Clear Sky Science · en
CHEK2 loss endows chemotherapy resistance to hematopoietic stem cells
Why some blood cells beat chemotherapy
As cancer treatments improve and more people survive chemo and radiation, doctors are discovering an unexpected side effect: years later, many survivors carry pockets of genetically altered blood cells that quietly took over parts of their bone marrow during therapy. This article asks a pointed question behind that mystery: why do certain mutant blood stem cells weather harsh cancer drugs better than normal cells, and what does that mean for future leukemia risk and treatment choices?
The hidden evolution inside our bone marrow
Our blood system is constantly renewed by a small pool of stem cells in the bone marrow. Over a lifetime, these cells gradually pick up random DNA changes. When a stem cell with a helpful change gains a survival edge, it can produce a large “clone” of descendant cells, a process called clonal hematopoiesis. This is surprisingly common in older adults and has been linked not only to blood cancers, but also to heart and other age-related diseases. Cancer therapies add intense stress to this system, killing many cells and unintentionally favoring those rare mutants that can better resist DNA damage.
A safety switch that backfires under treatment
The focus here is on a gene called CHEK2, which acts as part of the cell’s damage-sensing circuitry. In healthy cells, CHEK2 helps detect broken DNA, pause cell division, and, if the damage is too great, trigger self-destruction. Using a large-scale gene knockout screen in leukemia cells, the researchers systematically disabled thousands of genes to see which losses helped cells survive traditional DNA-damaging chemotherapy drugs. CHEK2 quickly stood out: when it was disabled, cells were far more likely to live through treatment with drugs such as cisplatin and melphalan. Without this safety switch, damaged cells continued to divide instead of stalling or dying.
Mutant stem cells that refuse to quit
To move beyond cell lines, the team engineered mice whose blood-forming stem and progenitor cells lacked Chek2. Under normal conditions, these mice produced blood in a perfectly ordinary way, suggesting the gene is not essential for day-to-day blood formation. That changed when the animals received repeated doses of chemotherapy. In that stressed setting, Chek2-deficient stem and early progenitor cells were depleted far less than their normal counterparts and gradually took over much of the blood system. These surviving cells carried more marks of DNA injury than normal cells, yet they persisted and recovered the bone marrow, illustrating a troubling trade-off: resistance to treatment at the cost of carrying extra genetic scars.
When leukemia drugs fuel resistant clones
The study went on to ask whether newer “gentler” drugs used for bone marrow disorders, known as hypomethylating agents, create similar pressures. These drugs, including azacitidine and decitabine, are generally thought of as epigenetic medicines that loosen chemical marks on DNA. The authors found that in fact they also cause a distinctive form of DNA damage by trapping an enzyme called DNMT1 directly onto DNA. In mixed cultures of normal and CHEK2-deficient cells, and in mouse stem cells grown outside the body, this damage again favored the mutants, which escaped cell-cycle arrest and death. When a separate DNMT1-blocking compound that does not damage DNA was added, the advantage of the CHEK2 mutants largely disappeared, underscoring that it is the DNA injury—not the demethylation itself—that selects for these resistant clones.
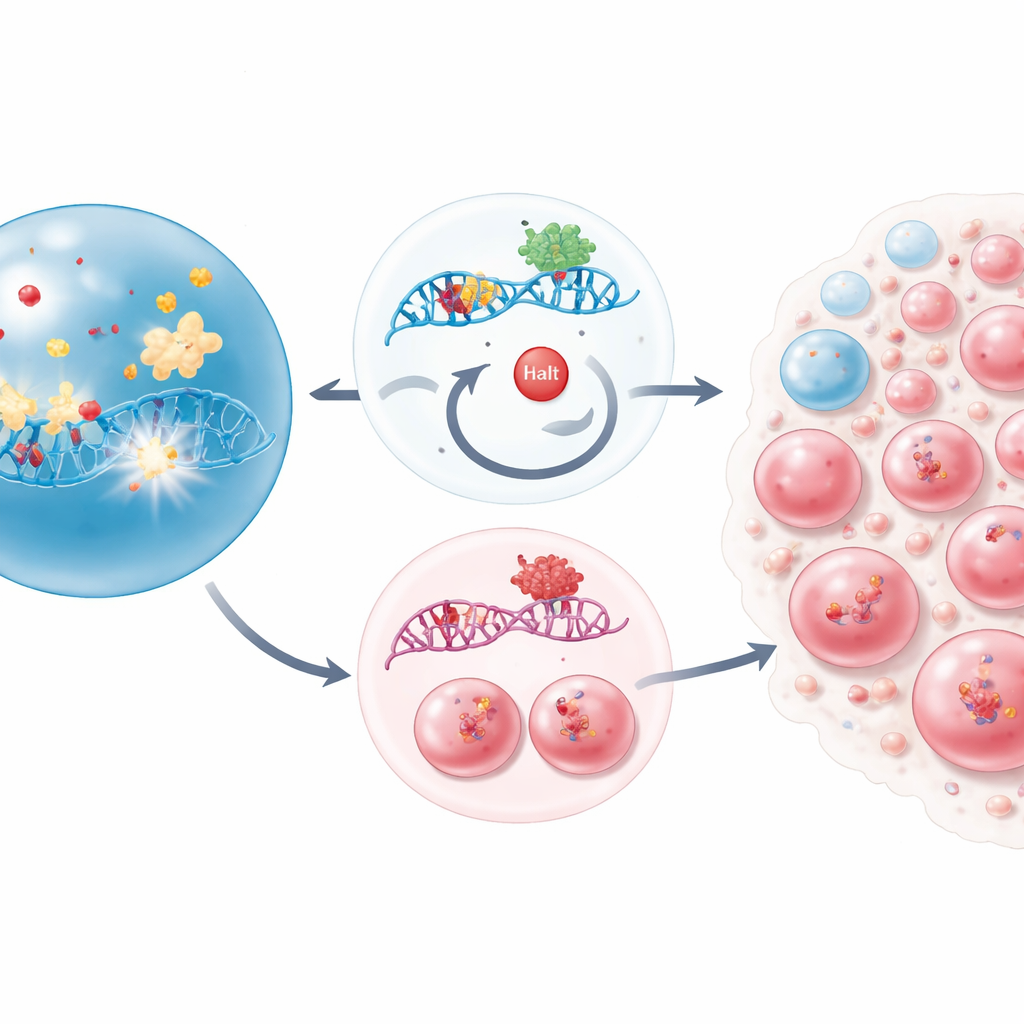
What this means for patients and future care
Taken together, the work paints a vivid picture of how a single damaged safety switch in blood stem cells can silently reshape the blood system after cancer therapy. Loss of CHEK2 allows stem cells to shrug off both classic chemotherapies and widely used bone marrow drugs, survive with lingering DNA damage, and expand into dominant clones. For patients, this helps explain why certain DNA repair gene mutations are often found in blood years after treatment and may foreshadow therapy-related leukemias. Understanding that specific drugs can unintentionally favor such mutant cells suggests that future treatment plans, and perhaps new targeted therapies, will need to account for the genetic “ecosystem” of a patient’s bone marrow, not just the cancer being treated.
Citation: Zhou, J., Hu, T., Li, D. et al. CHEK2 loss endows chemotherapy resistance to hematopoietic stem cells. Leukemia 40, 511–521 (2026). https://doi.org/10.1038/s41375-025-02850-w
Keywords: clonal hematopoiesis, CHEK2, chemotherapy resistance, DNA damage response, hematopoietic stem cells