Clear Sky Science · en
Robotic cell transportation system based on micropipette resistance modeling
Moving Tiny Cells Without a Microscope
Modern biology often depends on a surprisingly simple task: picking up a single cell with a glass tube and placing it somewhere else. This is essential for fertility treatments, embryo freezing, cell-based therapies, and basic research. Yet today it almost always requires a bulky microscope and a highly trained human operator watching every move. This paper describes a new robotic system that can move single cells between droplets of liquid without any microscopic view at all, opening the door to fully enclosed, automated “cell factories” that can work faster, cheaper, and with less damage to delicate cells.
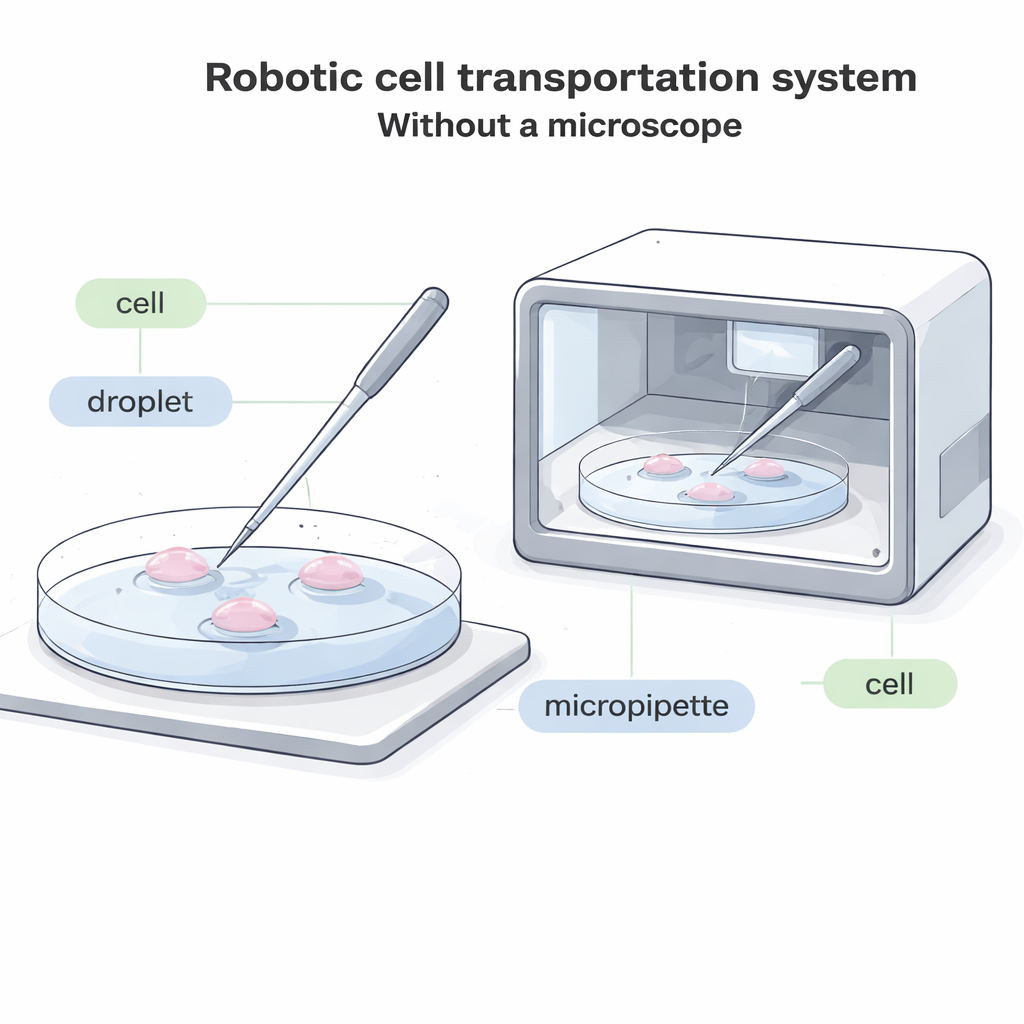
Why Cell Moving Needs a Makeover
In current labs, an operator looks through a microscope and uses a thin glass tube, called a micropipette, to land on the bottom of a dish, gently suck in one cell, shift it to a new droplet of fluid, and then push it out again. This process is slow, visually demanding, and hard to automate. In many future systems—such as compact, sealed devices that grow embryos or other cells inside a box—there simply is not enough room for a traditional microscope. In other cases, cells are labeled with glowing dyes that can fade or be damaged by light, so minimizing bright microscopic observation is important. Existing “blind” systems that don’t rely on images only work for unusually large cells, leaving most common cell types without a good automated solution.
