Clear Sky Science · en
Microfluidic continuous flow production of noble bimetallic nanoparticles stabilized on evolvable polymer microspheres for confined synergistic catalysis
Turning Tiny Plastic Spheres into Pollution-Fighting Helpers
Industrial wastewater often contains stubborn toxic chemicals that are hard to break down and expensive to treat. This study shows how engineers can build tiny, hollow plastic beads that carry precious metal nanoparticles inside them, then mass-produce these beads in a small spiral device to clean up water more efficiently. The work blends chemistry, materials science, and microfluidics to turn hazardous pollutants into useful products while using less time, energy, and chemicals than many traditional methods. 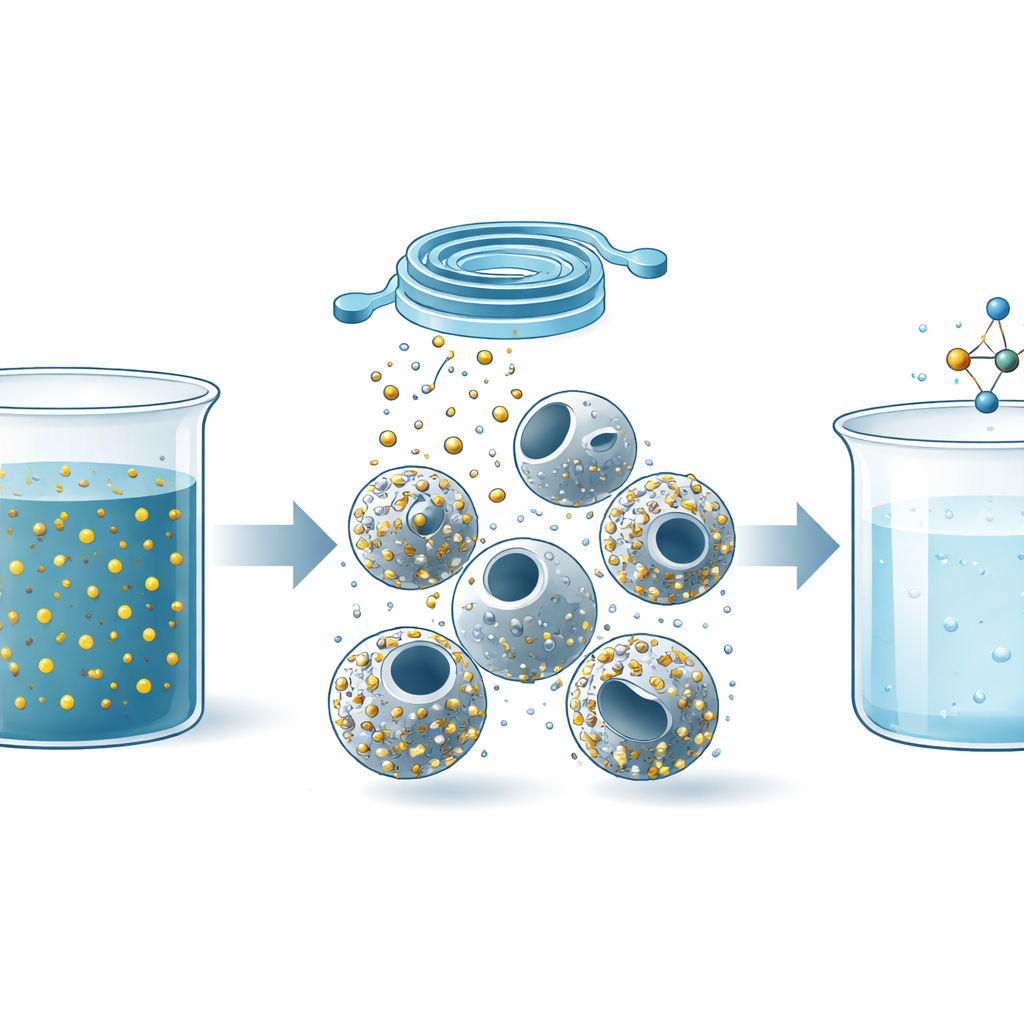
Building Tiny Hollow Beads
The researchers began with polystyrene, the same basic plastic found in foam cups, and reshaped it into microscopic spheres with empty interiors. By placing solid polystyrene beads in carefully chosen mixtures of water and ethanol and gently heating them, they caused solvent molecules to move in and out of the plastic. This motion pushed material from the center toward the outer shell, gradually creating a hollow core. By tuning the ratio of water to ethanol and the aging time, they could steer the beads through a sequence of shapes—from solid spheres to dimples, bowls, and finally fully hollow shells with very uniform sizes.
Carving Openings for Better Access
To make the beads even more useful, the team introduced a small amount of toluene, a solvent that swells polystyrene. When it concentrated in the dimpled areas of the shells, it stretched and weakened those spots until they ruptured, creating a single well-defined opening in each hollow sphere. These “open-hole” beads combine a large internal surface area with a direct entrance, forming tiny chambers where reactions can happen efficiently. Because the shapes form spontaneously from simple solvent changes rather than from complex templates or surfactants, the process is relatively clean, fast, and scalable.
Loading Precious Metals in a Spiral Channel
Next, the authors needed to decorate these hollow beads with noble metal nanoparticles—silver, gold, and platinum—because these metals are powerful catalysts. Instead of mixing everything in a big batch, they devised a continuous-flow strategy using a spiral microchannel made of soft polymer. Streams containing polystyrene beads, metal salts, and stabilizers were pumped through this narrow, winding channel. As they flowed, silver or combinations of silver–platinum and silver–gold nanoparticles formed and were drawn onto the bead surfaces by electrostatic attraction and gentle reduction chemistry. Within minutes, the beads emerged from the device coated with evenly spaced metal nanoparticles both inside and outside, something that would normally take many hours and often leads to clumping.
