Clear Sky Science · en
Highly mechanically stable PEDOT:PSS/PDA-modified microelectrode arrays reveal state-specific dynamic neural activity across sleep-wake
Why Better Brain Sensors Matter for Sleep
Sleep shapes how we think, feel, and stay healthy, yet the brain’s detailed activity during sleep and waking is still hard to see, especially deep inside the brain. This study tackles that challenge by building tiny, tougher brain sensors that can listen to individual nerve cells for weeks, then using them to explore how a key reward center, the ventral tegmental area (VTA), behaves across the sleep–wake cycle in mice.
Building a Tiny, Flexible Listening Device
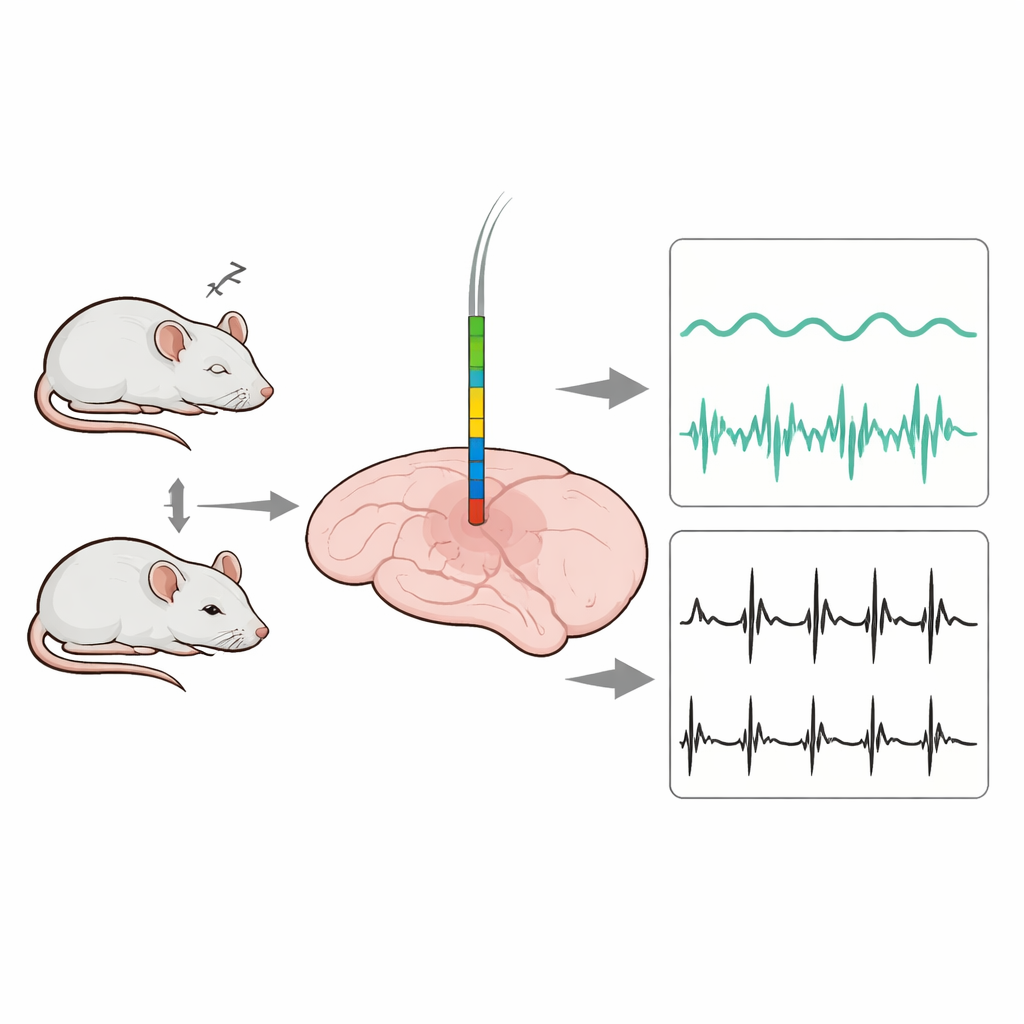
The researchers began by designing a slender microelectrode array—a comb-like strip with 16 microscopic recording sites—on a silicon base. Each site is about the size of a single neuron, allowing the device to capture both slow background waves and fast electrical spikes from individual cells. The probe is only 25 micrometers thick and a few hundred micrometers wide, so it can slide into deep brain regions like the VTA while minimizing damage and inflammation. The full system combines this deep probe with wires placed on the skull and neck to record standard brain waves (EEG) and muscle activity (EMG) at the same time.
