Clear Sky Science · en
PEGylated Cu-doped WS2 hybrid nanosheets for targeted multimodal cancer therapy
Why Smarter Cancer Treatments Matter
Many people with breast cancer still face harsh treatments that can damage healthy tissue while not fully stopping the tumor. This study describes a new kind of tiny engineered sheet, too small to see with the naked eye, designed to travel through the body, home in on breast tumors, and attack them in several coordinated ways at once. By combining heat, chemistry, and a classic cancer drug into a single targeted package, the researchers aim to make treatment both more powerful and gentler on the rest of the body. 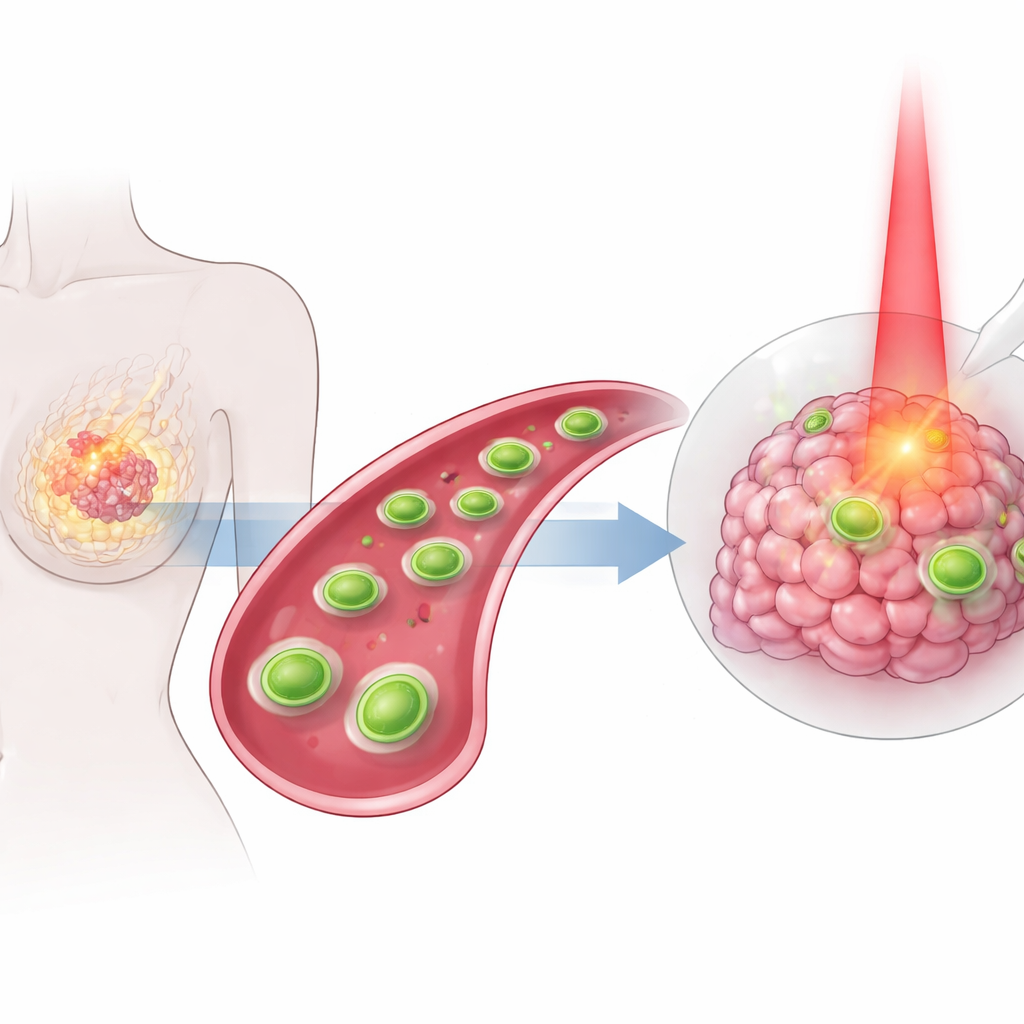
Tiny Sheets Built to Carry and Deliver Treatment
The heart of the approach is an ultra-thin material made from tungsten and sulfur, shaped like nanosized sheets. These sheets naturally absorb near‑infrared light and turn it into heat. The team first created these sheets and then carefully roughened their surface so they could hold other useful ingredients. Copper atoms were anchored in a highly dispersed way onto the sheets, and a soft, flexible coating similar to a molecular “raincoat” was added to help the particles mix well in body fluids and avoid clumping. Finally, they attached folic acid—a vitamin that many tumor cells avidly take up—as a homing device, and loaded the common chemotherapy drug doxorubicin onto the surface. The end result is a tiny, layered platform that can circulate in the blood, recognize tumor cells, and carry a hefty cargo of treatment.
