Clear Sky Science · en
Gli2 and Gli3 synergistically mediate HH-TGF-β crosstalk in mesenchymal progenitor cells to orchestrate tooth root morphogenesis
Why Tooth Roots Matter More Than You Think
Most of us only think about our teeth when they hurt or when we see them in the mirror. But what anchors each tooth in the jaw—the root—is just as important as the visible crown. Healthy roots keep teeth stable for chewing and speaking throughout life. This study explores how tiny signals inside jaw tissues tell immature cells how to build strong tooth roots, work that could one day help prevent or repair root defects that lead to loose teeth and tooth loss.
The Traveling Cells That Build Our Faces
Early in development, a special population of cells called cranial neural crest cells migrates into the forming face and jaws, where they transform into bone, cartilage, and tooth-supporting tissues. When this transformation goes wrong, babies can be born with serious facial and dental problems. A key signal that guides these cells is the Hedgehog pathway, which acts like a molecular GPS, helping cells decide where to go and what to become. The authors focused on two Hedgehog-controlled helper proteins, Gli2 and Gli3, to see how they jointly direct the cells that eventually build the roots of molar teeth in mice.
Two Gene Switches Working as a Team
Using finely engineered mouse models, the researchers selectively turned off Gli2, Gli3, or both in a group of root-forming progenitor cells located near the growing tips of the roots. Removing Gli2 alone barely changed root formation, but removing Gli3 produced noticeably shorter roots, delayed tooth eruption, and less supporting jawbone around the teeth. When both Gli2 and Gli3 were removed together, the problems became much more severe: roots were about half their normal length, tooth eruption was delayed, and the bone that normally cradles the roots was greatly reduced. These experiments show that Gli2 and Gli3 do not act in isolation; instead, they cooperate to shape the root and its surrounding support structures.
Guiding Cells Into the Right Jobs
To understand why roots failed, the team looked closely at what root progenitor cells normally become. In healthy teeth, these cells give rise to dentin-producing odontoblasts inside the root, fibrous ligament cells that tie the root to bone, and bone-forming cells in the socket. When Gli3, and especially both Gli2 and Gli3, were missing, these lineages were disrupted: key markers of dentin and ligament identity were greatly reduced, and fewer bone-forming cells appeared near the roots. Tracking the descendants of the progenitor cells showed that instead of populating the root and its ligament, many of these cells stayed higher up in the tooth area, indicating that their normal “career choices” had been derailed. At the same time, the number of dividing cells in the root region dropped, so there were fewer building blocks available to construct the root.
