Clear Sky Science · en
Rapid trapping and label-free optical characterization of single nanoscale extracellular vesicles and nanoparticles in solution
Seeing the Smallest Messengers
Our bodies and environment are filled with tiny particles far too small to see with a conventional microscope. Some of these, such as nanosized bubbles released by cells, carry crucial information about health and disease. Others include plastic fragments or engineered nanoparticles in water and air. This paper introduces a new chip-based tool that can catch single nanoparticles in liquid within seconds and read out both their size and chemical makeup without any added dyes, opening the door to faster medical tests and cleaner environmental monitoring.
Why Tiny Particles Matter
Cells constantly release nanoscale packages called extracellular vesicles and other related nanoparticles. These soft, bubble-like packets can carry proteins, fats, and genetic material that reveal the state of the cell that produced them, and they are being explored as vehicles to deliver drugs. At the same time, society is grappling with man‑made nanoparticles, from air pollution to nanoplastics in the oceans. To understand which particles are helpful, harmful, or simply different from one another, scientists need ways to look at single particles in solution, determine how big they are, what they are made of, and how diverse a sample really is. Existing tools can do parts of this job, but typically only slowly, one particle at a time, or by sticking particles to surfaces and tagging them with fluorescent labels that may alter their natural state.
A New Way to Catch and Hold Nanoparticles
The authors present a platform they call interferometric electrohydrodynamic tweezers (IET), which combines electric fields, fluid motion, and advanced light scattering on a single microfabricated chip. The chip consists of a very thin gold film patterned with a regular array of microscopic holes, separated from a transparent electrode by a narrow fluid channel. When a gentle alternating voltage is applied, it generates swirling flows