Clear Sky Science · en
Single-cell transcriptional atlas reveals distinct immune-chondrocyte crosstalk mechanisms in temporomandibular joint osteoarthritis induced by different types of occlusal disorder
Why your jaw joint matters
The small joints in front of your ears, called temporomandibular joints (TMJs), let you talk, chew, yawn and laugh. When they develop osteoarthritis, everyday actions can become painful and difficult. This study uses cutting‑edge single‑cell gene analysis in mice to look inside these jaw joints cell by cell, revealing how different bite problems trigger different kinds of damage—and how that might shape more precise treatments.
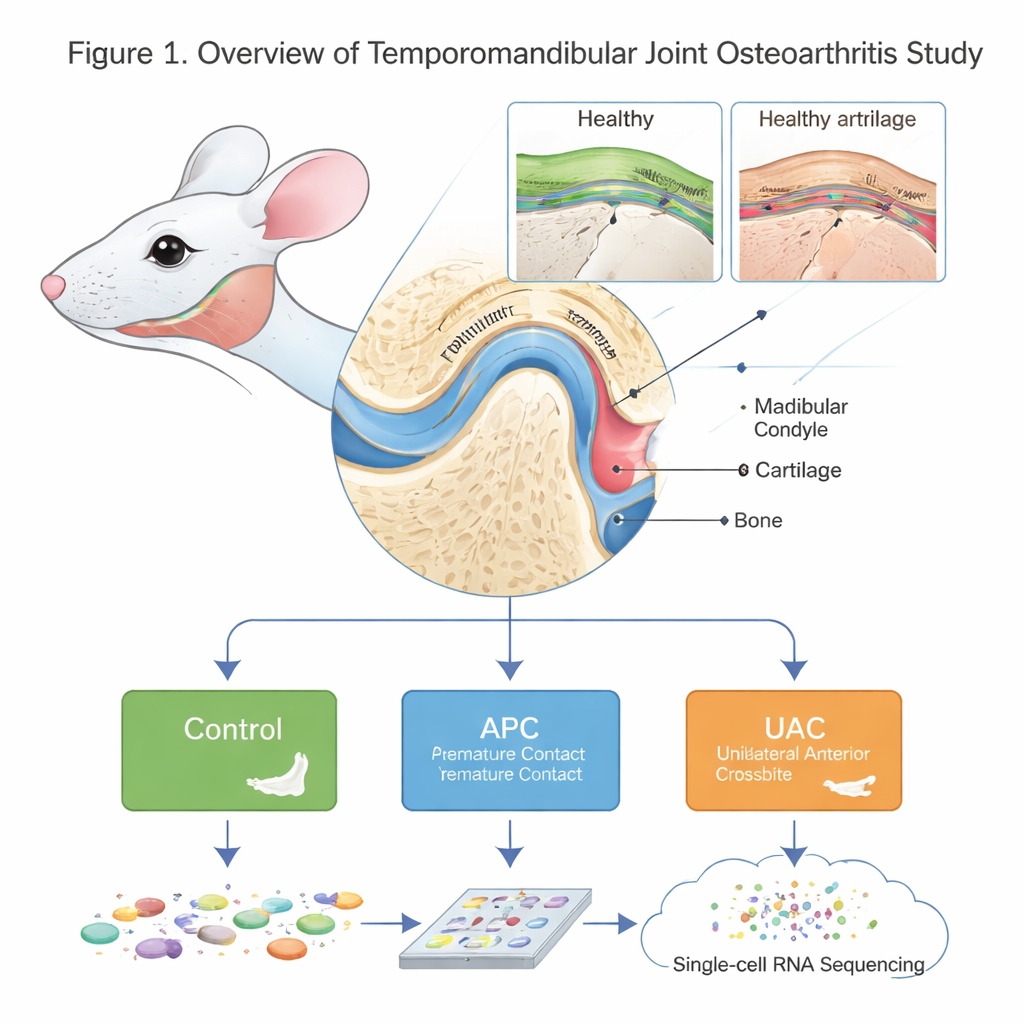
Looking closely at a tiny but complex joint
The TMJ is not a simple hinge. Its rounded end, the mandibular condyle, is covered by a special type of cartilage and supported by underlying bone. Osteoarthritis in this joint (TMJOA) causes pain, stiffness and loss of function, but its microscopic and molecular causes have been hard to untangle. The authors created a detailed “cell atlas” of the mouse condyle in health and disease using single‑cell RNA sequencing, a technique that reads which genes are active in thousands of individual cells. They compared healthy mice with two models of bite imbalance—anterior premature contact (APC), where the front teeth meet too early, and unilateral anterior crossbite (UAC), where one side of the front teeth bites in the wrong way—both known to overload the jaw joint.
