Clear Sky Science · en
Rapid-manufacturing and cost-effective single-layer microfluidic device for high-throughput three-dimensional hydrodynamic focusing
Why shrinking tiny streams matters
Modern medicine increasingly depends on looking at huge numbers of single cells one by one, for example to spot cancer cells in urine or blood. To do this quickly and cheaply, cells are often sent through hair-thin channels on microchips, where lasers or cameras inspect them. But to get sharp, reliable images at extreme speeds, every cell needs to pass through almost exactly the same tiny spot. This paper presents a new way to build such chips so that they can tightly herd cells into a narrow three-dimensional stream, even at very high speeds, using a device that is faster and cheaper to make than today’s standard systems.
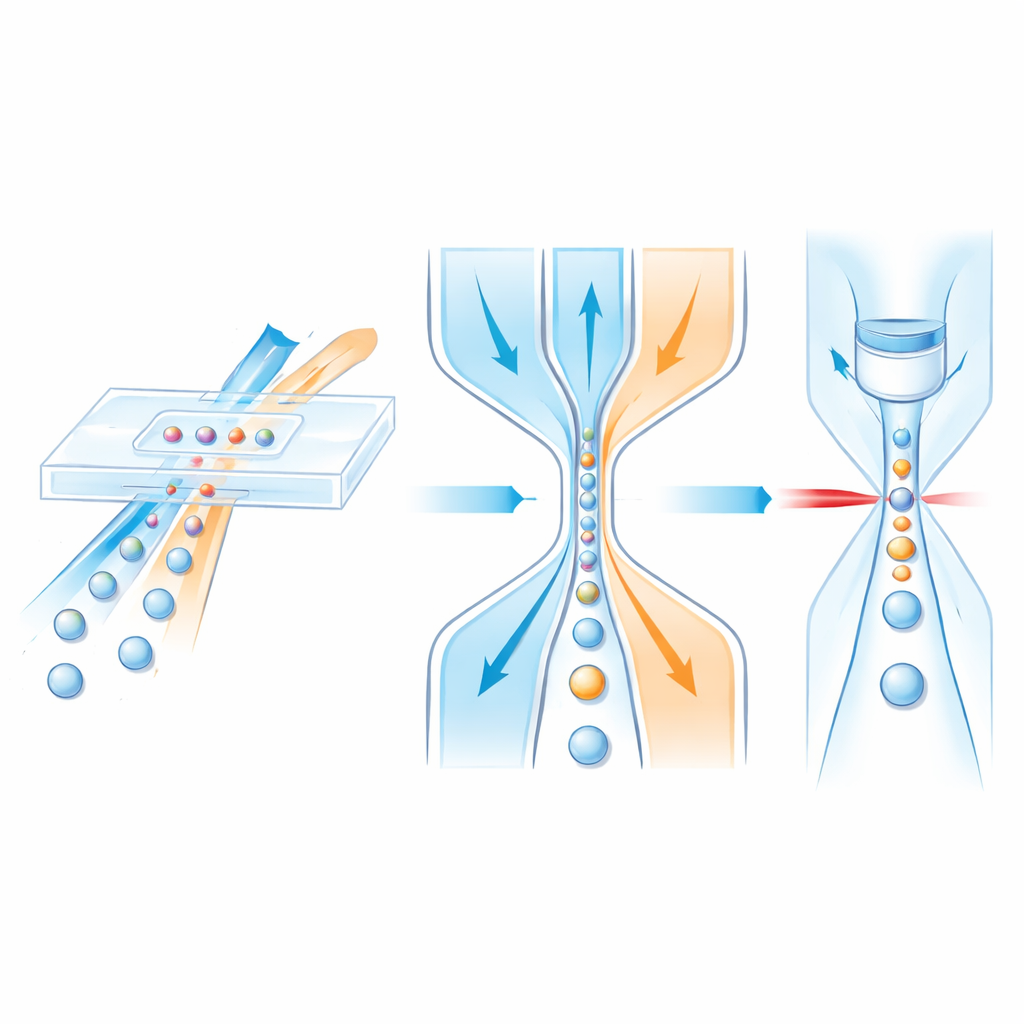
Guiding cells with flowing “traffic lanes”
Inside a microfluidic chip, cells ride in a central liquid stream while surrounding “sheath” fluids act like invisible guardrails, gently squeezing the sample toward the center. Earlier designs could usually focus cells only side-to-side, not up-and-down, or they relied on complex multi-layer structures that are slow and costly to manufacture. The authors instead design a single-layer channel that still achieves full three-dimensional control. First, the sample joins a vertical sheath flow at an angled T-shaped junction that narrows along its length. Because of the channel shape and the inertia of the liquids at higher speeds, the sample stream is pushed toward the top half of the channel. Next, two matching side sheaths further downstream squeeze from left and right, pinching the already lifted sample into a tight central filament that runs through a detection window.
