Clear Sky Science · en
Extreme sensitivity label-free biosensing platform based on topologically disruptive phase nano-optics
Why Seeing the Invisible Matters
Many diseases, including cancer and neurodegenerative disorders, release telltale molecules into our blood long before symptoms appear. Today’s diagnostic tests often miss these early signs because the molecules are too small or too scarce to detect reliably. This paper reports a new kind of optical sensor that can spot vanishingly small amounts of such biomolecules without using fluorescent tags or chemical labels. By engineering matter at the scale of a few atoms across, the authors harness subtle shifts in light to turn tiny biochemical events into large, easy-to-measure signals.
Turning Light Into a Super-Sensitive Detector
Conventional plasmonic biosensors work by shining light onto a thin metal film and watching how the reflected light changes when molecules land on the surface. These devices are already sensitive, but they struggle with very small molecules or extremely low concentrations. Instead of tracking brightness or color, the new platform focuses on the phase of light—the precise timing of its wave. Under special conditions, the reflected light almost disappears, and its phase changes extremely rapidly. These points, known as optical “dark” states or phase singularities, make the system extraordinarily responsive to even slight changes near the metal surface.
Building a Nanoscale Light Trap
To create these singularities, the team designed a layered structure just a few tens of nanometers thick. A glass base supports a 12-nanometer layer of aluminum oxide containing ultra-small silver nanoparticles less than 3 nanometers in diameter, all capped by a smooth, 48-nanometer gold film. The silver particles are carefully generated and embedded so that they remain crystalline, nearly spherical, and uniformly spaced at sub-nanometer distances. This arrangement lets the particles’ localized plasmon modes couple strongly both to each other and to traveling plasmon waves in the gold layer. The result is a kind of nanoscale optical cavity in which light energy becomes tightly confined and its phase extremely sensitive to the surrounding medium.
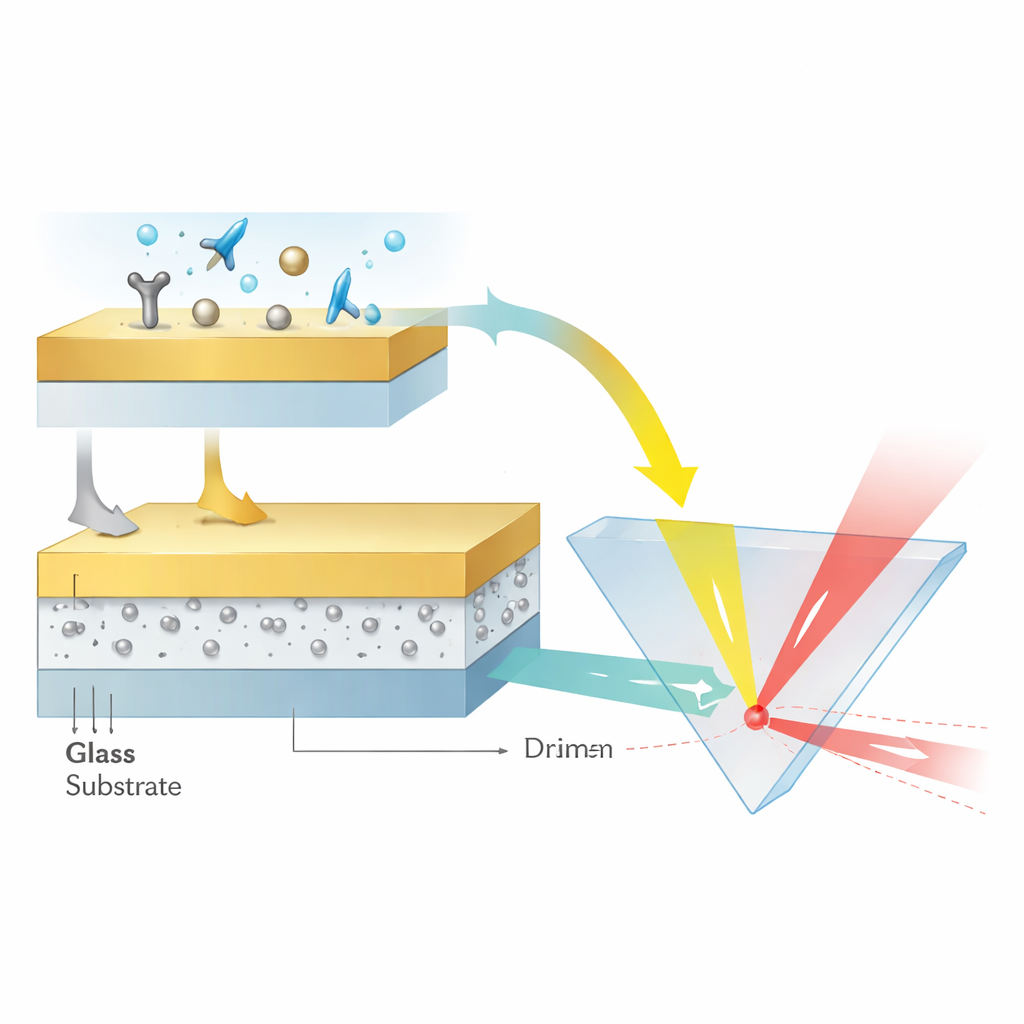
Making Light Slide Sideways
Rather than measuring angles or colors, the authors read out their sensor by tracking how far the reflected light beam shifts sideways along the surface—a phenomenon called the Goos–Hänchen shift. When a laser beam reflects under the right conditions, its energy peak can emerge slightly displaced from where simple geometry would predict. Near a phase singularity, that displacement grows dramatically. By tuning the silver nanoparticle concentration to about 16 percent, the team drove the reflectivity near zero and sharpened the phase jump until tiny refractive index changes, caused by molecules binding at the gold surface, produced lateral beam shifts hundreds of micrometers wide. In calibration tests using dilute glycerin solutions, the device reached a sensitivity equivalent to 3.27 × 10^8 nanometers of beam shift per unit change in refractive index and resolved changes as small as about four parts in ten million.