Making Electrodes That Last Inside the Brain
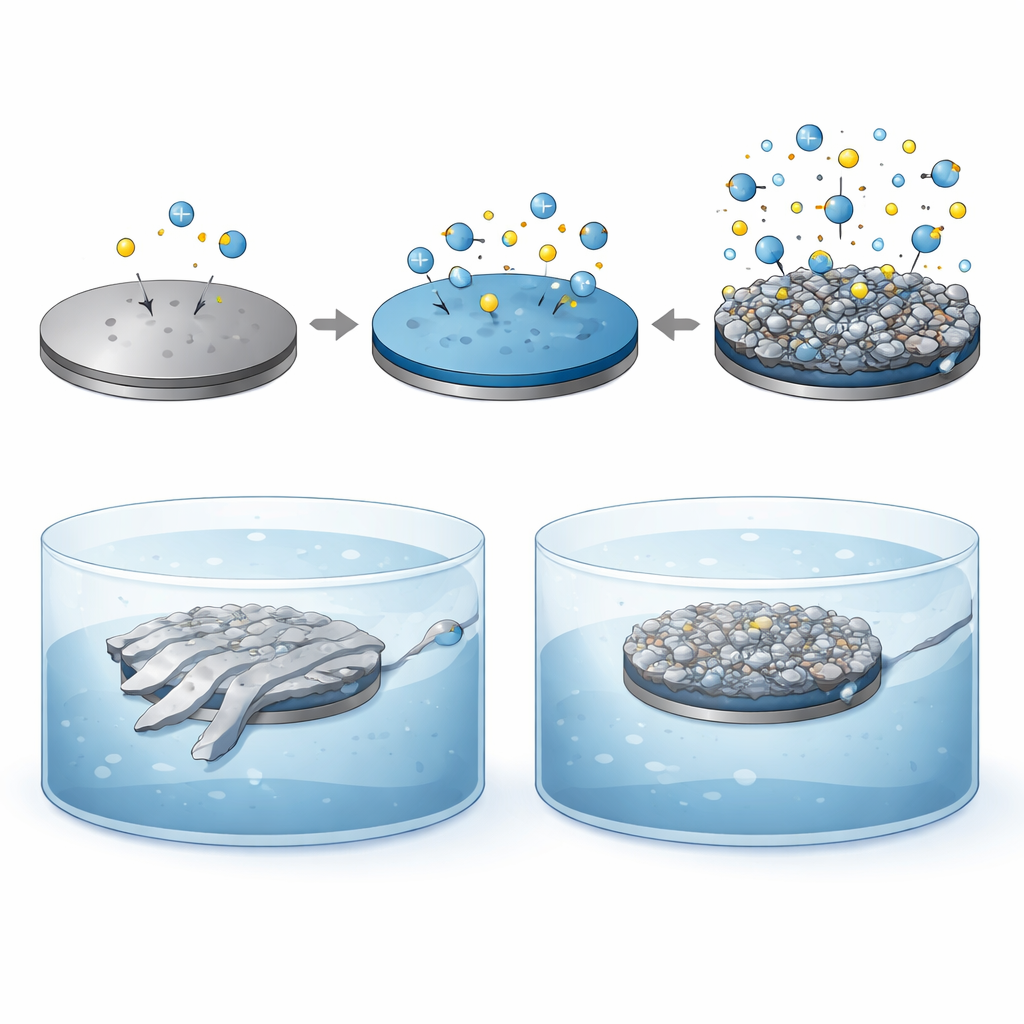
Listening to the brain over weeks is difficult because the metal surfaces of tiny electrodes often degrade, loosen, or irritate nearby tissue. To solve this, the team created a new coating that blends a well-known conductive plastic, PEDOT:PSS, with a sticky, biology-inspired material called polydopamine (PDA). Instead of layering them in separate steps, they co-deposited both in one electrochemical process, forming an interlocking network that clings tightly to the metal. This rough, sponge-like coating greatly increases the electrode’s effective surface area and adds chemical groups that attract water and cells, making the interface both more conductive and more welcoming to brain tissue.
Testing Strength, Stability, and Cell Friendliness
In the lab, the new coating transformed the electrical behavior of the electrodes. The resistance to neural signals dropped from about two million ohms for bare metal to roughly forty thousand with PEDOT:PSS alone, and to under thirty thousand when PDA was added. The amount of charge the electrode could safely store and exchange rose nearly thirty-fold compared with bare metal. Importantly, when the probes were shaken in an ultrasonic bath to mimic physical stresses inside the brain, the traditional PEDOT:PSS coating peeled and its performance collapsed, while the PEDOT:PSS/PDA coating stayed intact and stable. Tests with neural stem cells showed that the PDA-containing surface was far more hydrophilic and supported higher cell survival and growth over several days, indicating strong biocompatibility.
Watching Deep Brain Activity During Sleep and Wake
Armed with these improved probes, the scientists implanted them into the VTA of mice and recorded continuously for three weeks while also tracking EEG and EMG to label wakefulness, non-REM sleep, and REM sleep. The new coating delivered clearer neural spikes with about double the signal-to-noise ratio of standard coatings, and these clean signals remained stable over time. By sorting spike shapes and firing patterns, the team identified 87 individual neurons that fell into three groups: some fired most during wakefulness, others during sleep (both non-REM and REM), and a third group maintained similar activity across all states. At the same time, slow local field potentials in the VTA shifted from fast, low-amplitude activity in wakefulness to large, slow waves in non-REM sleep, and then back to faster rhythms in REM, with frequency bands that tracked sleep stages even more sharply than EEG recorded at the scalp.
What This Means for Sleep and Future Devices
Together, these results show that the VTA, long known for its role in motivation and reward, also contains distinct sets of neurons that track and possibly help control transitions between sleep and wake. The study also demonstrates a practical recipe for durable, gentle, and highly sensitive brain electrodes based on a PEDOT:PSS/PDA coating. For non-specialists, the takeaway is twofold: we now have clearer evidence that a deep reward center actively participates in shaping our sleep, and we have a promising technology for long-lasting brain sensors that could one day improve treatments for sleep disorders and support more reliable brain–machine interfaces.
Citation: Miao, J., Liu, Y., Wang, Y. et al. Highly mechanically stable PEDOT:PSS/PDA-modified microelectrode arrays reveal state-specific dynamic neural activity across sleep-wake. Microsyst Nanoeng 12, 105 (2026). https://doi.org/10.1038/s41378-026-01206-3
Keywords: sleep–wake regulation, ventral tegmental area, neural microelectrode arrays, conductive polymer coatings, local field potentials