Clear Sky Science · en
ANKRD1 sustains a neurogenic BMSC niche and counters cognitive aging
Why the Jaw Might Matter for an Aging Mind
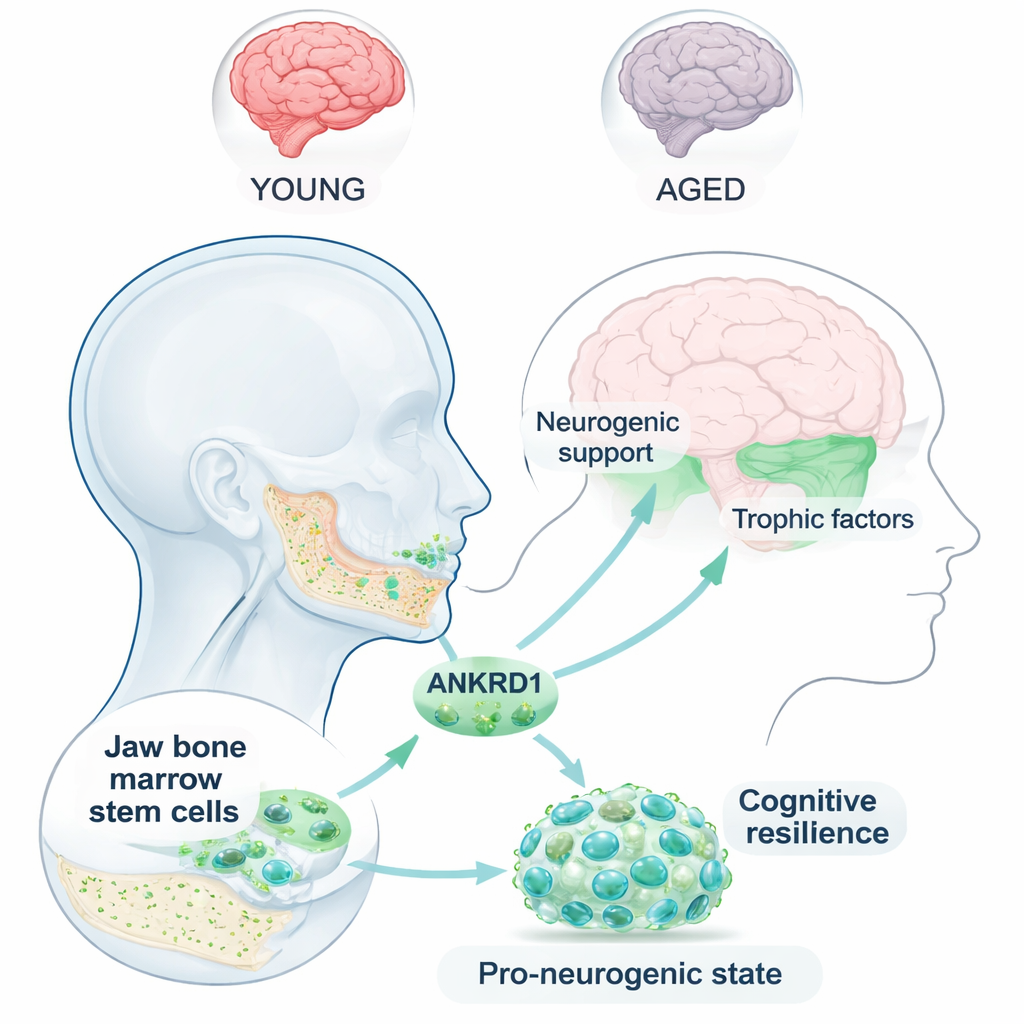
Most people think of the jaw bone as important for chewing and dentistry, not for memory. This study suggests a surprising link: stem cells hidden inside the jaw’s bone marrow may help protect the brain from age-related memory loss. The researchers focused on a protein called ANKRD1, showing that it helps these jaw-derived stem cells keep their “neural” potential and, in turn, supports brain health and spatial memory in aging animals.