A Special Straw With a Built-In Stop
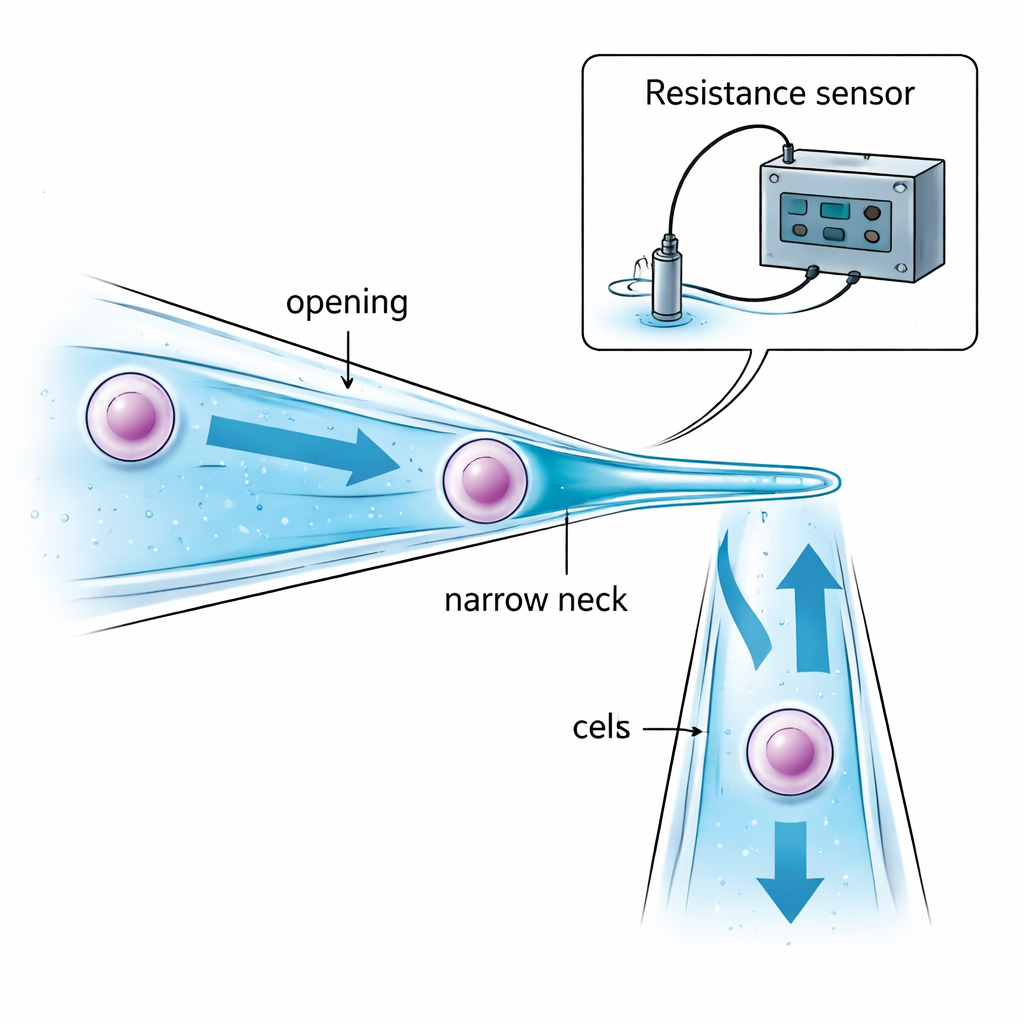
The authors attack the problem by redesigning the micropipette itself. Instead of a straight glass tube with the same diameter throughout, they create a narrow-necked micropipette. Its opening is slightly wider than the target cell so the cell can be drawn inside. Deeper in, the tube gently narrows to a “neck” that is smaller than the cell. When suction pulls the cell toward this constriction, the cell is stopped and held in place inside the tube—instead of dangling at the tip and being exposed to oil or air between droplets. Careful choice of the opening size, neck size, and distance between them ensures that the cell can be captured securely without being overly squeezed, and that only a small amount of liquid is carried from one droplet to another, reducing contamination.