along the gold surface that draw nanoparticles from the surrounding liquid toward specific “stagnation zones” located between holes, where the fluid speed drops almost to zero. At these spots, a balance between the drag from the fluid and electrical forces between the particle and the surface holds individual nanoparticles near the gold film without gluing them down permanently. Thousands of such trapping sites operate in parallel, allowing many particles to be captured within seconds even when they are present at low concentrations. 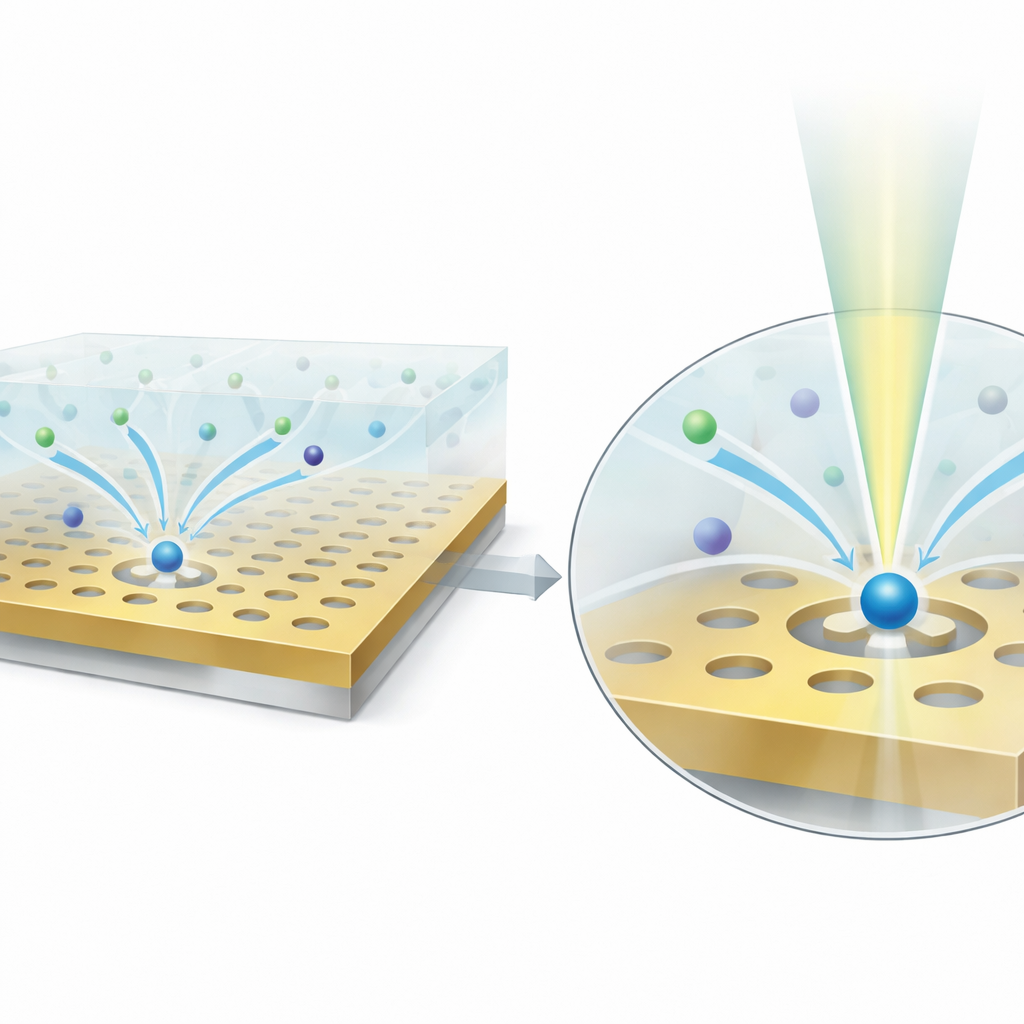
Reading Size and Shape with Light
Once particles are trapped, the IET chip uses a carefully tuned green laser to shine through the thin gold film from above. As light passes through, a small portion is scattered by each particle while the rest continues straight through the film. The camera records the interference between these two components, producing a bright‑and‑dark pattern whose contrast depends strongly on particle size and, to some extent, shape. Because the system collects forward‑scattered light, which grows nearly linearly with particle size over a broad range, the contrast signal provides a practical ruler for sizing nanoparticles. The team calibrated this relationship using plastic beads of known sizes, and could even see differences between spherical and elongated particles from the distinct patterns in their images. If the particle size is unknown, the electric field can be briefly turned off, letting the particles diffuse freely; by tracking their random Brownian motion, the researchers independently estimate their size and then correlate that with the contrast signal measured during trapping.
Fingerprinting Chemical Makeup Without Labels
Beyond size, the platform also probes chemical composition by adding a second, near‑infrared laser focused on any chosen trapping site. This light excites faint vibrational signals in the molecules that make up a trapped particle, a phenomenon known as Raman scattering. Each combination of proteins, fats, and other molecules produces a characteristic pattern