Building better chips in minutes, not hours
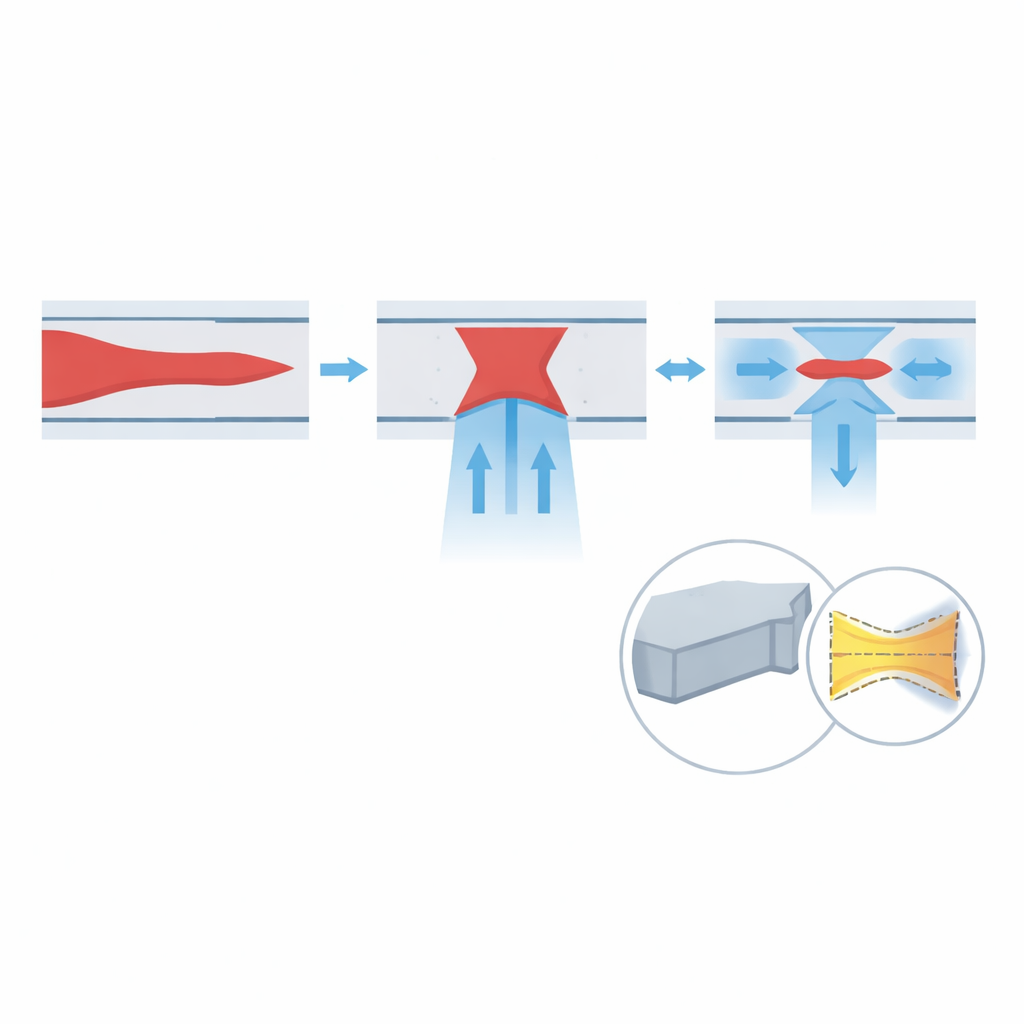
Most research microfluidic chips today are made from soft silicone (PDMS) using soft lithography, a process that requires multiple heating and curing steps and can take an hour or more per device. PDMS is easy to mold but bends under high pressure, causing the channels to puff outward and the focused stream to spread. The new device uses a hard plastic called polyurethane acrylate (PUA), patterned by a “double transfer” process. First, a reusable PDMS mold with raised channel features is cast from a silicon master. Liquid PUA is then poured into this mold, cured under ultraviolet light, and peeled off to form the channel layer. A separate flat PUA-coated glass slide serves as the base. The two PUA surfaces are aligned, pressed together, and bonded with another brief UV exposure. Because each curing step takes only seconds and no long baking is required, a complete chip can be produced in about five minutes, roughly ten times faster than traditional methods.
Testing the flow and taming deformation
To understand how well the design works, the team combines computer simulations with experiments. First, they simulate how changing the flow rates of the sample and sheath streams affects the shape of the focused core. The results show that increasing the vertical and side sheath flows both help to narrow the sample in height and width, and that higher overall speeds (higher Reynolds number) further improve focusing. They then simulate how the channel walls deform when made from soft PDMS versus stiff PUA. Under realistic high-speed conditions, PDMS walls bulge by over a hundred micrometers, enough to distort the flow so badly that the sample splits and drifts toward the corners. In contrast, PUA deforms by less than a hundred nanometers—effectively rigid at this scale—so the focused stream stays centered and tight even at high pressure.
Seeing real cells at extreme speeds
