Clear Sky Science · en
A comprehensive review of hydrogen sensor for thermal runaway monitoring: fundamentals, recent advancements, and challenges
Keeping Batteries Safe Before Trouble Starts
Lithium-ion batteries power our phones, cars, and energy storage farms, but they can fail in dramatic ways if they overheat. This review article explains how a tiny, invisible gas—hydrogen—can give an early warning before a battery bursts into flame. It walks readers through what happens inside a battery during a dangerous event called thermal runaway, why hydrogen appears first, and how new generations of miniature hydrogen sensors could spot trouble in time to prevent fires and explosions.
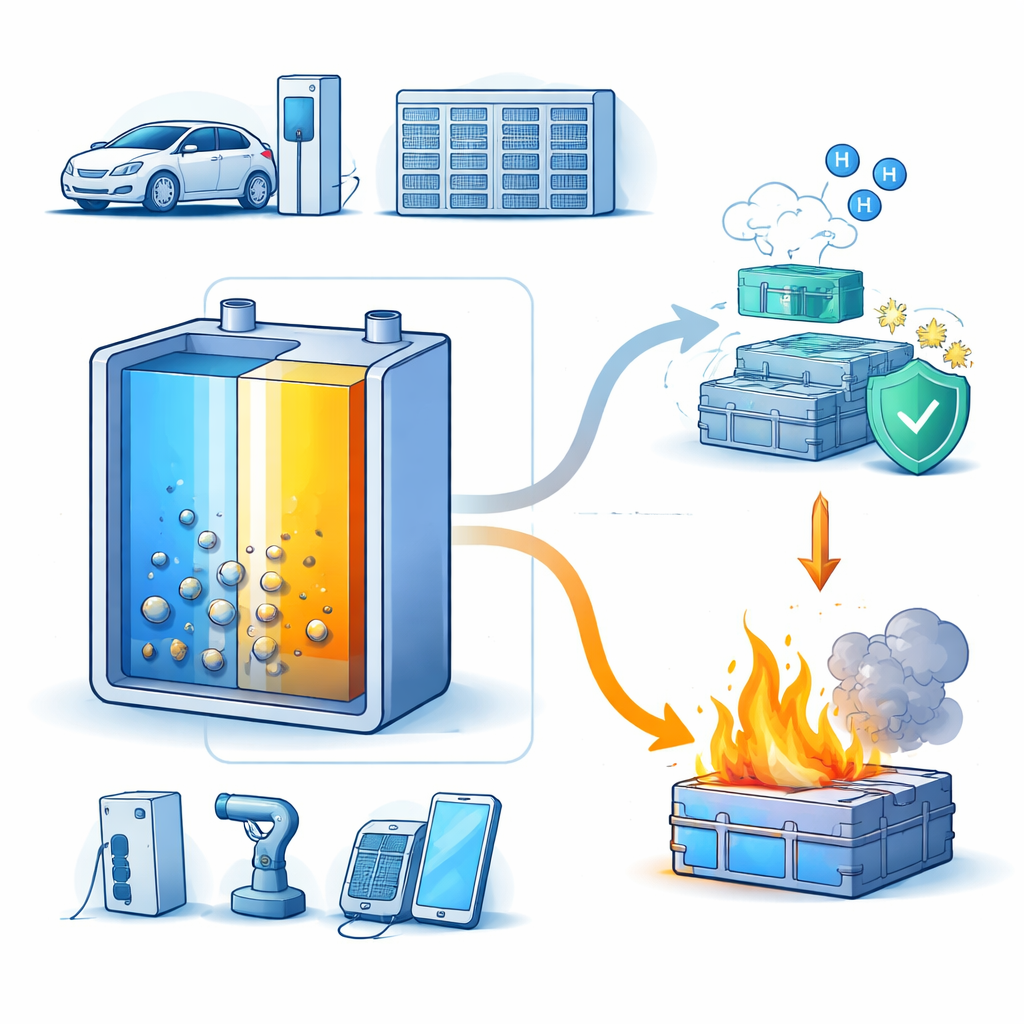
What Happens When a Battery Overheats
Inside a lithium-ion battery, thin layers of materials shuttle lithium back and forth to store and release energy. Under abuse conditions—such as crushing, hard impact, overcharging, or overheating—the cell can enter thermal runaway. In this process, internal reactions generate heat faster than it can escape. The authors describe three escalating stages: first, the battery shifts from normal to abnormal operation and its temperature starts to climb; second, protective layers and separators break down, releasing heat and gases; finally, flammable liquid components can ignite, leading to fire and even explosion. As one cell fails, it can trigger neighboring cells, turning a single fault into a large-scale accident.
Hydrogen as the Earliest Red Flag
As thermal runaway gets going, the electrodes, electrolyte, and protective films inside the battery decompose and release a cocktail of gases: hydrogen, carbon dioxide, carbon monoxide, hydrocarbons, and small amounts of corrosive species. Careful measurements with laboratory instruments show that hydrogen nearly always appears first, sometimes many minutes before the battery reaches a point of no return. Hydrogen is also linked to the growth of tiny lithium metal spikes, or dendrites, which can pierce separators and cause internal short circuits. Because hydrogen is both the earliest and a very specific sign of emerging damage, the authors argue it is one of the most powerful markers for early-warning systems in electric vehicles and energy storage units.
Why Chemiresistive Sensors Stand Out
There are many ways to monitor a battery—watching its voltage, measuring its temperature, or tracking pressure changes as it swells. But voltage often changes only after severe damage, surface temperature lags far behind the cell’s hot core, and pressure sensors can miss certain fault modes. In contrast, gas sensors respond directly to the first puffs of internal gas. This review focuses on chemiresistive hydrogen sensors, tiny devices whose electrical resistance changes when gas molecules touch their surface. They can be made cheaply, integrated onto microchips, and placed close to or even inside cells. The paper explains how different material families—precious metals like palladium, metal oxides, carbon nanostructures, ultrathin two-dimensional crystals, and wide band-gap semiconductors—each offer distinct trade-offs in speed, sensitivity, stability, and operating temperature.
