Clear Sky Science · en
Soluble Notch agonist enables human ameloblast maturation and enamel-like tissue formation for tooth regeneration
Why fixing tooth enamel is so hard
Enamel, the glassy white shell that coats our teeth, is the hardest material in the human body—but once it’s gone, it’s gone. More than 90 percent of adults have some enamel loss or damage, yet our teeth have no natural way to rebuild this protective armor. This study describes a way to grow human enamel‑producing cells in the lab, push them to a fully mature state, and even coax them to form enamel‑like mineral in animals. The work opens a path toward future therapies that might one day repair teeth with living tissue instead of fillings and crowns.
Building tooth enamel from stem cells
Enamel is made by specialized cells called ameloblasts, which die off once a tooth erupts through the gums. Without them, adult teeth cannot make new enamel. The researchers use human induced pluripotent stem cells—adult cells reprogrammed back into a flexible, embryo‑like state—to create “induced ameloblasts.” In earlier work, these lab‑grown cells could start to resemble natural enamel‑making cells but stalled before reaching full maturity, and they only functioned properly when grown in close contact with odontoblasts, the cells that form the underlying dentin. That raised a key question: what exactly do odontoblasts provide that ameloblasts need in order to finish their development?
The missing conversation between tooth cells
To decode this cellular dialog, the team mined single‑cell gene expression maps from developing human and mouse teeth. They looked for signaling pathways that seemed to run specifically from odontoblasts to ameloblasts during the key window when enamel forms. One pathway stood out: Notch, a direct cell‑to‑cell communication system used widely in embryonic development. In these tooth maps, Delta‑like ligands (especially DLL1 and DLL4) were mainly found on odontoblasts, while Notch receptors were concentrated on maturing ameloblasts. When the researchers blocked Notch activity with a drug in their co‑culture system, the ameloblasts sharply reduced production of enamelin, a major enamel protein—strong evidence that Notch signaling is essential for enamel‑forming cells to mature. 