of peaks in the scattered light, like a spectral fingerprint. In tests with plastic beads, the system quickly recovered the expected Raman features of polystyrene. More importantly, when the researchers trapped individual extracellular vesicles and related nanoparticles called supermeres from biological samples, they could measure their size, then record Raman spectra showing signatures of proteins, lipids, and nucleic acids. Different vesicles displayed noticeably different spectral patterns, highlighting the natural diversity of these biological messengers. 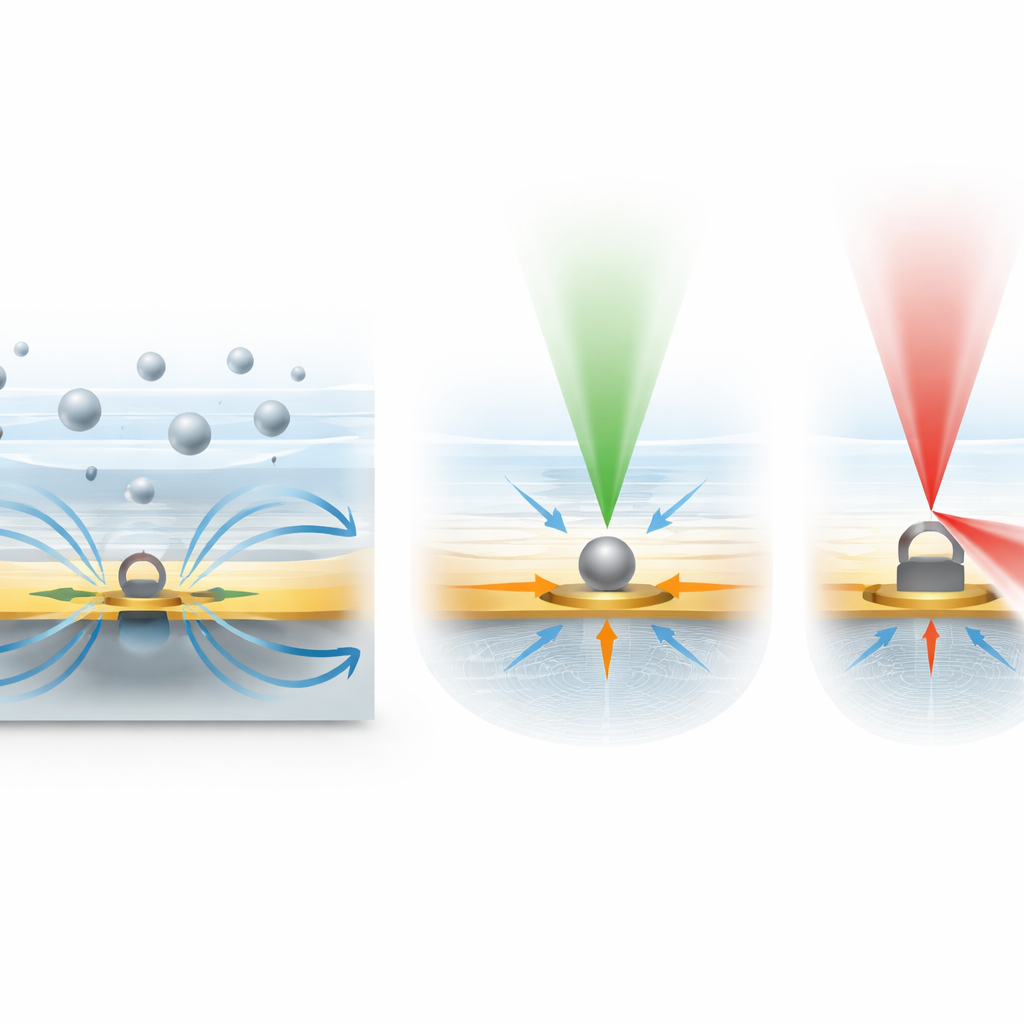
What This Means for Medicine and the Environment
By uniting rapid trapping, label‑free imaging, and chemical fingerprinting on a single chip, the IET platform offers a powerful new way to study nanoscale particles as they float freely in solution. It can capture a large fraction of available particles even at low concentration, determine their size in multiple ways, and reveal their overall molecular cargo, all within seconds rather than minutes. For biomedical research, this could help disentangle which extracellular vesicles carry specific genetic or protein messages, or assess how well drug‑loaded vesicles are prepared. For environmental science, similar measurements could distinguish between different types of nanoplastics or pollutants. Although the current system is best suited for particles larger than about 50 nanometers and low‑salt liquids, the authors outline paths to greater sensitivity and broader sample conditions. In essence, this work turns a tiny, patterned metal film into a fast laboratory for single nanoparticles, bringing detailed analysis of the invisible world one step closer to routine use.
Citation: Hong, I., Hong, C., Anyika, T. et al. Rapid trapping and label-free optical characterization of single nanoscale extracellular vesicles and nanoparticles in solution. Light Sci Appl 15, 180 (2026). https://doi.org/10.1038/s41377-026-02201-z
Keywords: extracellular vesicles, nanoparticle analysis, label-free spectroscopy, Raman tweezers, optofluidic trapping