Clear Sky Science · en
Ureteral stent sleeve for early detection of hydronephrosis
Why kidney swelling needs a better early warning
Kidney swelling, known medically as hydronephrosis, can quietly damage the kidneys when urine cannot drain properly. Many patients already live with small plastic tubes called ureteral stents that keep urine flowing, for example after kidney stone treatment. But if these stents clog, pressure in the kidney can rise for days or weeks before anyone notices, risking permanent loss of kidney function. Today, doctors can only check for this problem with hospital imaging scans, not with continuous, home-friendly monitoring. This study introduces a new add‑on device, called the UroSleeve, that aims to turn an ordinary ureteral stent into a smart early‑warning system for dangerous pressure build‑up.
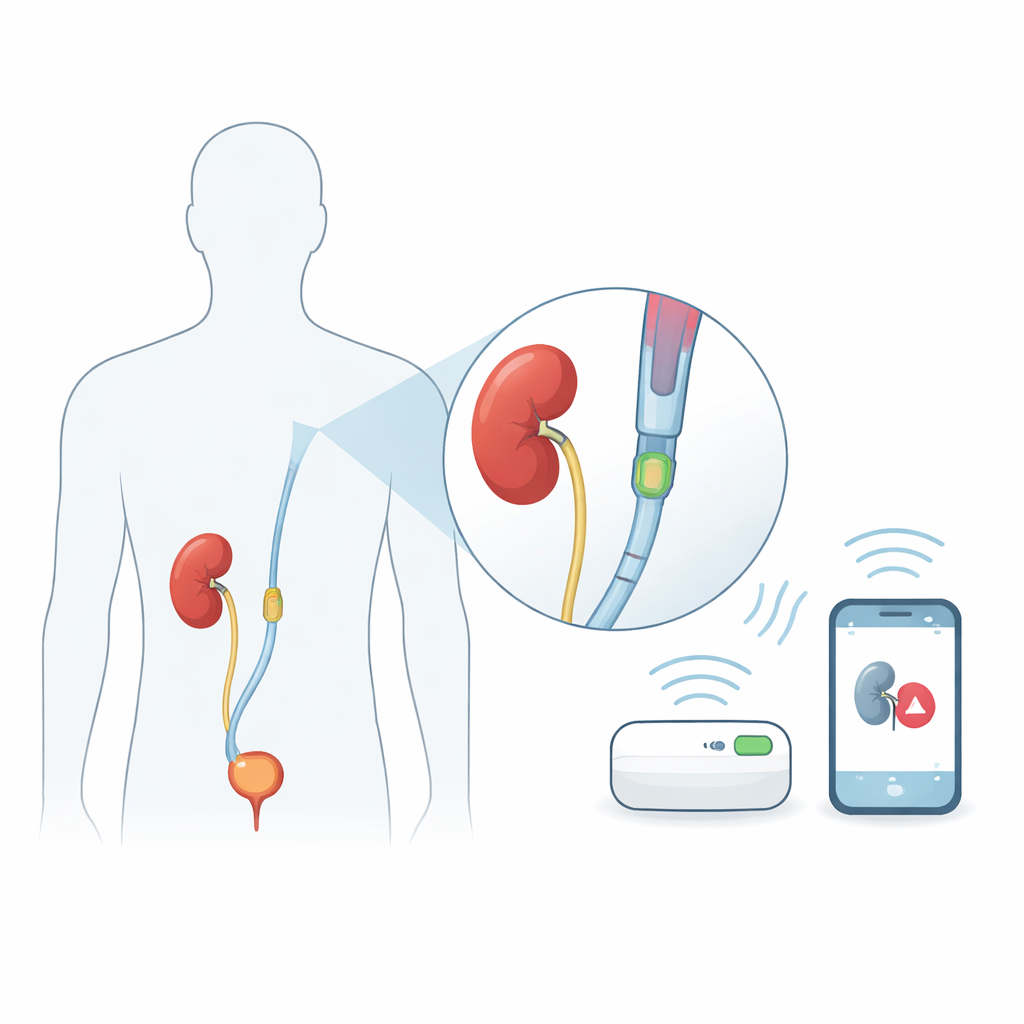
A smart sleeve that slips over existing tubes
Instead of redesigning the ureteral stent itself, the researchers built a thin sleeve that slides over the part of a standard stent that sits in the kidney. This modular add‑on approach means existing stent designs and manufacturing methods can stay the same, easing clinical adoption. Inside the UroSleeve are two main parts: a tiny pressure sensor that reacts to how hard the kidney fluid presses on it, and a spiral metal coil that acts like a small antenna. Together they form an electrical circuit whose natural "ringing" frequency shifts as the pressure changes. An antenna placed on the patient’s skin can listen for this shift, allowing wireless readings of kidney pressure without any battery or wires inside the body.
