Clear Sky Science · en
Boron-doped diamond solution-gate field-effect transistor (BDD-SGFET) biosensor for gene mutation detection
Why tiny chips and tiny DNA changes matter
Many cancers begin with tiny changes in our DNA—single “letters” in the genetic code that are swapped, added, or lost. Spotting these changes early can guide treatment and even save lives, but today’s standard tests often need large machines, expert staff, and time-consuming sample preparation. This paper introduces a new kind of miniature electronic sensor, built from a special form of diamond, that can electrically read out whether a stretch of DNA is normal or carries subtle mutations linked to lung cancer.
A new kind of electronic nose for genes
The authors focus on mutations in the EGFR gene, an important marker in non-small cell lung cancer. Instead of using optical labels or complex chemistry, their device works like an “electronic nose” for DNA. It is a solution-gated field-effect transistor—essentially a very small electronic switch—whose active channel is made of boron-doped diamond shaped into slender microwires. When DNA molecules in a drop of liquid bind to the surface of this channel, their electrical charge subtly shifts the current flowing through the device. By monitoring this current, the sensor can tell whether incoming DNA strands are perfectly matched or contain errors in their base pairs.
Why diamond makes a better sensor surface
Conventional transistor-based biosensors often use silicon or metal oxides, which can corrode, drift, or generate distracting background signals in salty or acidic liquids like those found in real biological samples. Boron-doped diamond behaves differently. It has an unusually wide electrochemical “window,” meaning it produces very little unwanted current while still allowing useful signals to pass. It is also hard, chemically stable, and friendly to biomolecules. The team used computer simulations to tune the length and width of the diamond microwires, showing that making them wider and shorter improves how strongly the gate (the liquid-facing surface) can control the current. Guided by these simulations, they fabricated three-dimensional microwire structures that increase the effective surface area where DNA can attach, boosting the device’s sensitivity.
From simulation to working gene sensor
After growing a thin, highly conductive boron-doped diamond layer, the researchers carved microwires using photolithography and plasma etching, added metal contacts, and protected non-sensing regions with an insulating layer and epoxy. They then carefully studied how the device behaved in salty buffers of different acidity and strength, finding conditions—around physiological pH and moderate salt concentration—where the transistor’s response is strongest and most stable. Under these optimized conditions, the sensor reached high current levels and large transconductance (a measure of how strongly the gate controls current) while operating at low voltages, making it well suited for delicate biological measurements.
