Clear Sky Science · en
Interferometric Image Scanning Microscopy for label-free imaging at 120 nm lateral resolution inside live cells
Seeing Living Cells Without Added Dyes
Modern biology often relies on glowing fluorescent labels to reveal the hidden architecture inside our cells. But these labels can stress cells, alter their behavior, and sometimes can’t be used at all in fragile or hard-to-engineer samples. This paper introduces a new way to watch life unfold inside living cells with high detail—without adding any dyes or genetic tags—promising gentler, longer, and more natural observations of how cells really work.
Looking at Cells by How They Scatter Light
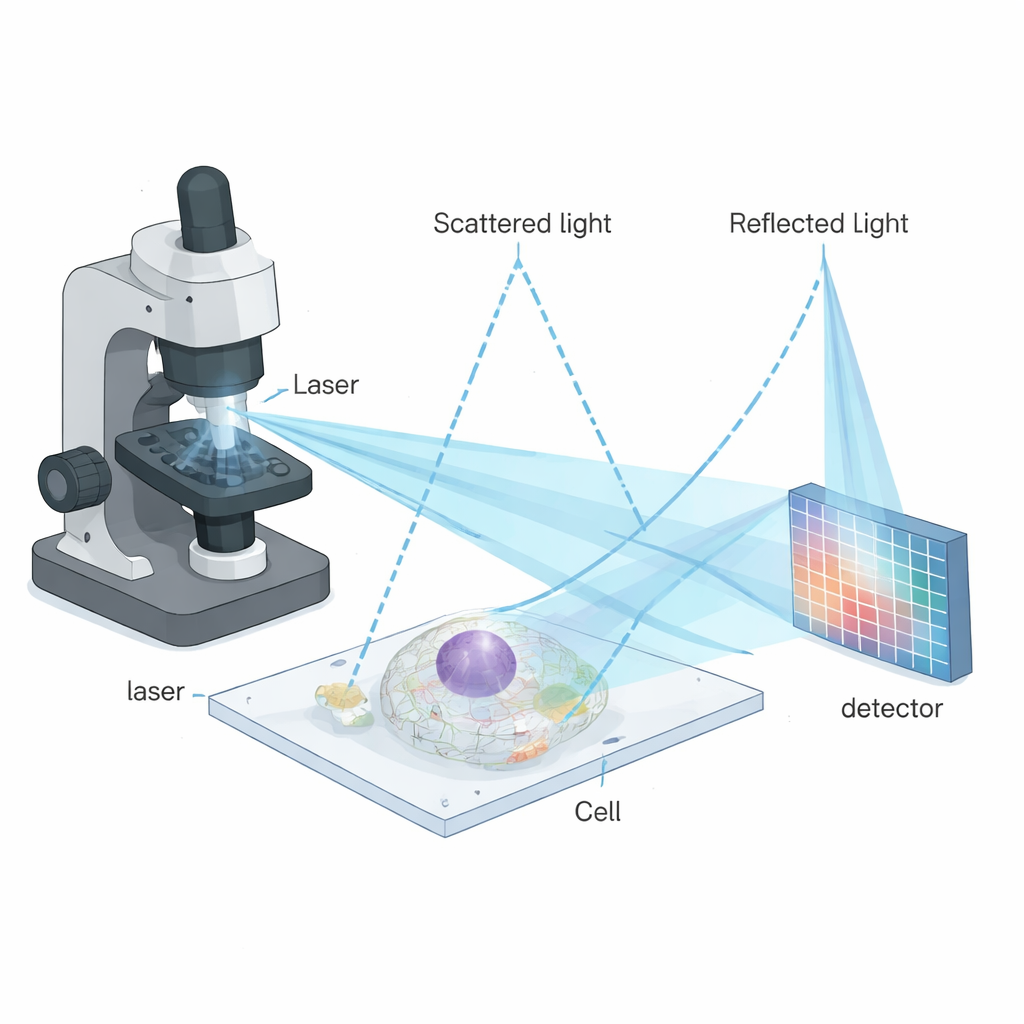
The method builds on a family of techniques that do not rely on fluorescence, but instead measure how tiny structures scatter light. One such method, interferometric scattering microscopy (iSCAT), mixes light scattered by a nanoscale object with a reference reflection from a glass surface. The resulting interference pattern is extremely sensitive to very small particles like proteins, viruses, or vesicles. iSCAT has previously worked best on simple, clean samples such as isolated particles on glass. However, applying it deep inside living cells has been difficult because cells are crowded and messy: many overlapping scattering events produce a speckled background that hides fine details.
