Clear Sky Science · en
Repression of miR-29 via MYC leads to increased CD40 signaling in transformed follicular lymphoma
When Cell-to-Cell Talk Turns Dangerous
Follicular lymphoma is a slow-growing blood cancer that can smolder for years, yet in some patients it suddenly shifts gears and becomes a fast, life‑threatening disease. This study asks a simple but crucial question: what changes inside these cancer cells, and in their cellular neighborhood, to flip that switch? By following both the cancer cells’ genes and tiny RNA regulators across this transition, the researchers uncover a hidden control circuit that tunes how strongly tumor cells respond to help from nearby immune cells — and show that when this circuit fails, the cancer grows harder to treat.
A Quiet Cancer That Sometimes Accelerates
Follicular lymphoma arises from B cells, the immune cells that normally make antibodies. Most patients live with an indolent, or slow, form of the disease, but each year a fraction of cases transform into an aggressive lymphoma that behaves more like diffuse large B‑cell lymphoma. Genetic mutations had been linked to this change, yet they did not tell the whole story. The tumor cells live in a rich microenvironment full of helper T cells and other immune partners, constantly exchanging signals. The authors suspected that small regulatory molecules called microRNAs, which fine‑tune how genes are used, might be key to how cancer cells adapt to this shifting neighborhood and make the leap to a more dangerous state.
Small RNA Brakes Go Missing
To test this idea, the team performed the first matched profiling of both messenger RNAs and microRNAs in paired samples taken from the same patients before and after transformation. Comparing 11 follicular lymphoma samples with their 11 transformed counterparts, they found more than a thousand genes and 19 microRNAs that changed. One family stood out: all three members of the miR‑29 family were consistently reduced in transformed tumors. In earlier work in other blood cancers, miR‑29 had acted as a brake on cell growth and survival. Here, its loss emerged as a hallmark of transformation, hinting that turning down this microRNA family might release growth‑promoting pathways in the lymphoma cells.
A Boosted Signal From Helper Cells