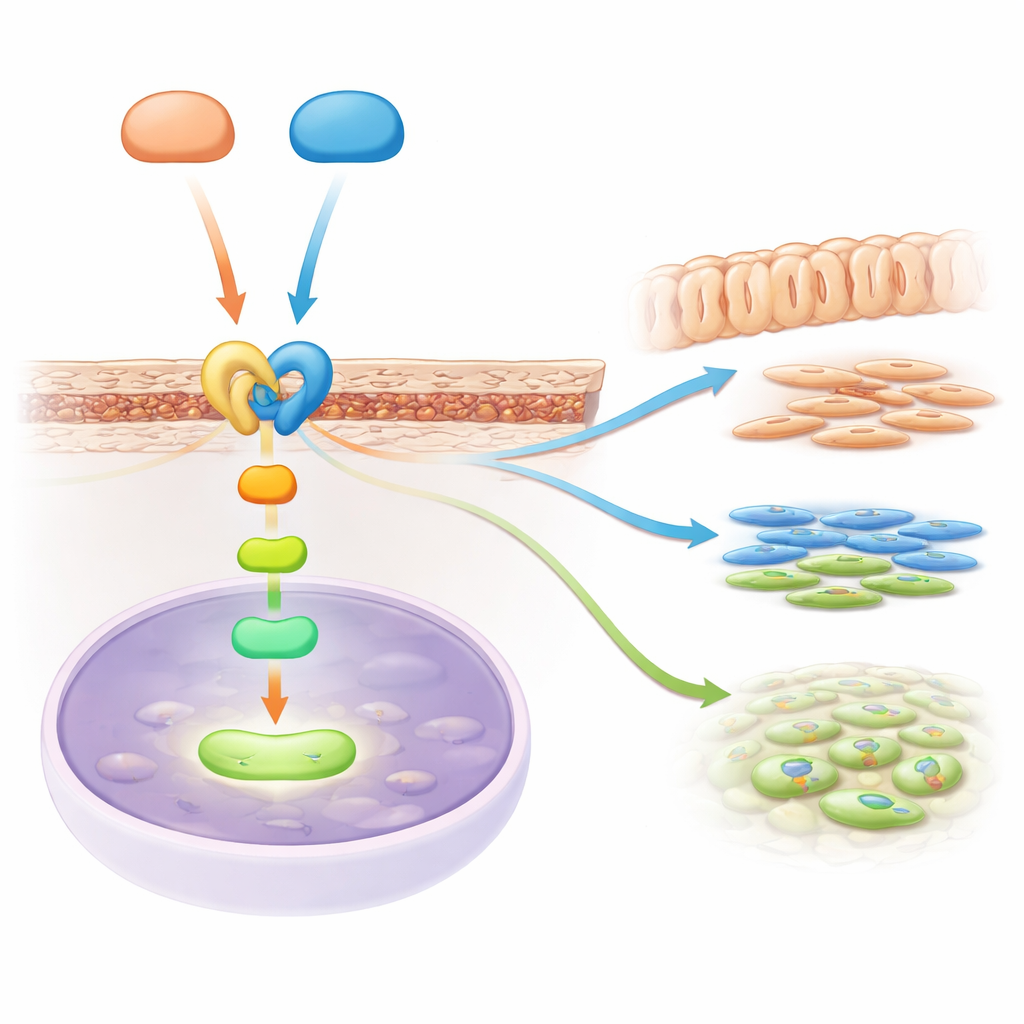
A Signal Relay Linking Two Major Pathways
The story did not end with Hedgehog signaling. By reading out which genes were turned on or off, the scientists discovered that another major pathway, TGF-beta signaling, was dampened when Gli2 and Gli3 were lost. TGF-beta is well known for promoting the formation of dentin and bone. The team pinpointed a key relay molecule, a receptor called Acvr2b, which sits at the surface of root progenitor cells and helps pass along TGF-beta family signals into the cell. They showed that both Gli2 and Gli3 physically bind to the control region of the Acvr2b gene, boosting its activity. Without these helpers, Acvr2b levels in the root region dropped, and the downstream TGF-beta messengers inside cells were less active, weakening the instructions that drive root growth and differentiation.
Partially Rewiring a Faulty Root-Building Program
To test whether reviving TGF-beta signaling could help, the researchers treated mice lacking both Gli2 and Gli3 with a drug that stimulates this pathway. While it could not fully restore normal roots, the treatment did lengthen roots, increased the amount of surrounding bone, and improved the formation of dentin and ligament tissue compared with untreated mutant mice. This partial rescue suggests that the Gli2/Gli3–Acvr2b–TGF-beta chain is a major route by which Hedgehog signals are converted into the structural growth of tooth roots.
What This Means for Future Dental Care
In simple terms, this work shows that two genetic “switches,” Gli2 and Gli3, work together to help young jaw cells sense Hedgehog signals and, through a relay involving TGF-beta, decide to become the specialized cells that build and support tooth roots. When this communication breaks down, roots are short, teeth erupt poorly, and supporting bone is thin. Understanding this signal network not only clarifies how normal tooth roots form, but also points toward new strategies to guide stem or progenitor cells in repairing damaged roots or regenerating them altogether in future dental therapies.
Citation: Zhou, T., Huang, L., Xie, Y. et al. Gli2 and Gli3 synergistically mediate HH-TGF-β crosstalk in mesenchymal progenitor cells to orchestrate tooth root morphogenesis. Int J Oral Sci 18, 30 (2026). https://doi.org/10.1038/s41368-026-00427-6
Keywords: tooth root development, cranial neural crest cells, Hedgehog signaling, TGF-beta pathway, dental stem cells