Clear Sky Science · en
Boltzmann luminescent nanothermometry: mechanistic criteria and predictive design of thermally coupled levels
Taking the Temperature of the Tiny World
Knowing exactly how hot something is at the scale of cells, microchips, or tiny reactors is crucial, but ordinary thermometers are far too bulky and intrusive. This paper explores a new way to measure temperature using glowing nanoparticles whose color balance shifts with heat. By uncovering the rules that govern this glow, the authors turn a once trial‑and‑error technique into a predictable, designable tool for future biomedical devices, advanced batteries, and space technology.

Light as a Heat Gauge
In luminescent nanothermometry, light replaces wires and metal probes. Special nanoparticles doped with rare‑earth atoms are illuminated with a laser, and they respond by emitting light of different colors. Two closely spaced internal energy states act like neighboring shelves where electrons can sit. As temperature rises, more electrons hop to the higher shelf. Because each shelf produces light of a slightly different color, the ratio of their brightness directly reflects the temperature. This makes a “self‑calibrating” thermometer that is resistant to changes in laser power or particle amount, a big advantage for measurements deep in tissue or inside sealed devices.
Why Simple Theories Fall Short
The standard explanation says that the balance of electrons between the two shelves follows a textbook rule known as the Boltzmann distribution. In practice, however, many materials that should obey this rule simply do not. The color ratio curves bend the wrong way, different labs report conflicting sensitivities, and the same rare‑earth ion looks reliable in one crystal but not in another. The authors show that the culprit is often ignored energy levels lurking just below the two chosen shelves, as well as competing non‑glowing pathways that drain energy away. When these hidden levels sit too close, they upset the delicate balance between thermal hopping and light emission, breaking the simple law that designers have relied on.
New Rules for Building Better Light Thermometers
To tame this complexity, the team builds a detailed population‑dynamics model that tracks how electrons move among energy levels, emit light, or lose energy as vibrations in the host crystal. From this, they define a “thermal coupling window” that specifies the temperature range over which the color ratio truly follows Boltzmann behavior. A striking design rule emerges: for stable operation, the nearest lower energy level must lie at least about twice as far below the lower shelf as the gap between the two shelves themselves. If this condition is not met, the lower level behaves like a leak, and the thermometer becomes unreliable. The authors also connect the key energy gap to simple measures of the chemical bonds in the host material, introducing a splitting factor that links microscopic bonding to macroscopic sensitivity. This turns host selection from guesswork into something that can be estimated in advance.
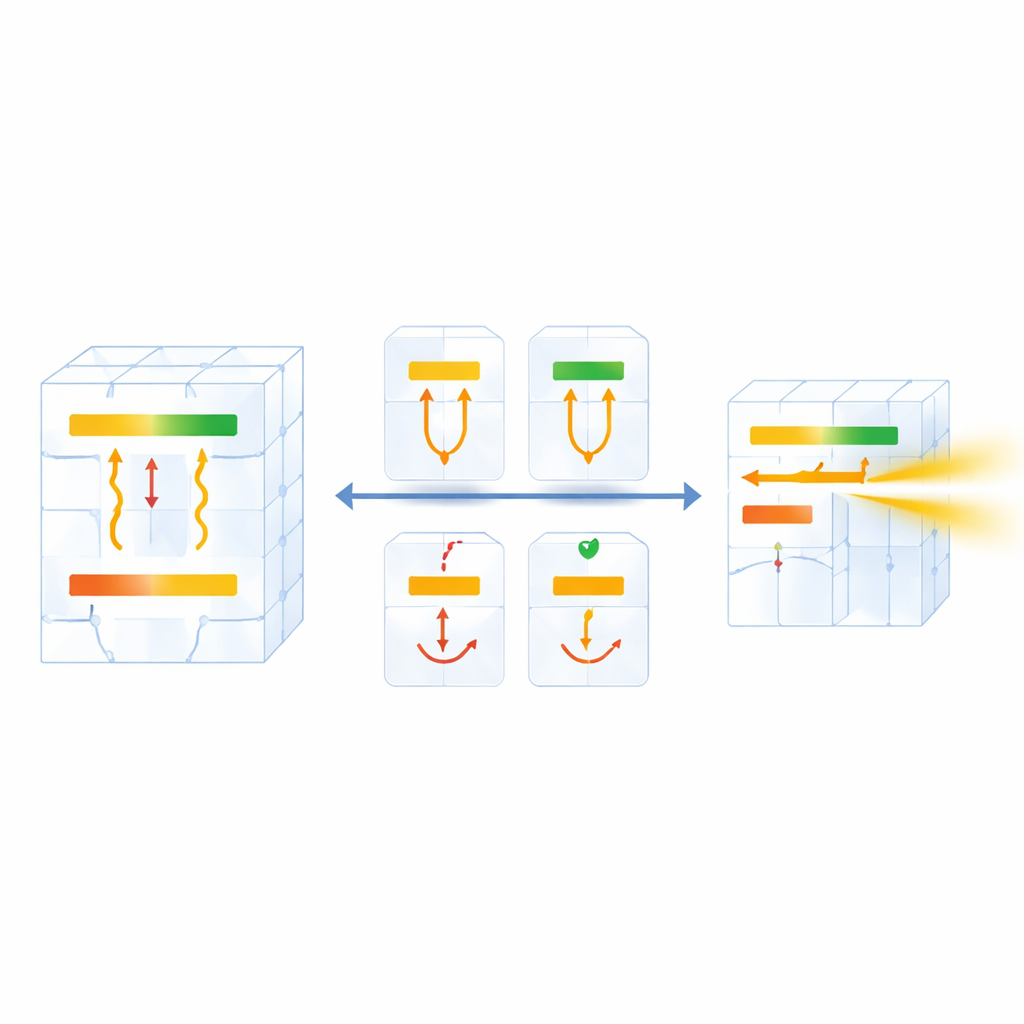
Engineering Sharper and Smarter Glow
Armed with these guidelines, the researchers go beyond passive material choice and actively reshape the energy landscape. By slightly distorting a fluoride crystal with extra lithium ions, they fine‑tune how the rare‑earth levels split, effectively widening the critical energy gap and boosting sensitivity beyond what the host alone would allow. They then combine two different rare‑earth ions, one whose emission fades with heat and another whose emission strengthens, so that their light intensities change in opposite directions. This dual‑color strategy dramatically amplifies the change in ratio with temperature, achieving record sensitivities of more than six percent change per kelvin and temperature resolution better than a tenth of a degree in the relevant range.
From Theory to Flexible Heat‑Sensing Patches
To show that these ideas work outside the lab bench, the team embeds their optimized particles into an ultrathin, flexible silicone patch. The film glows bright green under a modest infrared laser, despite being only about two tenths of a millimeter thick. Because it bends and conforms, it can be wrapped onto curved glassware or delicate components. Mounted inside a reaction flask, the patch tracks tiny temperature fluctuations during simulated nanoparticle synthesis without disturbing the sealed environment. Its readings differ from a reference thermocouple by less than a degree and remain highly repeatable over many heating–cooling cycles. In everyday terms, the work lays out a recipe for building small, bright, and accurate light‑based thermometers that can be tailored to specific tasks, opening the door to precise heat mapping in places where traditional sensors simply cannot go.
Citation: Li, K., Zhao, J., Jia, M. et al. Boltzmann luminescent nanothermometry: mechanistic criteria and predictive design of thermally coupled levels. Light Sci Appl 15, 181 (2026). https://doi.org/10.1038/s41377-026-02260-2
Keywords: luminescent nanothermometry, rare-earth nanoparticles, temperature sensing, upconversion phosphors, flexible thermal sensors