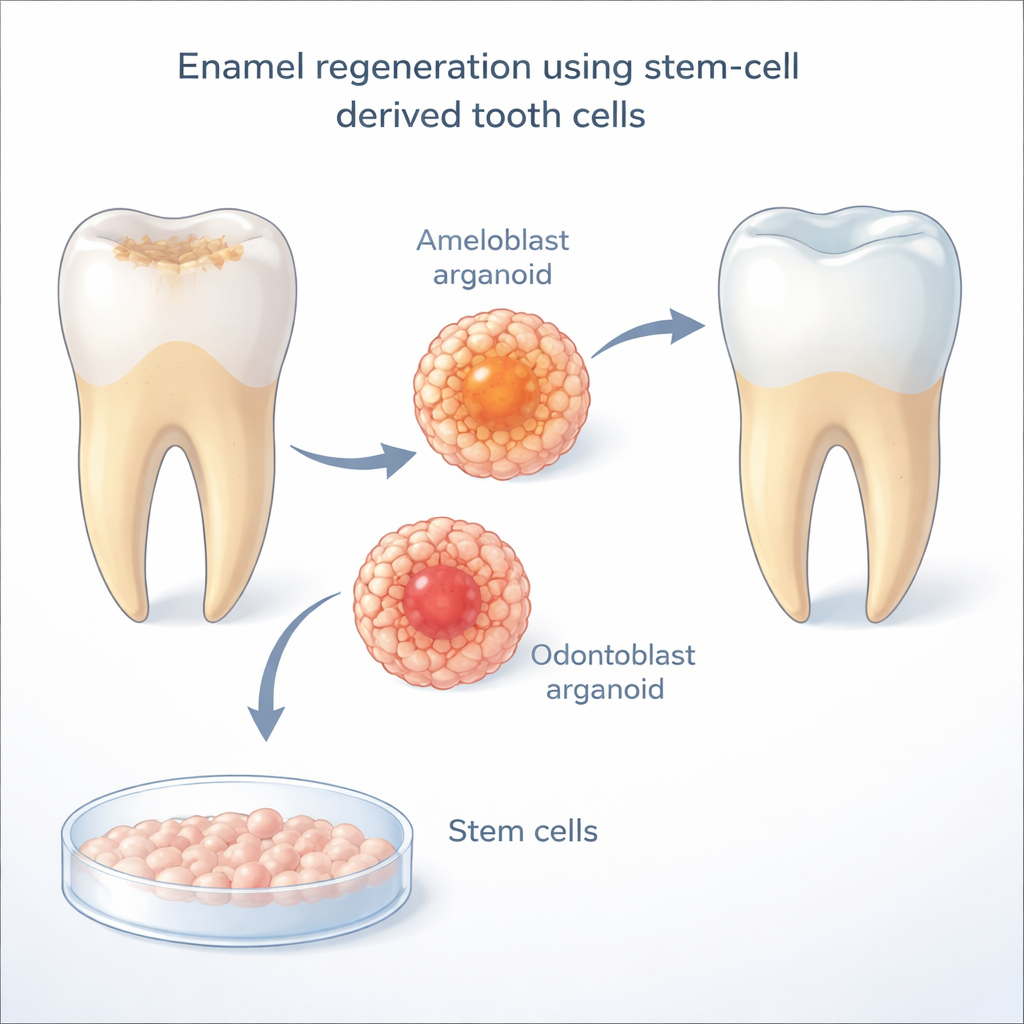
An AI‑designed protein switch for enamel cells
To go further, the team wanted to turn Notch on in a precise and controllable way, without relying on real odontoblasts. They used an artificial protein called C3‑DLL4, designed with computational tools. This soluble molecule arranges three copies of the DLL4 signaling domain in a rigid, three‑armed scaffold, allowing it to cluster and activate Notch receptors on nearby cells much like a natural neighboring cell would. In reporter cell lines, C3‑DLL4 strongly switched on Notch‑dependent genes. When added to early ameloblast organoids—tiny 3D cell clusters grown from stem cells—it drove a sweeping shift in gene activity. The organoids moved from an immature profile toward one that matched secretory and fully mature ameloblasts, turning on hallmark genes such as AMELX, ENAM, MMP20, ODAM, KLK4, TUFT1, and WDR72. Removing Notch activity had the opposite effect, stalling maturation and disrupting enamel protein production.
From organoids to enamel‑like tissue
Strikingly, when these Notch‑activated ameloblast organoids were transplanted under the kidney capsule of immune‑deficient mice—a safe, well‑supplied niche often used to test human tissues—they began to deposit dense, mineralized material. High‑resolution micro‑CT scans and classic bone‑staining techniques confirmed the presence of calcified nodules where no such tissue normally exists. Microscopy showed organized, polarized layers of human cells expressing key enamel proteins and WDR72, a factor linked to proper enamel mineralization. Although this material is best described as “enamel‑like” rather than fully natural enamel, it demonstrates that engineered human ameloblasts can, in principle, lay down hard mineral in a living body. 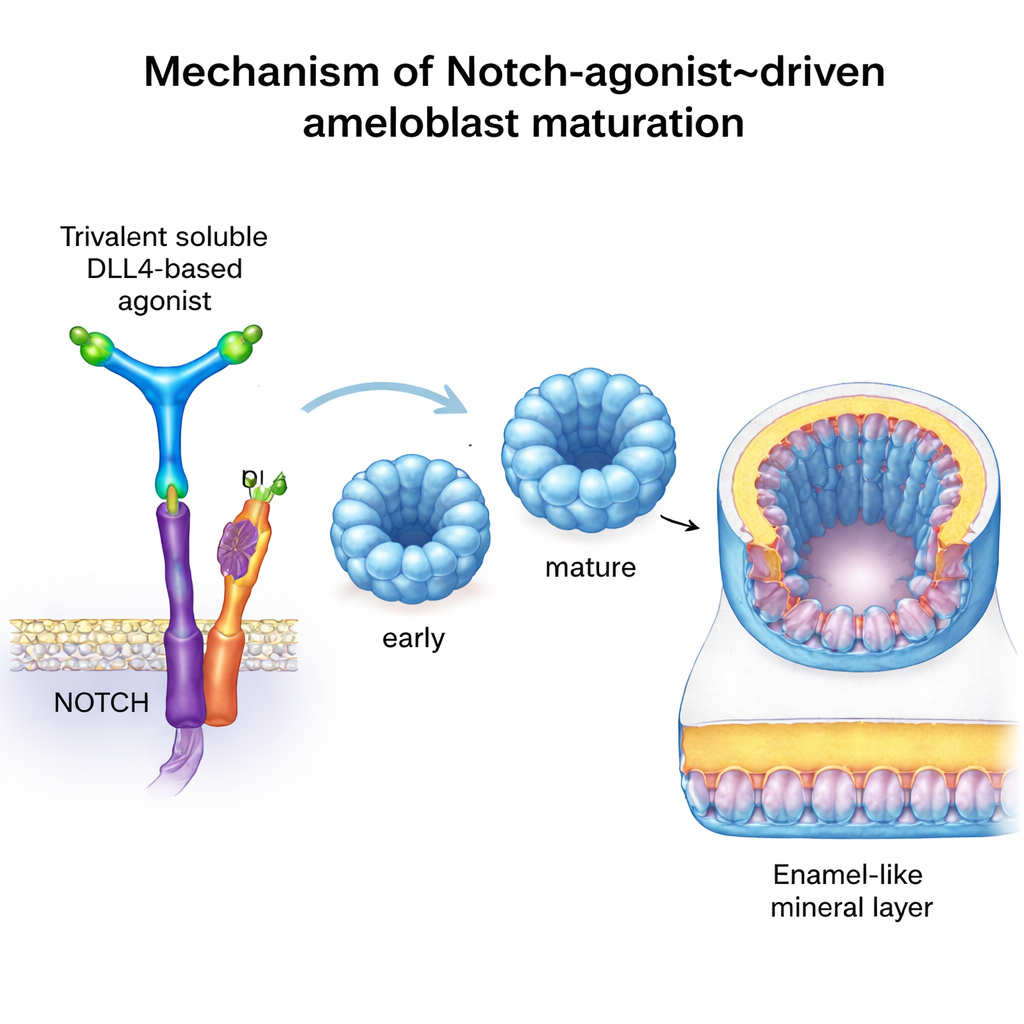