Listening to Electrical Signals Instead of Looking
Without a microscope, the robot must still know three things: when the glass tip is close enough to the dish bottom, when the cell has been successfully captured at the neck, and when it has been fully released into the new droplet. The team solves this by monitoring tiny changes in electrical resistance in the liquid inside and around the micropipette. As the slanted pipette approaches the dish bottom, the gap of liquid between them shrinks and the electrical resistance rises in a predictable way, signaling a safe landing just before contact. When a cell blocks the narrow neck, the electrical path through the liquid is constricted and the resistance suddenly jumps; when the cell is pushed out, the resistance drops just as sharply. Mathematical models of these “gap,” “aspiration,” and “injection” resistances allow a computer to interpret these signals in real time and decide when to stop suction or pressure, all without visual feedback.
Putting the Robot to the Test
To see whether this approach works in practice, the researchers built a full robotic setup that combines motion control, pressure control, resistance sensing, and a host computer. They tested it on small cancer cells known as HeLa cells (around 10 micrometers across) and much larger porcine egg cells (about 150 micrometers). The system could reliably detect landing, capture, and release purely from resistance signals over a wide range of pipette sizes. In head-to-head comparisons, the robot moved HeLa cells with a 90 percent success rate—better than a previous vision-based automated method—and transported porcine eggs with a 95 percent success rate, higher than a standard microscopic approach. Operation times per cell were similar to, or faster than, human-controlled methods, mainly because the narrow neck removes the need for repeated fine-tuning of suction to position the cell.
Keeping Cells Alive and Scaling Up
Any robotic helper in the biology lab must avoid harming the cells it handles. After transport by the new system, both HeLa cells and porcine egg cells were cultured for a day and stained with a dye that lights up only in living cells. Survival rates for the new method matched or closely approached those from traditional microscope-guided transfer and from untouched control cells, indicating that the gentle mechanical squeeze at the narrow neck does not noticeably reduce cell viability when thresholds are set correctly. Looking ahead, the authors suggest that the same resistance-based sensing and pressure control can be built into compact microfluidic chips. That would allow many channels to operate in parallel, enabling high-throughput, fully enclosed cell handling systems suited for automated reproductive medicine, cell therapies, and long-term cell culture devices where microscopes cannot easily go.
What This Means for Future Cell Work
For non-specialists, the main message is that moving single cells no longer has to depend on a person watching through a microscope. By carefully shaping a glass tube and “listening” to how electricity flows through the surrounding liquid, a robot can feel when it has landed, grabbed a cell, and let it go—all in the dark and in tight spaces. This makes it much more practical to design closed, self-running cell culture systems that protect fragile samples, simplify clinical workflows, and bring laboratory-quality cell manipulation closer to automated, factory-like operation.
Citation: Zhao, Q., Liu, M., Zhu, R. et al. Robotic cell transportation system based on micropipette resistance modeling. Microsyst Nanoeng 12, 65 (2026). https://doi.org/10.1038/s41378-026-01193-5
Keywords: cell manipulation, micropipette, robotics, microfluidics, automation