A Hidden Nerve-Friendly Niche in Jaw Bone
Jaw bone marrow contains mesenchymal stromal cells—versatile stem cells best known for making bone and cartilage. Because the jaw develops from a structure called the neural crest, the authors wondered whether its stem cells might retain some nerve-forming ability. Using single-cell RNA sequencing, they mapped thousands of individual jaw stem cells and discovered a distinct subpopulation that looked like neural progenitors: highly proliferative, rich in genes linked to neural development, and poised to become nerve-like cells. Within this niche, ANKRD1 emerged as a standout marker, strongly associated with both growth and neurogenic potential.
ANKRD1 as a Guardian Against Aging and Premature Specialization
As we age, many stem cells lose their flexibility and slide toward exhaustion. The team showed that ANKRD1 levels drop sharply in jaw stem cells from older donors and in lab-grown cells pushed into a senescent, or aged, state. When ANKRD1 was experimentally reduced in young cells, classical aging markers rose, and the cells began behaving like old ones. Conversely, boosting ANKRD1 in aged cells dialed down these senescence signals. The protein also helped keep the cells from committing too quickly to bone or fat lineages: low ANKRD1 drove up bone and fat markers, while high ANKRD1 suppressed them. In simple terms, ANKRD1 helps jaw stem cells stay youthful and versatile instead of burning out or locking into a single fate.
Keeping Neural Genes Accessible at the DNA Level
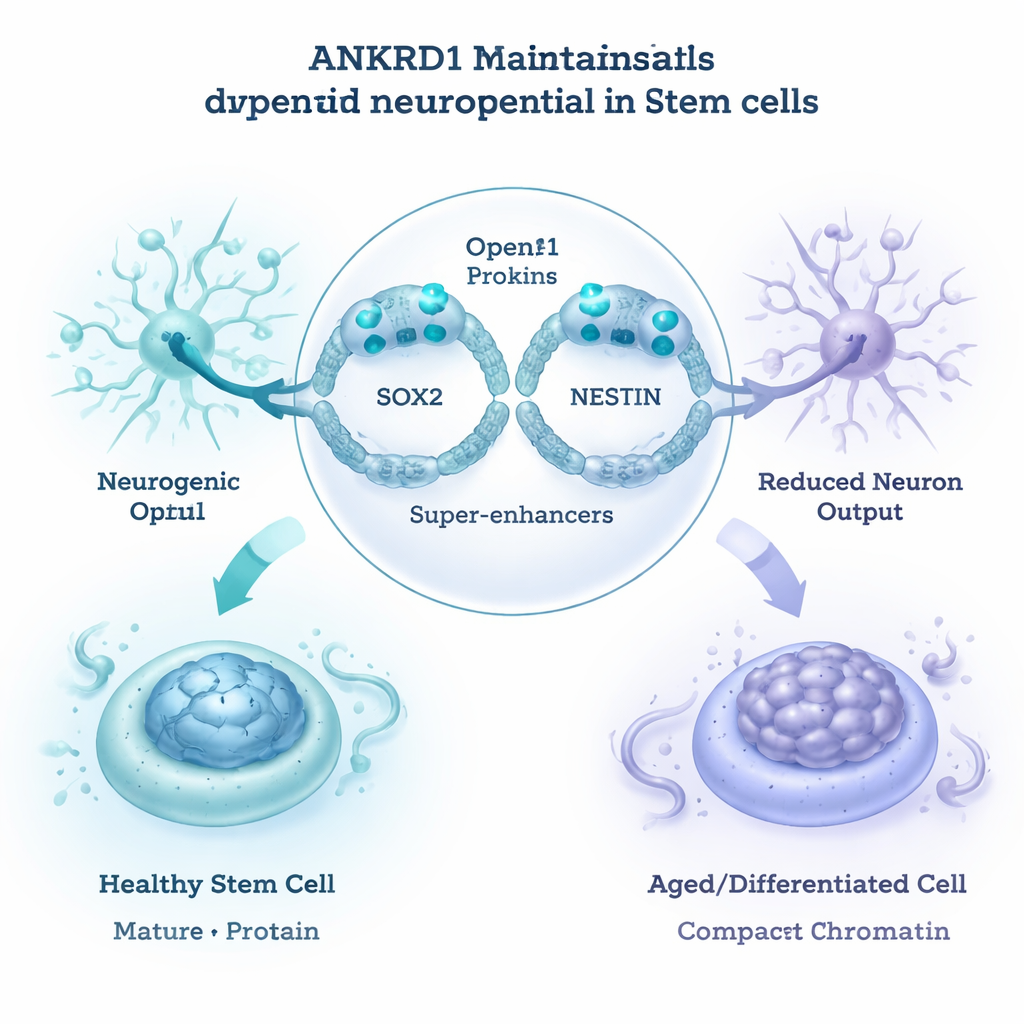
The authors then asked how ANKRD1 exerts such broad control. By profiling where ANKRD1 sits on the genome, they found that it binds strongly to powerful regulatory regions known as super-enhancers, especially those controlling two key neural stem cell genes, SOX2 and NESTIN. In young, undifferentiated cells, ANKRD1 occupancy at these sites coincided with open, accessible chromatin—the relaxed DNA architecture that allows genes to be turned on. As cells differentiated or aged, ANKRD1 binding at distant control regions fell, the chromatin became more compact, and neural pathways were less active. Reporter experiments confirmed that ANKRD1 can directly boost the activity of SOX2 and NESTIN control elements. Combined with 3D genome mapping, these results suggest that ANKRD1 maintains a “neurogenic reservoir” in jaw stem cells by holding key neural genes in a ready-to-use, open configuration.