Cracking a genetic mystery behind weak enamel
The study also tackles a long‑standing puzzle around a gene called DLX3, mutations in which cause certain inherited enamel defects and raise cavity risk. DLX3 is active in both ameloblasts and odontoblasts, making it hard to tell which cell type is most affected when things go wrong. By using their odontoblast‑free organoid system and CRISPR gene editing, the researchers removed DLX3 specifically in the ameloblast lineage. Early steps of ameloblast formation still occurred, but the final maturation phase collapsed: key enamel proteins such as enamelin, AMELX, MMP20, KLK4, TUFT1, and WDR72 were drastically reduced at both RNA and protein levels. This shows that DLX3 is directly required inside human ameloblasts to complete the enamel‑making program, helping to explain why variants near this gene are tied to fragile, cavity‑prone teeth.
What this could mean for future dental care
Together, these findings outline a step‑by‑step roadmap from stem cells to enamel‑producing organoids to enamel‑like tissue in vivo, controlled by a tunable Notch “on switch” and the DLX3 gene. While clinical tooth regeneration is still far off, this work provides a powerful testing ground for understanding genetic dental diseases, screening new treatments, and refining strategies to rebuild lost enamel with living cells instead of inert materials. For patients, the long‑term hope is that visits to the dentist might one day involve repairing worn or decayed teeth with bioengineered enamel that behaves much more like the real thing.
Citation: Patni, A.P., Mout, R., Alghadeer, A. et al. Soluble Notch agonist enables human ameloblast maturation and enamel-like tissue formation for tooth regeneration. Int J Oral Sci 18, 25 (2026). https://doi.org/10.1038/s41368-026-00429-4
Keywords: enamel regeneration, ameloblast organoids, Notch signaling, DLX3, dental stem cells