How Light, Chemistry, and Drug Work Together
Once these particles reach a tumor, several things happen in concert. When the tumor area is illuminated with a gentle red laser, the tungsten–sulfur sheets heat up efficiently, raising the local temperature enough to stress and damage cancer cells without overheating the rest of the body. At the same time, the copper atoms on the sheets react with natural peroxide molecules that are already present at higher levels inside tumors. This reaction turns the peroxide into very aggressive short‑lived oxidants that punch holes in cellular components from the inside. Tumors also tend to be slightly more acidic than healthy tissue; in this sourer environment, and under heating by the laser, the bond between the sheet and the doxorubicin weakens, allowing more of the drug to be released right where it is needed. These three effects—heat, chemical attack, and focused chemotherapy—are designed to reinforce one another. 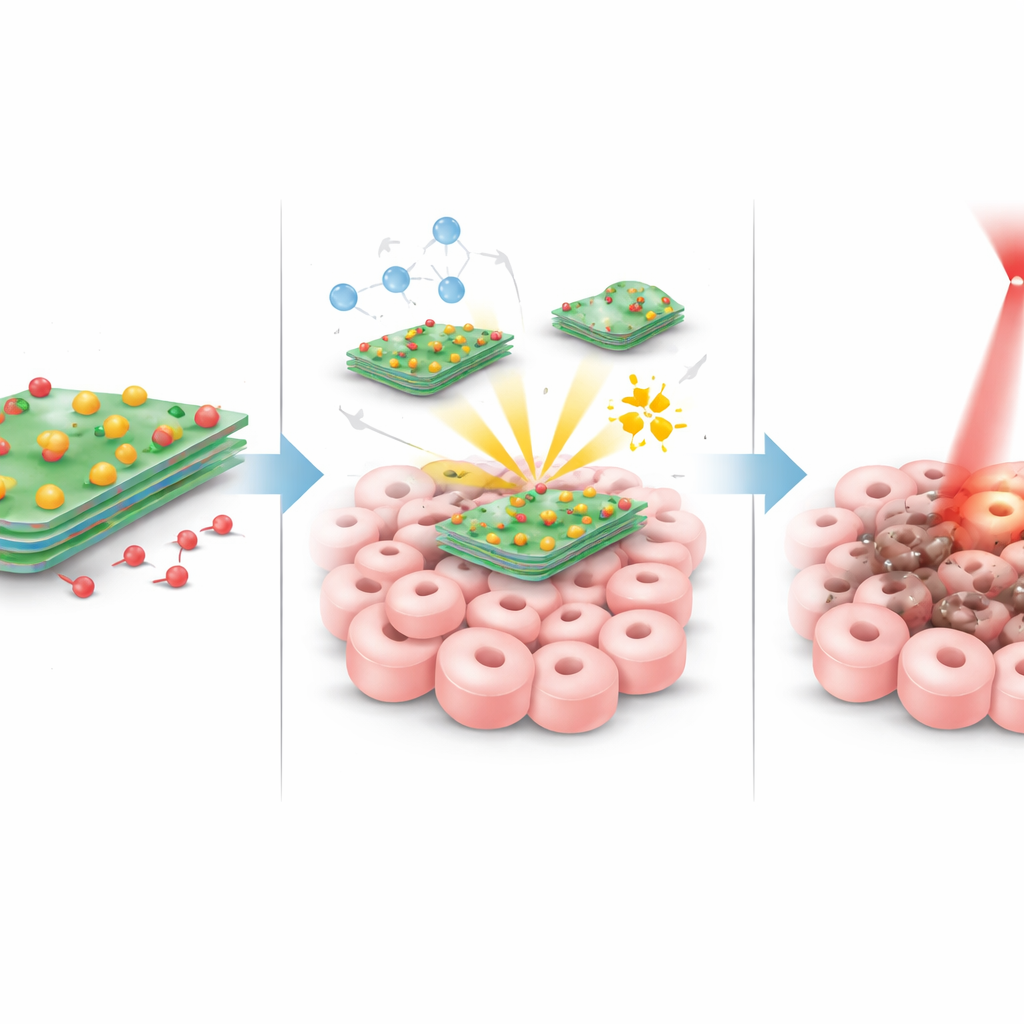
Evidence from Cells and Tumor‑Bearing Mice
In lab dishes, the coated sheets alone showed little harm to normal cells, suggesting good basic safety. But when loaded with doxorubicin and exposed to laser light in the presence of peroxide, they caused strong death of breast cancer cells, much greater than any single treatment alone. The researchers also showed that the particles generate a surge of reactive oxidants inside tumor cells, confirming that the copper chemistry is active. In mice carrying breast tumors, particles with the folic‑acid coating accumulated much more strongly and for longer in tumor tissue than non‑targeted particles. When the mice received the full combination—targeted particles plus red‑light exposure—their tumors shrank dramatically, their survival time increased, and their body weight and organ health remained largely normal, indicating limited side effects compared with standard drug treatment.
What This Could Mean for Future Cancer Care
Taken together, the findings suggest that these engineered sheets act like a multitool for cancer treatment: they seek out tumors, heat them, poison them from within via reactive chemistry, and deliver a proven drug more precisely, all while sparing much of the rest of the body. The work is still at the animal‑study stage, and important questions remain about long‑term safety, breakdown, and how best to manufacture such particles for human use. However, the design shows how combining several modest treatments into a single smart package tailored to the tumor’s own environment can produce a much stronger overall effect. If future studies confirm their safety and effectiveness, such multifunctional nanoplatforms could help make breast cancer therapy more targeted, more efficient, and less punishing for patients.
Citation: Li, D., Wen, C., Wu, H. et al. PEGylated Cu-doped WS2 hybrid nanosheets for targeted multimodal cancer therapy. Microsyst Nanoeng 12, 121 (2026). https://doi.org/10.1038/s41378-026-01218-z
Keywords: breast cancer therapy, nanomedicine, photothermal treatment, targeted drug delivery, chemodynamic therapy