How the hidden sensor feels pressure
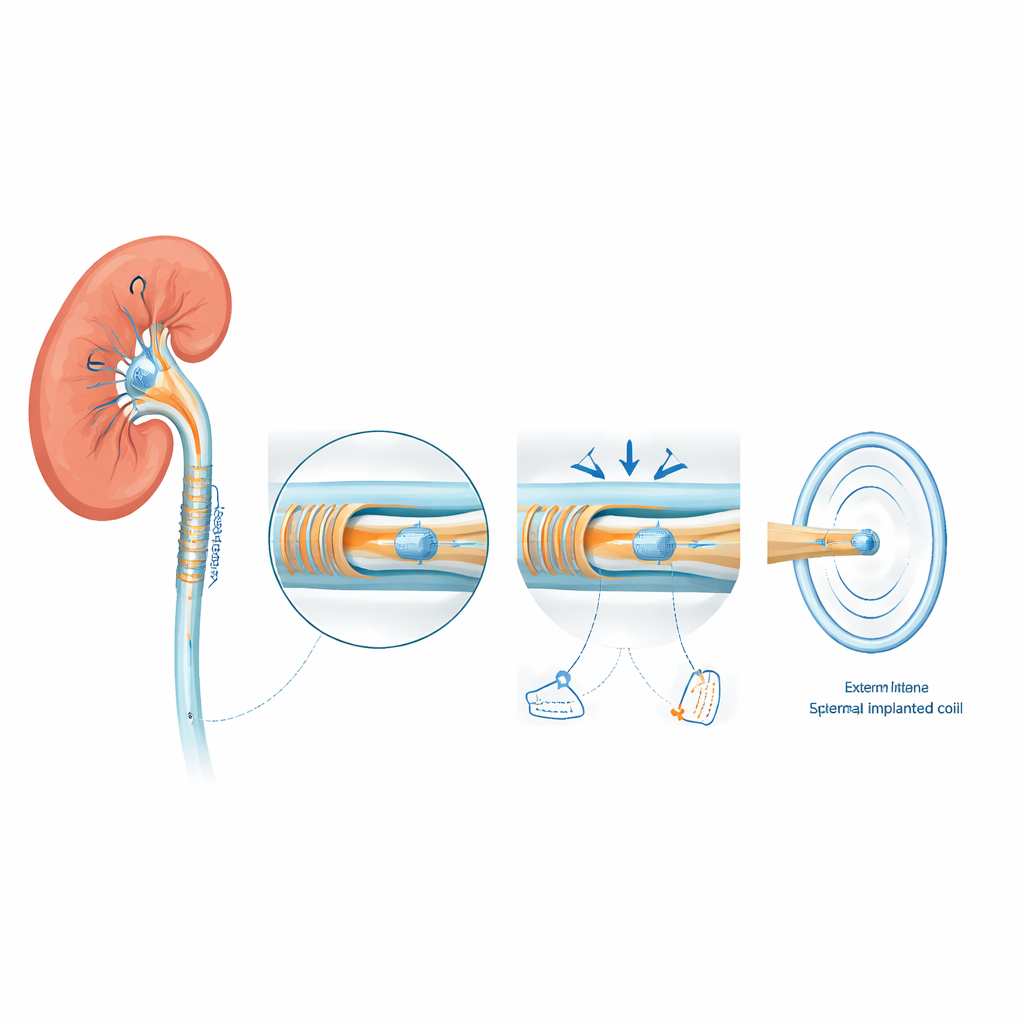
At the heart of the UroSleeve is a special pressure sensor made using micro‑scale manufacturing similar to that used in computer chips. The sensor contains a thin flexible membrane facing a rigid surface, with a tiny sealed cavity in between. As pressure in the kidney rises, the membrane is pushed down until it gently touches the insulated surface beneath over a growing area. This change in contact area alters the sensor’s electrical properties much more strongly than simply squeezing the gap, making the signal easier to detect. To build and seal this delicate cavity, the team used one‑way microchannels shaped like tiny fluid "valves" that let sacrificial material escape during fabrication, but then become blocked when a protective coating is applied, leaving a clean vacuum pocket that improves sensitivity.
Turning pressure into a wireless signal
The membrane‑based pressure sensor is wired to a flexible spiral coil patterned on a thin plastic film that can wrap around the stent without making it too stiff. Together they behave like a classic inductor‑capacitor circuit whose resonant frequency depends directly on the sensor’s state. When an external reader coil is brought close to the patient’s side, energy is passed magnetically between the two coils. At the exact frequency where the implanted circuit resonates, the reader detects a sharp dip in its own electrical response. As kidney pressure increases, the sensor’s capacitance changes, shifting this dip to a lower frequency. By tracking how the dip moves over time, the system can follow pressure inside the kidney continuously, without any surgery to access the device after implantation.
Testing the sleeve in a realistic kidney model
To see whether this concept works under realistic conditions, the team implanted UroSleeve‑equipped stents into pig kidneys that had been removed right after euthanasia and kept in a nourishing solution. They then mimicked a blocked ureter by clamping the outlet and slowly pumping water into the kidney through the stent, while watching pressure with a standard lab sensor. At the same time, an external coil placed against the kidney wall listened to the UroSleeve’s resonant signal through water. As pressure rose from normal to clearly harmful levels, the device’s resonant frequency shifted steadily downward, with a sensitivity on the order of several kilohertz per millimeter of mercury. After the experiment, the kidneys showed obvious swelling and a widened ureter, confirming that the model reproduced hydronephrosis‑like conditions.
What this could mean for patients
The study shows that a thin sleeve added to existing ureteral stents can wirelessly track pressure inside the kidney with no onboard power source and no need to alter the stent’s proven design. For patients, a mature version of this technology could provide an early warning when a stent begins to fail, prompting a timely stent change before lasting kidney damage occurs. The current work is an early proof‑of‑concept in ex vivo organs, and future steps include long‑term animal tests, deeper safety studies, and improvements in the external reader for everyday use. If these hurdles are cleared, UroSleeve‑like devices could fit seamlessly into standard urology care, turning a passive drainage tube into an active guardian of kidney health.
Citation: Shalabi, N., Searles, K., Herout, R. et al. Ureteral stent sleeve for early detection of hydronephrosis. Microsyst Nanoeng 12, 97 (2026). https://doi.org/10.1038/s41378-026-01224-1
Keywords: hydronephrosis, ureteral stent, wireless pressure sensor, kidney monitoring, implantable devices