Clear Sky Science · en
Organic small-molecule NIR-II fluorophores for tumor phototheranostics
Light That Sees Deep Inside the Body
Doctors have long dreamed of a way to see cancers deep inside the body and treat them at the same time, using only beams of light and tiny drug-like molecules. This review article explains how a new class of glowing compounds, which shine in a special "second" near‑infrared color band, could bring that vision much closer to reality. By slipping through tissue with less scattering and glare than visible light, these dyes promise sharper images, gentler treatments, and more precise surgery for tumors that are otherwise hard to detect and remove.
A New Color Window for Medicine
Most hospital imaging is built around X‑rays, ultrasound, or visible light. But visible light is easily scattered and absorbed by blood and other pigments, which blurs images and limits how deep doctors can see. The dyes described here emit light in a region called NIR‑II, just beyond what our eyes can see. In this zone, tissues are more transparent and natural background glow is lower, so cameras can pick up clearer signals from several centimeters below the surface. That means blood vessels, lymph nodes, and tumors can be tracked in real time, even during surgery, with much higher contrast than older near‑infrared dyes such as indocyanine green.
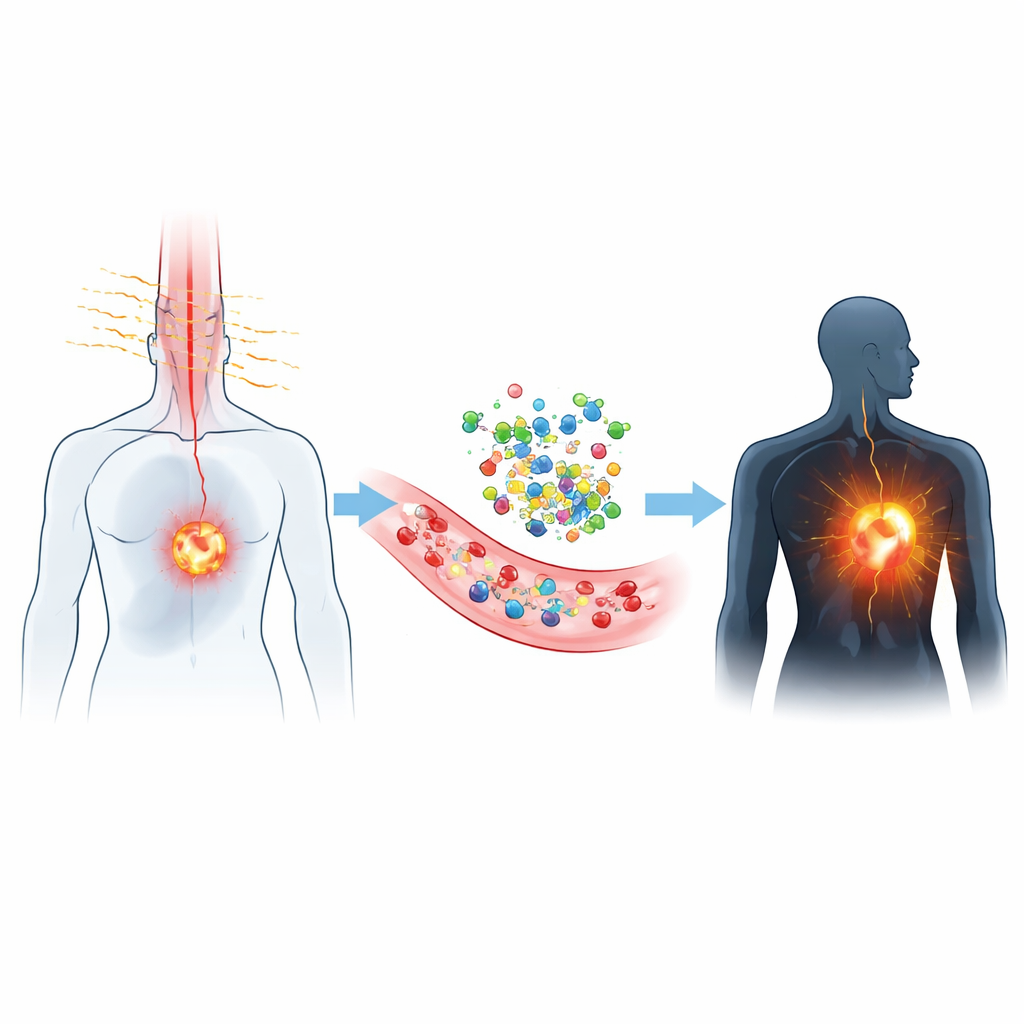
Tiny Tailor‑Made Light Bulbs
These advances rely on exquisitely engineered small molecules that act like microscopic light bulbs. Chemists build them on a few recurring frameworks—such as cyanine, benzobisthiadiazole, BODIPY, xanthene, cyano‑rich scaffolds, and even compact metal complexes—and then fine‑tune their behavior by adding or swapping side groups. By lengthening or twisting parts of the backbone, strengthening electron‑pushing or electron‑pulling segments, or forcing molecules to adopt more rigid shapes, they can shift color deeper into the NIR‑II region, brighten the glow, or convert more of the absorbed light into heat. Other designs let the dyes cluster together in tiny particles that become brighter—not dimmer—when packed, an effect known as aggregation‑induced emission.