Turning a Toxic Dye into a Valuable Product
To test how well these composite beads work as catalysts, the team chose a common model pollutant: 4-nitrophenol, a toxic compound often found in industrial wastewater. In the presence of a reducing agent (sodium borohydride), noble metal nanoparticles can help convert 4-nitrophenol into 4-aminophenol, a useful chemical building block for medicines and dyes. The researchers found that beads carrying only silver already sped up this reaction, but beads carrying two metals together—silver–platinum or silver–gold—were much more effective. The best performer was the open-hole hollow bead loaded with silver–platinum nanoparticles, which achieved a high reaction rate and kept working for at least five cycles with almost no loss of activity. The hollow architecture concentrates reactants near the metal surfaces, and the two metals share the work: one binds the pollutant well, while the other generates highly active hydrogen species. 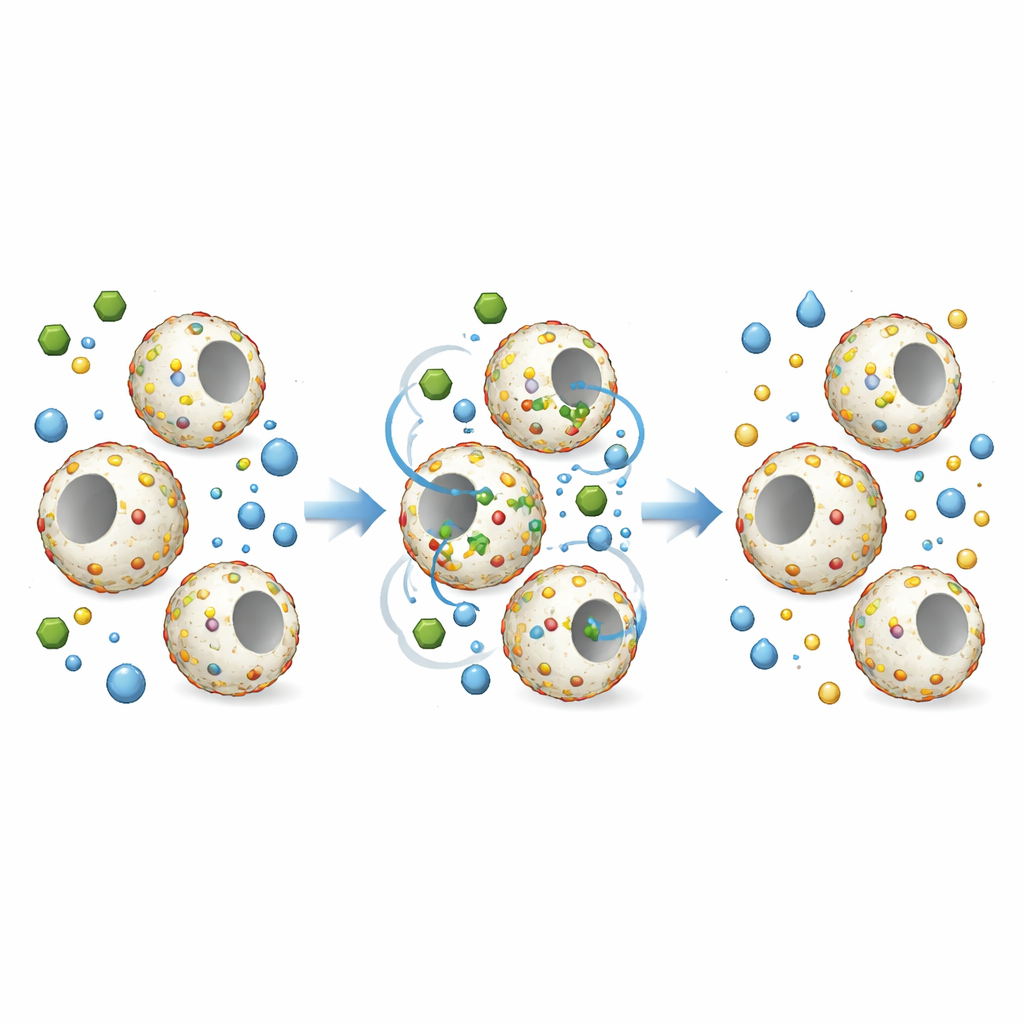
From Wastewater Problem to Reusable Solution
Overall, the study demonstrates a compact and controllable way to make large quantities of finely engineered catalyst beads, simply by adjusting solvent mixtures and flowing ingredients through a spiral microreactor. These hollow, open-hole polystyrene spheres, studded with pairs of noble metals, can rapidly convert a stubborn toxic pollutant into a valuable product and then be separated and reused. For non-specialists, the key message is that by carefully shaping materials on the microscale and guiding how different components assemble in flow, it becomes possible to clean water more efficiently, reduce waste, and recover useful chemicals from streams that would otherwise be environmental burdens.
Citation: Ma, L., Hou, J., Luo, Z. et al. Microfluidic continuous flow production of noble bimetallic nanoparticles stabilized on evolvable polymer microspheres for confined synergistic catalysis. Microsyst Nanoeng 12, 99 (2026). https://doi.org/10.1038/s41378-026-01176-6
Keywords: microfluidic catalysis, hollow polymer microspheres, bimetallic nanoparticles, wastewater treatment, 4-nitrophenol reduction