Digging into the gene‑expression data, the researchers discovered that transformed tumors showed a strong rise in activity of the CD40 pathway, a signaling route that is triggered when a helper T cell engages a B cell. In healthy lymph nodes and in indolent follicular lymphoma, this interaction helps B cells grow and survive in a controlled way. Surprisingly, in transformed samples, CD40‑related genes were switched on in about 90 percent of cases, even though the overall number of T cells in the tumor areas was actually lower. Single‑cell analyses confirmed that a subset of T cells still provided the CD40‑activating signal, but the lymphoma cells themselves seemed to have become much more sensitive to that signal.
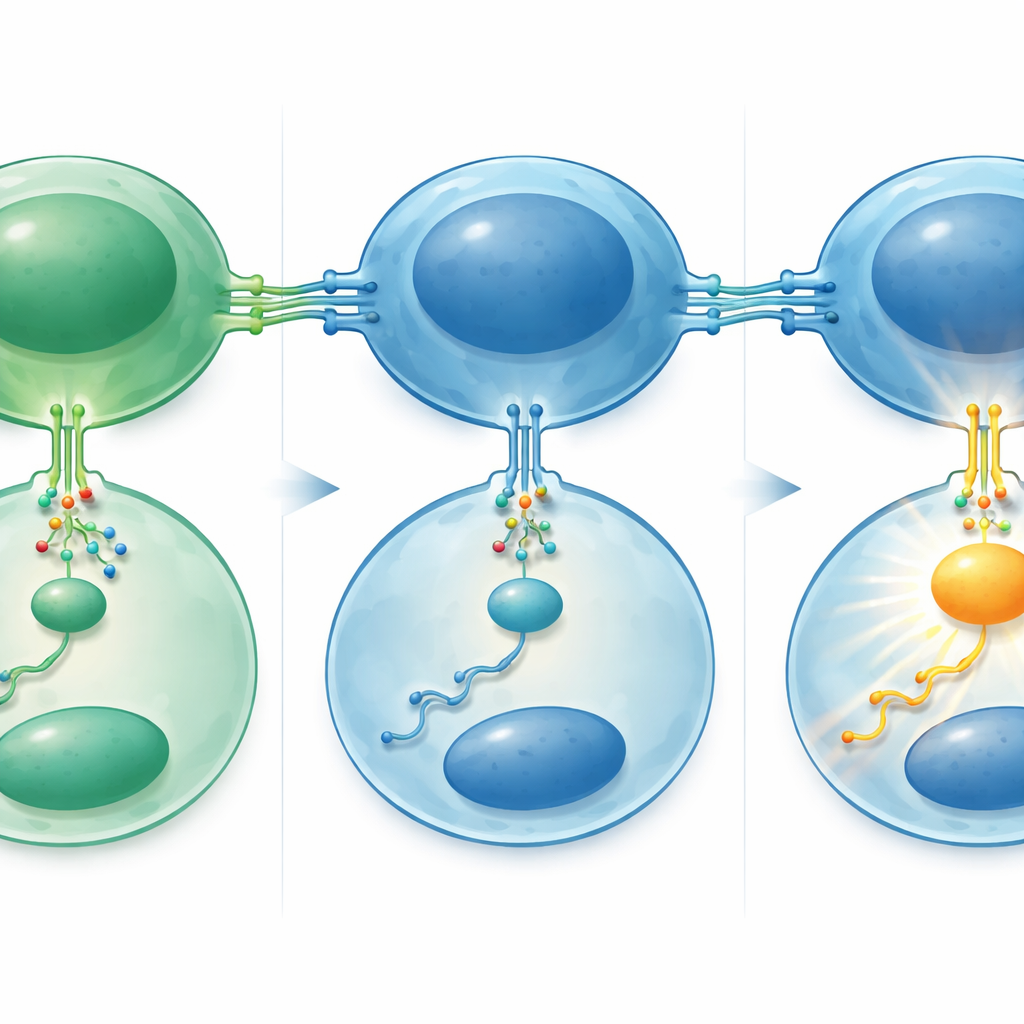
How a Three‑Step Circuit Amplifies Growth
The authors then pieced together the molecular circuit behind this heightened sensitivity. Laboratory experiments showed that miR‑29 normally keeps levels of a protein called TRAF4 in check inside B cells; TRAF4 is an internal adaptor that helps transmit the CD40 signal deeper into the cell. When miR‑29 levels were experimentally raised in lymphoma cell lines, TRAF4 levels dropped and the cells responded less strongly to CD40 stimulation, with weaker activation of downstream signaling and slower proliferation. In patient samples, the opposite pattern appeared: transformed tumors and higher‑grade follicular lymphomas had more TRAF4 and higher cell‑division rates. Another major player, the well‑known cancer‑promoting protein MYC, sat upstream. MYC levels and activity rose during transformation, and MYC was shown to bind directly to the DNA regions controlling miR‑29, acting as a switch that turns this microRNA family down.
What This Means for Patients
Putting these pieces together, the study outlines a three‑step chain: MYC activation suppresses miR‑29, loss of miR‑29 lifts the brake on TRAF4, and extra TRAF4 makes lymphoma cells hyper‑responsive to CD40 signals from the few remaining helper T cells. This amplified “help” drives stronger growth and survival, contributing to transformation. Clinically, patients whose tumors had lower levels of miR‑29 — especially the miR‑29c member — tended to have shorter survival and quicker relapses, findings confirmed in an independent treatment trial. Because miR‑29 can be measured reliably even in old, preserved biopsy samples, and because drugs that mimic miR‑29 or block CD40 signaling are already being explored, this work points toward new ways to identify high‑risk follicular lymphoma patients and to design treatments that cut the dangerous conversation between tumor cells and their immune neighbors.
Citation: Filip, D., Litzmanova, K., Michaelou, A. et al. Repression of miR-29 via MYC leads to increased CD40 signaling in transformed follicular lymphoma. Leukemia 40, 759–772 (2026). https://doi.org/10.1038/s41375-026-02868-8
Keywords: follicular lymphoma, microRNA, CD40 signaling, MYC, tumor microenvironment