Beyond colored dye tests, the authors evaluate the device using optical time-stretch (OTS) microscopy, a technique that converts ultrafast laser pulses into rapid line scans, allowing millions of line images per second. They send treated urine samples from bladder cancer patients through the chip at increasing flow rates, while OTS records two-dimensional images of each passing cell. Because the optical system has a very thin focal region, any cell that strays up or down appears blurry, providing a direct measure of vertical focusing. Across speeds from 3.3 to 16.7 meters per second, the fraction of sharply focused images climbs, reaching 98.4% at the highest tested speed. Lateral focusing is assessed by measuring how far cell centers deviate from the channel’s midpoint; this offset shrinks with speed, corresponding to about 95.0% lateral focusing efficiency at 16.7 meters per second.
What this means for future cell analysis
In simple terms, the researchers show that a straightforward, single-layer plastic chip can reliably herd cells into a tight, well-controlled stream in all directions, even under the demanding conditions required for ultra-high-speed imaging. By pairing a deformation-resistant material with a clever arrangement of sheath flows, they avoid the mechanical limits of soft silicone devices while dramatically shortening fabrication time. This makes it easier to produce many identical chips for clinical and industrial use, and to run large-scale, high-throughput tests on real patient samples. As a result, the technology offers a practical route toward faster, more precise cell screening tools that could benefit diagnostics, cancer monitoring, and other applications that depend on looking closely at vast numbers of individual cells.
Citation: Yan, R., Wei, S., Weng, Y. et al. Rapid-manufacturing and cost-effective single-layer microfluidic device for high-throughput three-dimensional hydrodynamic focusing. Microsyst Nanoeng 12, 87 (2026). https://doi.org/10.1038/s41378-026-01212-5
Keywords: microfluidic flow cytometry, 3D hydrodynamic focusing, high-throughput single-cell analysis, polyurethane acrylate microfluidic chips, optical time-stretch microscopy