Combining Two Ideas for Sharper, Gentler Imaging
To overcome these limitations, the authors combined iSCAT with a powerful imaging strategy called image scanning microscopy (ISM). In ISM, the sample is scanned point by point with a focused beam, and instead of using a single detector, an array of detector pixels records many slightly shifted views of each point. By cleverly realigning and combining these views, one can sharpen the final image without throwing away precious photons. The new technique—interferometric image scanning microscopy, or iISM—adapts this idea to the more complex, phase-sensitive signals of interferometric scattering. The microscope uses a blue laser, special optics to make the light polarization symmetric, and a sensitive camera to record the scattered and reflected light for every scan position. A custom computational workflow then reassigns the pixel information in a way that respects the wave-like nature of the signal, yielding images with about 120-nanometer lateral resolution, roughly twice as fine as standard diffraction-limited optics.
Smart Algorithms Turn Noisy Patterns into Clear Pictures
Because interferometric signals carry both brightness and phase information, the usual processing tricks from fluorescence imaging are not enough. The authors designed an adaptive pixel-reassignment (APR) procedure tailored for coherent light. First, they transform each small interferometric pattern into a “radial variance” map that highlights centers of symmetry without caring about whether the signal is positive or negative. This step effectively converts complicated interference fringes into images that behave more like conventional intensity pictures. Then, using open-source software, they determine how much each detector pixel’s image is shifted relative to the center and shift them back accordingly before adding them up. This refined alignment concentrates the useful signal while averaging down noise, boosting the contrast-to-noise ratio by about a factor of four compared to a tightly pinholed confocal iSCAT image, all at the same light level.
Watching Organelles and Cytoskeletons in Action
With these technical advances, the team turned to living COS-7 cells to test how well iISM performs in practice. At very low illumination power—about ten times lower per focused spot than conventional confocal microscopes typically use—they could clearly discern key organelles: the endoplasmic reticulum, mitochondria, vesicles, the actin cytoskeleton, plasma membrane, and thin leading-edge structures called lamellipodia. Because the interferometric contrast depends sensitively on vertical position, similar organelles could appear with positive or negative contrast, revealing subtle height differences of only a few hundred nanometers. By recording time-lapse sequences, they tracked vesicles moving and endoplasmic reticulum tubules remodeling over many minutes, without obvious photodamage and with no sign that the imaging itself was disturbing the cells’ behavior.
