Clear Sky Science · en
Alnuctamab, a bivalent B-cell maturation antigen-targeting T cell engager for patients with relapsed or refractory multiple myeloma: results from a phase 1, first-in-human study
Why this new cancer drug matters
For people living with multiple myeloma, a blood cancer that almost always comes back after treatment, every new option can mean more time and better quality of life. This study tests a next‑generation immune therapy called alnuctamab, designed to help the body’s own defender cells hunt down and kill myeloma cells. The research asks a practical question that matters to patients and doctors alike: can this powerful therapy be given as a simple under‑the‑skin shot, with strong anti‑cancer effects but fewer dangerous side effects?
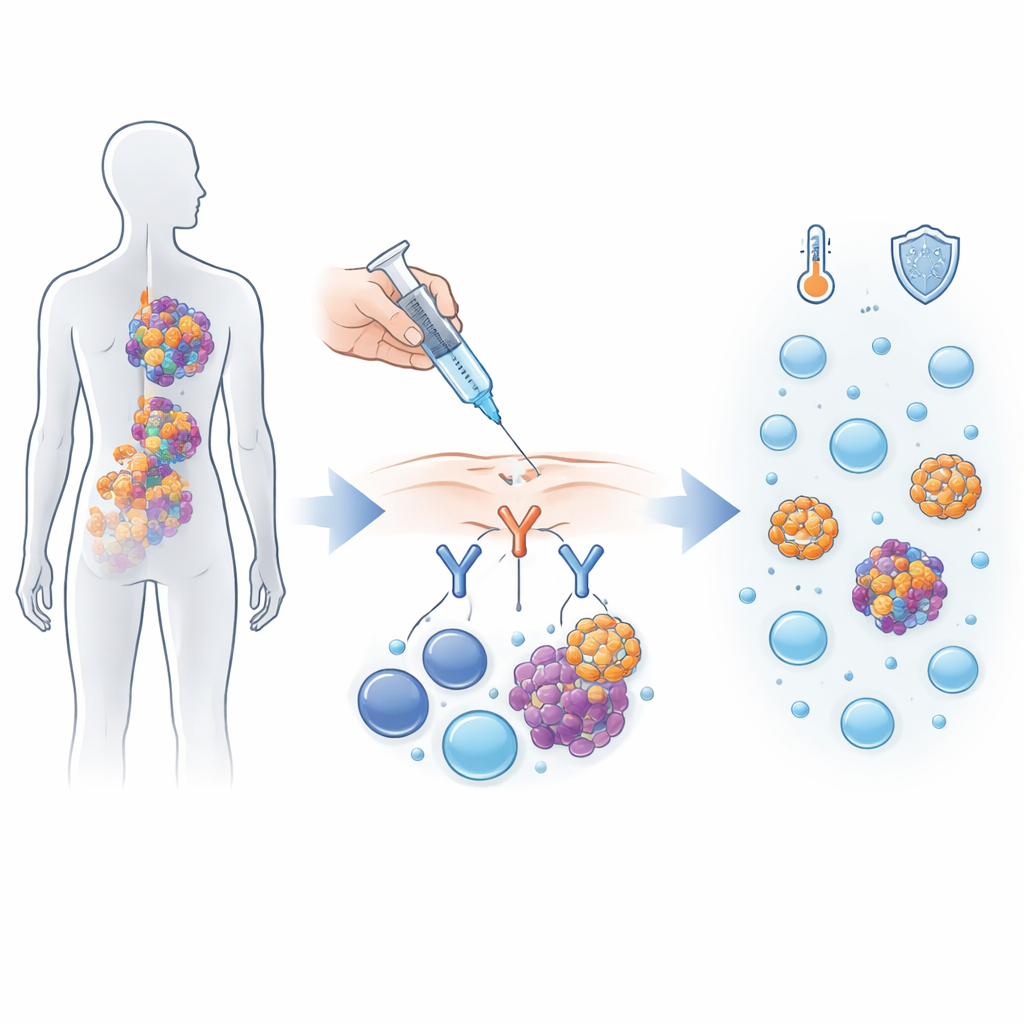
A clever way to redirect the immune system
Alnuctamab belongs to a new class of drugs that act like molecular matchmakers between cancer cells and immune cells. Multiple myeloma cells carry a marker called BCMA on their surface, while the body’s T cells carry a different marker, CD3. Alnuctamab is built with two “arms” that latch onto BCMA and one arm that latches onto CD3, pulling T cells into close contact with myeloma cells so they can attack them. In this first‑in‑human trial, 165 people with myeloma that had come back after at least three different kinds of treatment received alnuctamab, either by intravenous drip into a vein or by subcutaneous injection under the skin.
From infusion to injection
Early on, the drug was given by vein, but this approach caused more intense immune reactions, including a life‑threatening episode of a complication called cytokine release syndrome, where the immune system suddenly pours out large amounts of inflammatory signals. Because of these safety concerns and only modest benefit, the study shifted to under‑the‑skin dosing, which releases the drug more gradually. Ninety‑five patients were treated this way using a “step‑up” schedule: small starter doses followed by higher target doses, with the frequency of injections slowly reduced over time from weekly to once every four weeks if treatment continued.