From Jaw Stem Cells to Better Memory in Old Mice
If ANKRD1 keeps neural options open in stem cells, can it actually improve brain function? To test this, the researchers delivered ANKRD1 specifically to neurons in naturally aged mice using a viral vector designed to cross the blood–brain barrier. The treatment did not change movement or anxiety-like behavior, but it significantly improved performance in the Morris water maze, a classic test of spatial learning and memory. Treated mice learned the platform location faster and searched more precisely in the correct quadrant. In parallel lab experiments, raising ANKRD1 levels made jaw stem cells express more neural markers, adopt neuron-like shapes, and secrete higher amounts of brain-supporting growth factors such as NGF and BDNF, suggesting a two-pronged benefit: better intrinsic neurogenic capacity and a richer supportive environment.
Rewiring Brain Activity Patterns in Key Memory Regions
To see how ANKRD1 reshapes brain function, the team mapped activity of neurons across the whole mouse brain using c-Fos, a protein that lights up in recently active cells. ANKRD1-treated mice showed stronger activation in regions crucial for cognition, including the hippocampal formation, hypothalamus, and the outer brain cortex. These areas not only became more active but also more synchronized with each other, shifting from fragmented patterns toward more integrated, positively correlated networks. This coordinated activation pattern is thought to support efficient information processing and may underlie the improved memory seen in behavior tests.
What This Means for Future Brain Health
Altogether, the study paints ANKRD1 as a key molecular switch that preserves nerve-forming capacity in jaw bone marrow stem cells and helps stabilize brain networks during aging. By keeping crucial neural genes accessible and encouraging both neuron production and protective factor release, ANKRD1 supports resilience of memory circuits. While translation to humans will require caution, the work opens the door to therapies that tap into the unique developmental origins of jaw stem cells and their chromatin “memory” to counter cognitive aging.
Citation: Wang, Z., Liu, X., Zhen, W. et al. ANKRD1 sustains a neurogenic BMSC niche and counters cognitive aging. Int J Oral Sci 18, 23 (2026). https://doi.org/10.1038/s41368-026-00428-5
Keywords: cognitive aging, jaw bone marrow stem cells, ANKRD1, neurogenesis, spatial memory