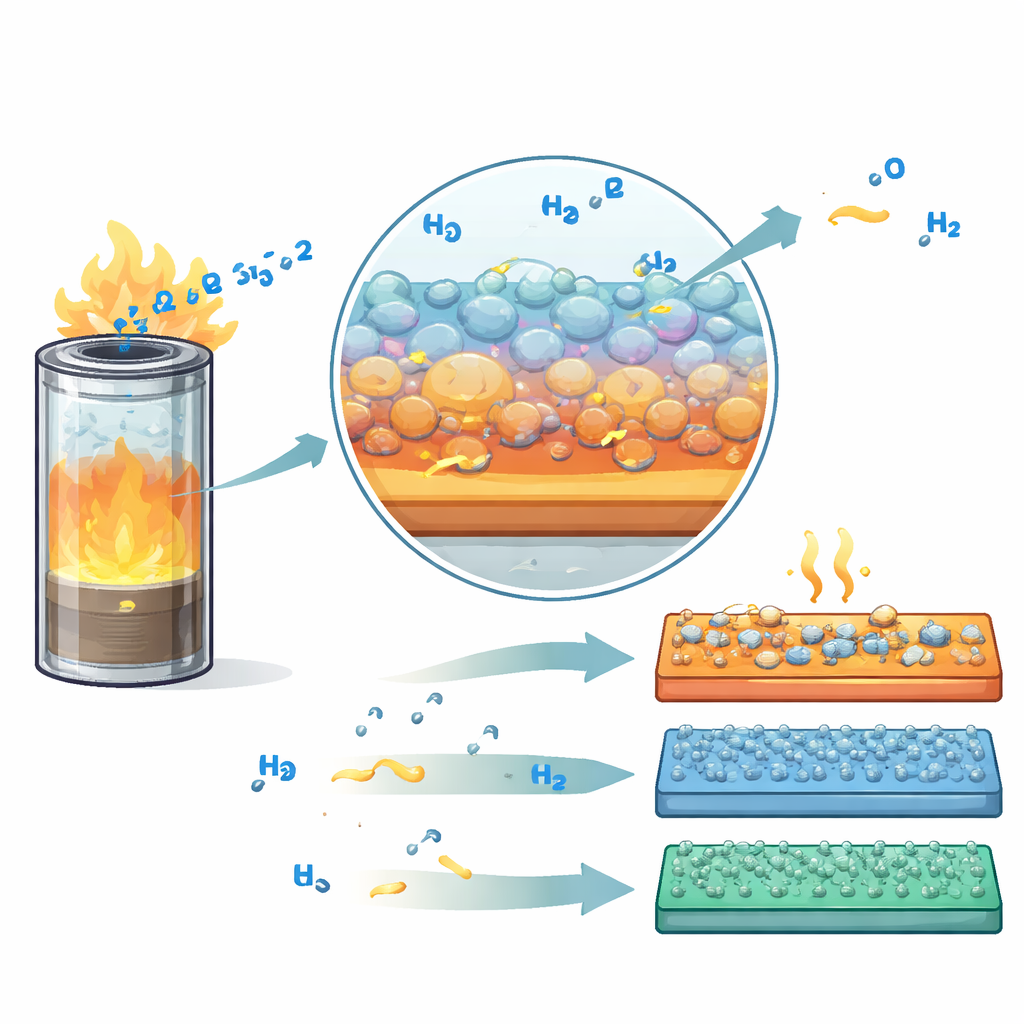
Engineering Materials to See Hydrogen Faster
Much of the review explores how to sculpt matter at the nanoscale so that it “feels” hydrogen more sharply and more quickly. For palladium-based sensors, shrinking particles, carving controlled nanogaps, and alloying with other metals tame unwanted phase changes and hysteresis that would otherwise blur the signal. For metal oxides, researchers tailor crystal facets, introduce oxygen vacancies, and build porous networks to give hydrogen more landing spots and shorter paths. Decorating these oxides or carbon and 2D materials with tiny clusters or even single atoms of noble metals like palladium and platinum lowers the energy barrier for hydrogen to react, speeding up response and recovery. Clever device structures, micro-heaters, and even machine-learning algorithms that extrapolate from the first fraction of a second of data push total detection times toward the one-second goal set by the U.S. Department of Energy.
From Laboratory Prototypes to Real-World Guardians
The authors emphasize that early-warning sensors for batteries must be not just sensitive, but also selective, durable, and inexpensive. Real packs operate over wide temperature and humidity ranges and contain many interfering gases that can foul catalysts or mask the hydrogen signal. Promising strategies include molecular sieving layers that let hydrogen through while blocking larger molecules, passivation shells that protect fragile 2D materials, and multi-sensor arrays whose combined outputs are interpreted by artificial intelligence. Ultimately, the article concludes that chemiresistive hydrogen sensors—especially when combined with temperature, voltage, and pressure data—are poised to become key guardians of battery safety, offering precious extra minutes to intervene before a smoldering cell turns into a fire.
Citation: Liu, L., Guo, C., Wang, Y. et al. A comprehensive review of hydrogen sensor for thermal runaway monitoring: fundamentals, recent advancements, and challenges. Microsyst Nanoeng 12, 108 (2026). https://doi.org/10.1038/s41378-026-01171-x
Keywords: hydrogen sensors, thermal runaway, lithium-ion batteries, chemiresistive gas sensing, battery safety monitoring