How well patients did and what they faced
Among all patients who received the under‑the‑skin shots, about 59% saw their cancer shrink by at least half, and nearly half became “minimal residual disease negative,” meaning highly sensitive tests could no longer find myeloma cells in their bone marrow. At the recommended dose of 30 milligrams, responses were even stronger: about 71% responded and more than half achieved minimal residual disease negativity. Many patients maintained control of their disease for close to a year or more, even as the dosing schedule became less intense. Side effects were common but mostly manageable. Fever‑like immune reactions occurred in roughly three out of five patients, but all were mild or moderate. Low white blood cell counts and infections did occur, reflecting both the drug’s strength and patients’ fragile immune systems, yet serious infections were less frequent than reported with several similar drugs.
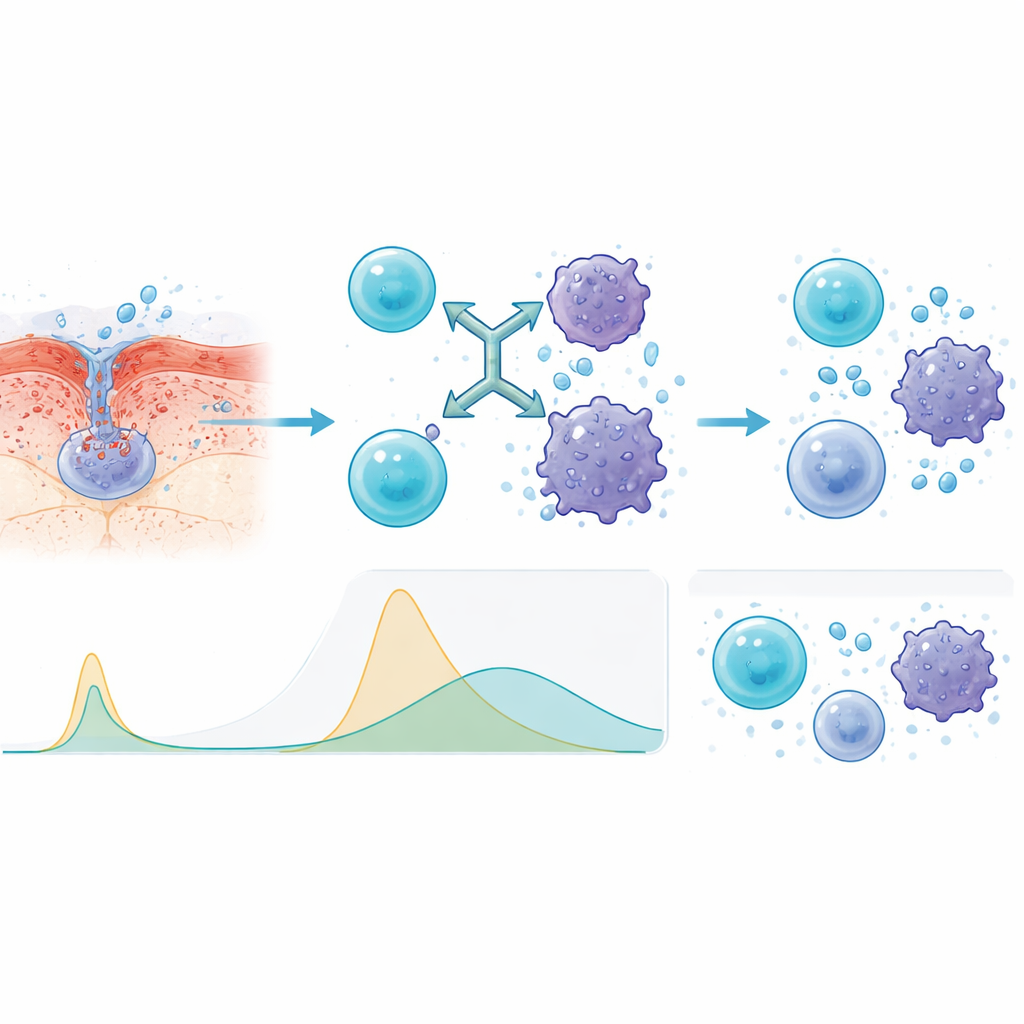
Why the injection route was safer
To understand why the under‑the‑skin method behaved better, the researchers measured immune signaling molecules in the blood. Compared with infusion into a vein, injections produced slower, lower spikes in key inflammatory signals such as interleukins and tumor necrosis factor. This flatter profile means the immune system is still activated against the cancer, but without the same sudden surge that can make patients dangerously ill. The team also found that people whose blood showed lower levels of shed BCMA at the start—a sign of less aggressive or less advanced disease—tended to respond better and stay in remission longer.
What this means for future myeloma care
Although the company has since decided not to continue developing alnuctamab for strategic reasons, the lessons from this trial are important. The study shows that a carefully designed immune engager can be given as a simple shot, on a schedule that becomes less demanding over time, while still delivering deep and lasting responses in many people with hard‑to‑treat myeloma. It also suggests that gradually ramping up the dose and avoiding sharp peaks in immune activation can limit serious reactions without dulling anti‑cancer power. These insights are likely to guide the design of future immune‑based treatments, offering hope that more patients with relapsed myeloma will have effective therapies that are not only powerful, but also safer and easier to live with.
Citation: Bar, N., Martin, T., Hofmeister, C.C. et al. Alnuctamab, a bivalent B-cell maturation antigen-targeting T cell engager for patients with relapsed or refractory multiple myeloma: results from a phase 1, first-in-human study. Leukemia 40, 481–490 (2026). https://doi.org/10.1038/s41375-025-02841-x
Keywords: multiple myeloma, immunotherapy, bispecific antibody, BCMA, T cell engager