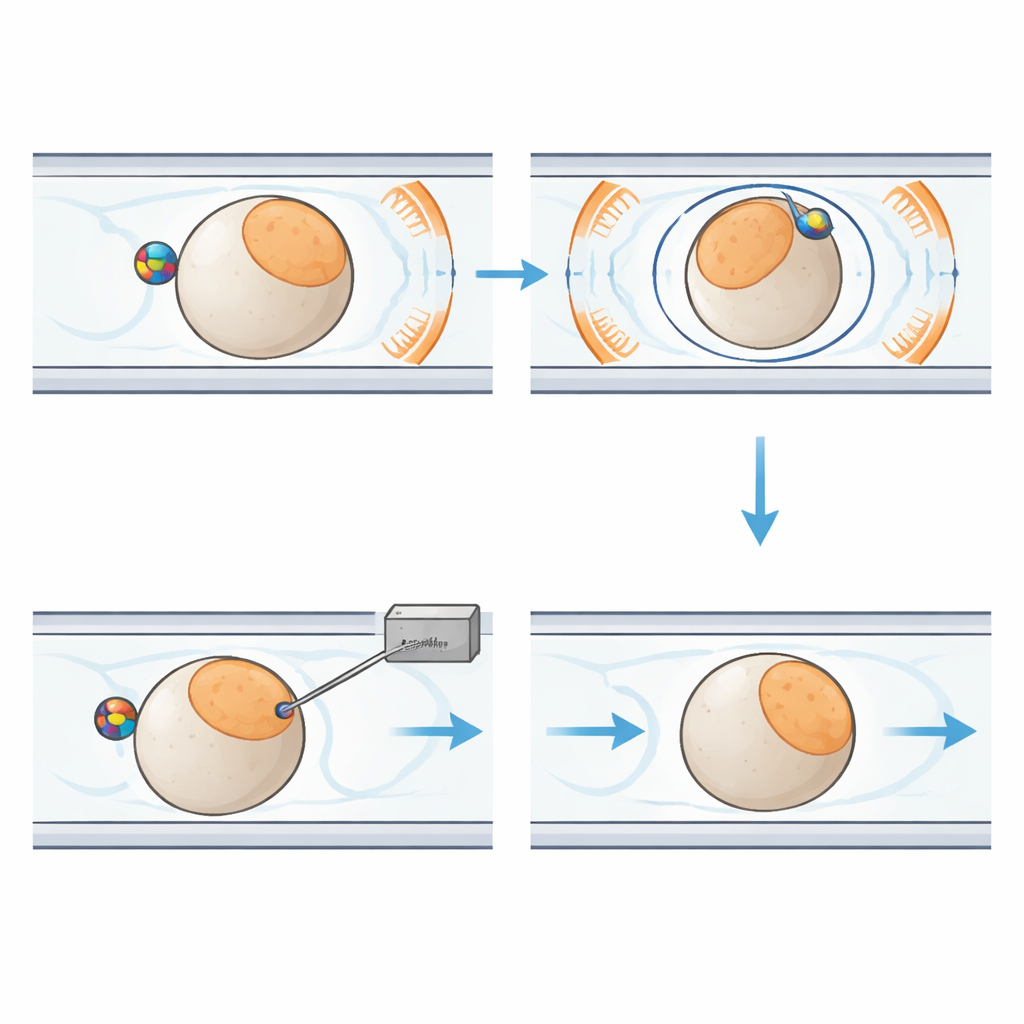
At the heart of the new system is a palm-sized plastic chip containing narrow channels that guide individual embryos along a fixed path. Close to each embryo sits a small magnetic bead. Around the chip, six carefully arranged coils generate a uniform magnetic field in three dimensions. By changing the direction of this field over time, the system causes the bead to rotate. Thanks to friction between the bead and the embryo’s surface, the entire cell turns with it, like two gears locked together. This allows the robot to tilt the embryo out of the imaging plane or spin it within the plane until the sensitive region is safely positioned away from the incoming needle. Liquid and gentle air pressure move and hold embryos in place, while a separate module controls the injection needle and the tiny flow of genetic material.
Seeing clearly in a tiny field of view
To make this precise choreography possible, the researchers also designed an advanced computer vision system, called the microscopic manipulation perception network. It analyzes images from a microscope camera in real time to spot each embryo, detect the fine needle tip, and distinguish the two opposite “poles” of the egg where key developmental structures lie. The network uses modern deep-learning tricks—such as dual feature-extraction paths, attention modules that highlight important details, and multi-scale fusion of image information—to handle the cluttered, low-light world inside a microfluidic chip. In tests on thousands of images, it correctly detected objects and segmented embryo regions with about 98–99% accuracy, providing the reliable feedback needed to steer both magnetic rotation and needle placement.
Proving it works in living zebrafish
The team compared their automated system to experienced human operators using 100 zebrafish embryos each. The robot completed its injections in about 33.8 seconds per embryo, more than twice as fast as manual work, and successfully punctured every cell. It also oriented each embryo with an average angular error of just 2.1 degrees, roughly half the error of human operators. Most importantly, 88% of the robot-injected embryos survived to hatch as larvae, compared with 66% after manual injection. Under a fluorescence microscope, the larvae from the automated system glowed green, showing that the introduced genetic material had been taken up and expressed as planned. By rotating embryos magnetically rather than squeezing them with mechanical tools, the system minimized deformation and damage while maintaining high throughput.
What this means for future studies
In simple terms, this work shows that a carefully engineered combination of microchannels, magnetic control, and smart image analysis can inject fragile fish embryos more quickly and with better survival than a skilled person using traditional tools. Because zebrafish are a key platform for studying development, disease, and drug effects, a reliable high-throughput injector could speed up large genetic screens and long-term biological studies that were previously limited by manual labor. With suitable design changes to the chip and control settings, the same strategy could be adapted to other cell types and small organisms, helping to shift delicate cellular surgery from an art practiced by a few specialists into an automated, standardized process.
Citation: Guo, X., Wang, F., Zhao, A. et al. Design of an automated cell batch microinjection system based on magnetic tweezers for zebrafish embryos. Microsyst Nanoeng 12, 113 (2026). https://doi.org/10.1038/s41378-026-01230-3
Keywords: zebrafish embryos, microinjection, magnetic tweezers, microfluidics, robotic cell manipulation