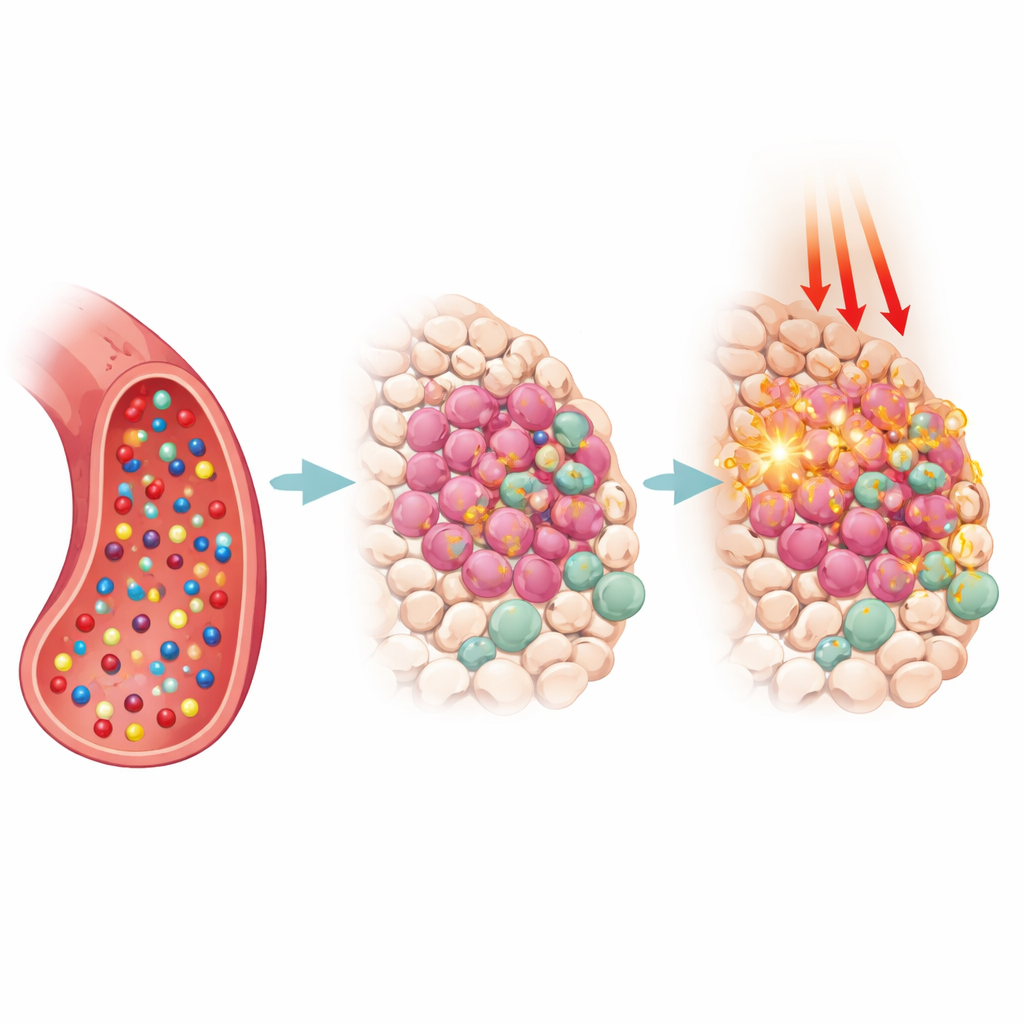
Smart Probes That Switch On Only at Tumors
One of the most powerful ideas in this field is to make the dyes respond only when and where disease is present. Many NIR‑II probes are now "activatable": they remain dim in the bloodstream but switch on inside a tumor’s acidic pocket, in thickened or sluggish fluids, or when they meet telltale chemicals such as glutathione, hydrogen sulfide, nitric oxide, or disease‑linked enzymes. Others carry small homing tags that latch onto structures on cancer cell surfaces, the tumor’s blood supply, or specific cell compartments like mitochondria. By combining smart chemistry with biological targeting, researchers dramatically boost contrast, reduce false alarms from the liver and other organs, and open the door to tracking subtle changes in tumor chemistry over time.
Imaging, Heating, and Killing—All with One Agent
Beyond simple imaging, many of these molecules double as treatment tools. When illuminated, some funnel their energy into oxygen to form reactive species that poison cancer cells (photodynamic therapy), while others dump the energy as heat (photothermal therapy), cooking tumors from within. The review describes examples where a single NIR‑II probe guides surgeons to hidden lymph nodes, maps blood‑brain barrier leaks after stroke, visualizes kidney damage, or outlines tiny tumor vessels—and then, under controlled laser light, helps destroy the marked tissue. Some systems package chemotherapy drugs or immune‑stirring agents alongside the dye, so that light, heat, reactive molecules, and drugs all work together to shrink tumors and awaken the body’s defenses.
From Lab Benches to Hospital Rooms
While the progress is striking, the authors emphasize that real‑world use still faces hurdles. Many NIR‑II dyes lose brightness in water, are hard to formulate without clumping, or clear too slowly or too quickly from the body. Others remain permanently bright, which can blur images, or have trouble crossing the barrier that protects the brain. Future work aims to boost light output, keep molecules compact and water‑friendly, build in precise on‑off switches and targeting features, and prove that these agents are safe and effective in realistic animal models and, ultimately, patients. If these challenges can be solved, NIR‑II small‑molecule fluorophores could become key tools for earlier cancer detection, cleaner surgery, and gentler, more targeted light‑based therapies.
Citation: Xiang, D., Wang, Z., Zheng, H. et al. Organic small-molecule NIR-II fluorophores for tumor phototheranostics. Light Sci Appl 15, 173 (2026). https://doi.org/10.1038/s41377-026-02212-w
Keywords: near-infrared imaging, tumor phototherapy, fluorescent probes, molecular imaging, light-guided surgery