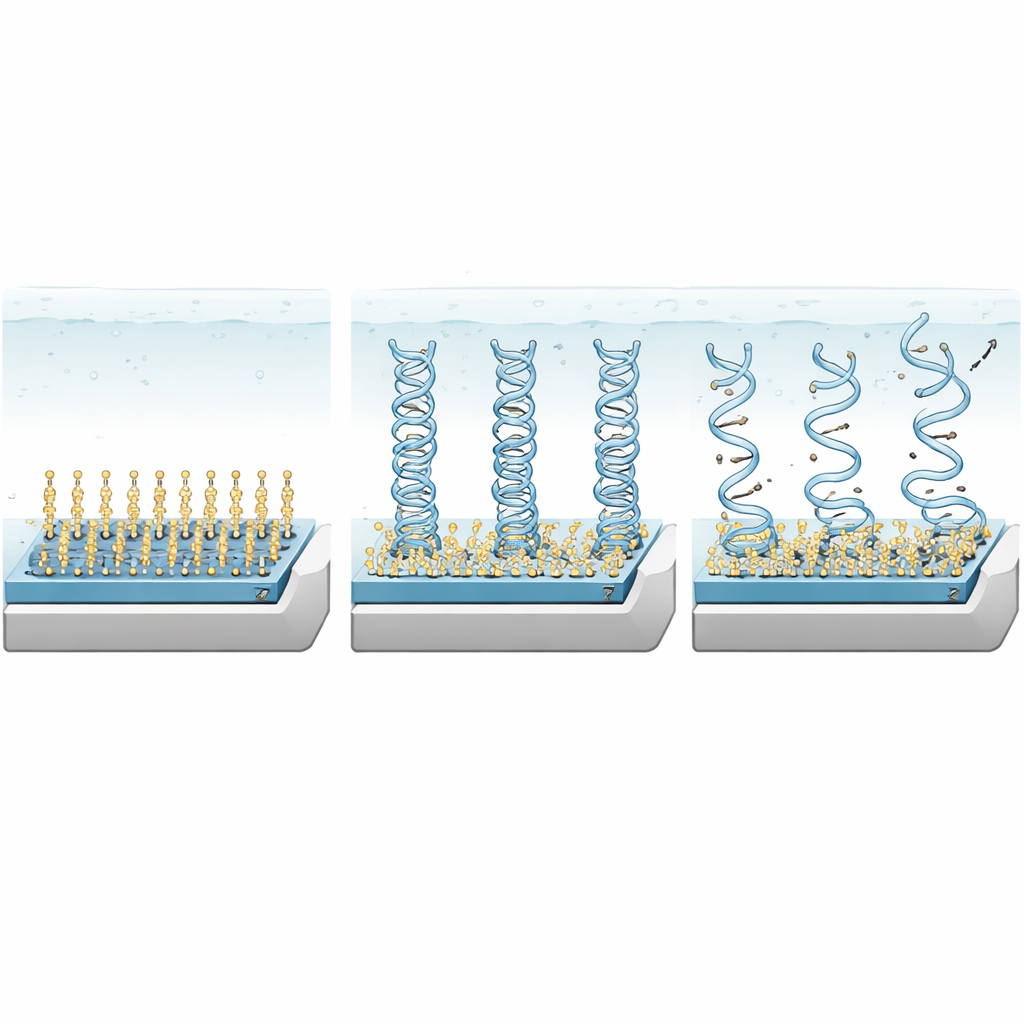
Listening for tiny differences in the genetic code
To turn the diamond chip into a gene mutation detector, the team chemically attached short “probe” DNA strands from an EGFR region that is commonly mutated in lung cancer. When a solution containing target DNA is introduced, perfectly matching strands form tight, rigid double helices close to the diamond surface, creating a dense layer of negative charges that markedly alters the channel current. If the target DNA contains one or more mismatched bases, the resulting double strands are looser, more flexible, and partly frayed. Their negative charges sit farther from the surface and are more spread out, leading to a smaller change in current. By tracking how the current‑versus‑voltage curve shifts, the device can not only detect DNA down to 10 picomolar concentrations but also distinguish sequences with two, four, or even eight mismatched bases.
Robust performance in messy, real-world conditions
Beyond sheer sensitivity, a practical medical sensor must be stable, repeatable, and resistant to interference from other molecules. The researchers repeatedly cycled the device through DNA binding and release steps and found that its responses remained highly consistent. They also monitored performance over days of storage, observing only moderate signal decline, and tested behavior in the presence of a positively charged protein that might otherwise clog or confuse the surface. The diamond microwire sensor maintained its ability to separate normal from mutated DNA even with this added biological “noise,” demonstrating strong anti-interference capabilities and reliable operation.
What this means for future cancer testing
In everyday terms, the authors have built a tiny, durable, diamond-based electronic chip that can feel the difference between correctly paired DNA and strands that hide cancer-related mutations, all without labels or bulky optics. Its combination of high sensitivity, ability to resolve even small numbers of base mismatches, and robustness in complex solutions suggests a promising path toward portable, point-of-care tests for genetic changes. While more work is needed to integrate such sensors into complete clinical devices, this study shows how carefully engineered diamond microwire electronics could become a powerful new tool for earlier, simpler detection of disease-driving gene mutations.
Citation: Lin, Z., Zheng, Y., Chen, Y. et al. Boron-doped diamond solution-gate field-effect transistor (BDD-SGFET) biosensor for gene mutation detection. Microsyst Nanoeng 12, 89 (2026). https://doi.org/10.1038/s41378-026-01184-6
Keywords: gene mutation detection, diamond biosensor, field-effect transistor, EGFR lung cancer, DNA mismatch sensing