Detecting Tiny Molecules at Vanishing Concentrations
To demonstrate practical biosensing, the researchers first targeted biotin, a vitamin-sized molecule with a very low molecular weight. Standard surface plasmon sensors cannot reliably see biotin even at micromolar levels. Here, by decorating the gold surface with streptavidin, which binds biotin strongly, the new platform clearly tracked real-time binding at concentrations down to 1 femtomolar—roughly one molecule among 10^15 solvent molecules. The signal increased steadily with each tenfold rise in concentration, confirming that the beam shift scales predictably with coverage of such tiny analytes.
Hunting Cancer Markers in the Attoworld
The team then moved to a clinically relevant target: tumor necrosis factor-alpha (TNF-α), a cytokine associated with inflammation and cancer, present in patient serum at around 10^−13 molar. They functionalized the gold surface with short DNA strands (aptamers) that specifically capture TNF-α and blocked remaining areas to suppress nonspecific binding. Under these conditions, the sensor registered clear, stable signals for TNF-α at concentrations as low as 0.1 attomolar (10^−19 molar) and produced a shift of nearly 47 micrometers at 10^−13 molar, squarely within the medically relevant range. Control tests with another cytokine, interleukin-6, yielded almost no lasting signal, confirming that the response was both highly sensitive and selective.
What This Means for Future Medical Tests
Put simply, this work shows that carefully arranged silver nanoparticles hidden beneath a thin sheet of gold can turn an almost imperceptible change at the surface into a large sideways movement of light that is easy to measure. By operating at phase singularities, the platform bypasses the need for fluorescent labels and pushes sensitivity into the zepto- to attomolar regime for real biological targets. If translated into robust, user-friendly devices, such sensors could enable blood tests that detect disease markers long before current methods, opening new windows for early diagnosis and real-time monitoring of health.
Citation: Du, F., Gireau, M., Youssef, J. et al. Extreme sensitivity label-free biosensing platform based on topologically disruptive phase nano-optics. Microsyst Nanoeng 12, 106 (2026). https://doi.org/10.1038/s41378-026-01222-3
Keywords: label-free biosensing, plasmonic sensor, nanoparticles, early disease detection, cancer biomarkers