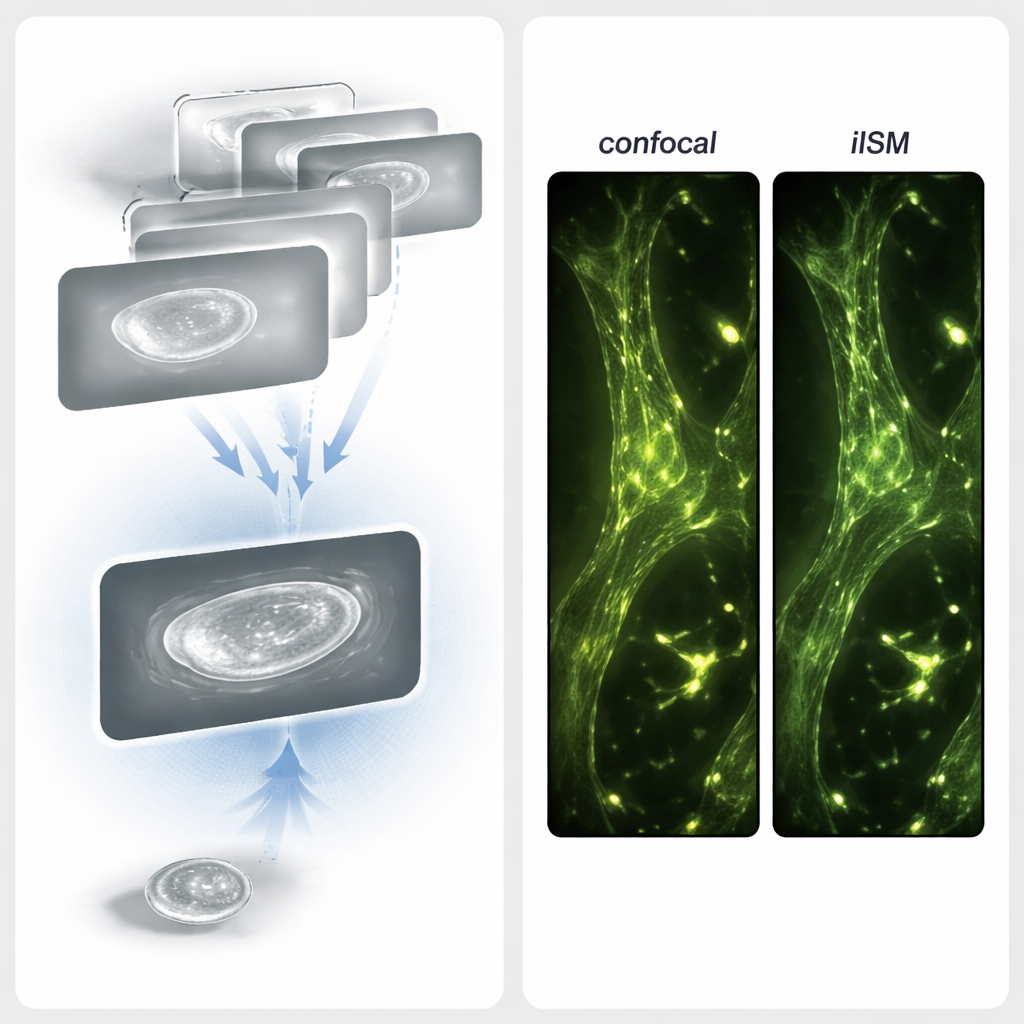
Matching Label-Free Views to Fluorescent Maps
To check that the label-free images truly reflect real cellular structures, the researchers also carried out combined iISM and fluorescence ISM measurements in fixed cells. They stained the actin cytoskeleton with a red fluorescent dye and recorded super-resolved fluorescence images alongside label-free iISM images of the same region. When they overlaid the two, bright actin filaments in the fluorescence channel lined up closely with filamentous features in the iISM images. In some regions, iISM even revealed additional scattering details—such as variations along filaments or nearby unlabeled structures like focal adhesions—that were invisible in the fluorescence channel. Together, these results show that iISM can both confirm known structures and uncover extra information about unlabeled surroundings.
A New Window Into Cells, With Less Disturbance
For non-specialists, the key message is that iISM offers a way to see fine details inside living cells without making them glow artificially. It combines the sensitivity of interferometric scattering with the sharpening power of image scanning, achieving roughly 120-nanometer resolution while using far less light than many existing microscopes. Because it is built from components already common in advanced confocal systems, it can, in principle, be added to commercial instruments. In the future, iISM could be paired with traditional fluorescence, fast detectors, or even machine-learning “virtual stains” to follow infections, cargo transport, or cytoskeletal rearrangements in cells under conditions that are closer to their natural, unperturbed state.
Citation: Küppers, M., Moerner, W.E. Interferometric Image Scanning Microscopy for label-free imaging at 120 nm lateral resolution inside live cells. Light Sci Appl 15, 129 (2026). https://doi.org/10.1038/s41377-026-02210-y
Keywords: label-free imaging, live-cell microscopy, interferometric scattering, super-resolution, cellular organelles