Many kinds of cartilage cells, not just one
Traditionally, condylar cartilage has been described in broad layers—proliferating, mature and hypertrophic cells. This study shows the real picture is far more varied. The team identified multiple chondrocyte (cartilage cell) subtypes, each with its own gene activity and likely role. There were progenitor cells near the surface that behave like a repair pool, fibrotic chondrocytes involved in scar‑like matrix and healing, inflammation‑associated chondrocytes producing inflammatory signals, mineralization‑associated and hypertrophic chondrocytes that help turn cartilage into bone, and reparative cells that dampen stress and inflammation. In healthy joints, these subtypes sit in an orderly spatial pattern and follow a smooth developmental “trajectory” from progenitor to more mature states.
When bite problems scramble the joint’s cellular order
Both APC and UAC models led to thinner cartilage, surface roughening and typical osteoarthritis‑like lesions in the condyle. At the cellular level, the proportion of chondrocytes fell while immune cells rose. In APC, there was a surge of certain mineralizing and hypertrophic chondrocytes and a disruption of the normal progression from progenitor to mature cells. In UAC, there was also an increase in fibrotic and reparative chondrocytes, hinting at a stronger repair and remodeling attempt. Microscopy confirmed that cells lost their orderly layers and became compacted and disorganized in diseased cartilage, matching the disturbed developmental paths inferred from the gene‑expression data.
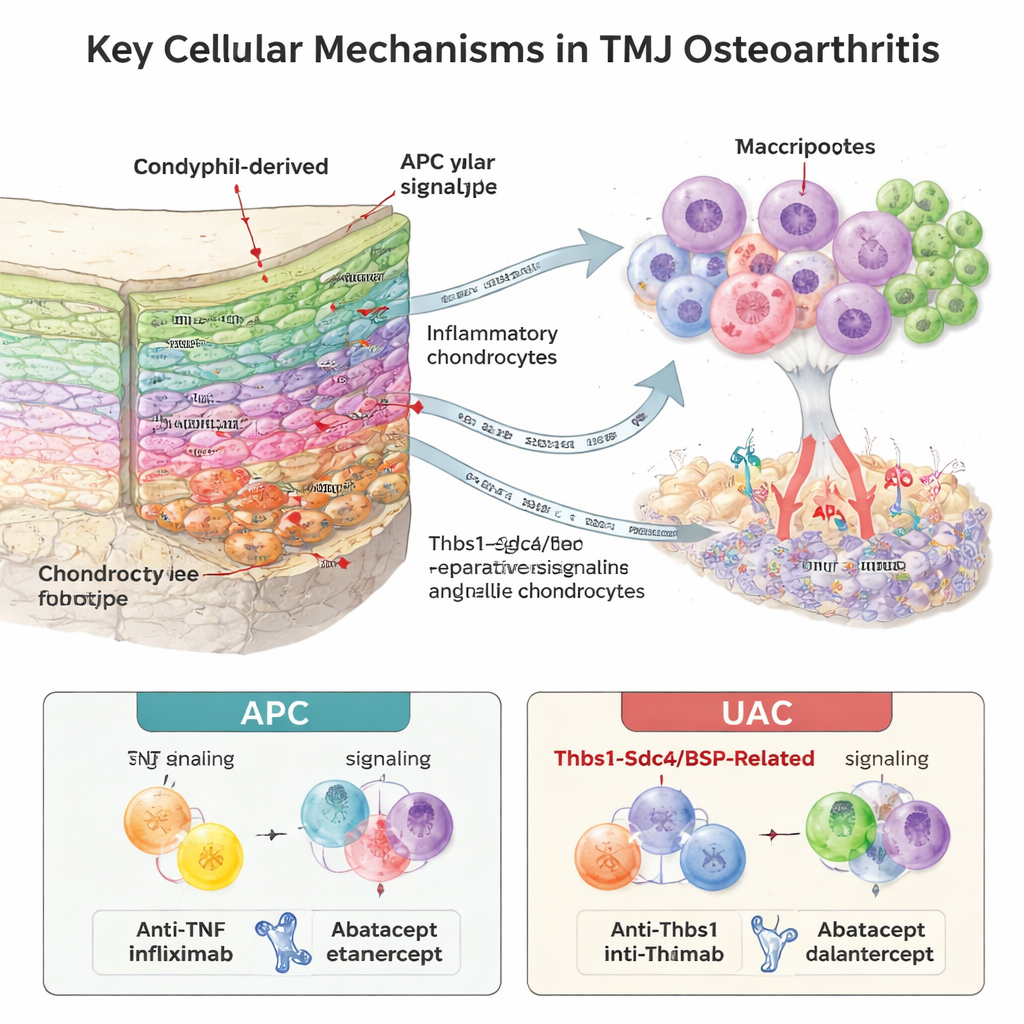
Immune cells, blood vessels and cartilage talk to each other
The condyle’s supporting bone and marrow harbored a surprisingly rich immune “microenvironment.” The researchers found nine immune cell types, with neutrophils (a fast‑acting white blood cell) expanding strongly in diseased joints, especially a specific maturation stage. By analyzing which signaling molecules and receptors were co‑expressed, they inferred intense “crosstalk” between immune cells and chondrocytes. In the APC model, neutrophils became the main source of TNF‑alpha, a potent inflammatory signal, sending strong damaging messages to cartilage cells. In the UAC model, a different pattern emerged: bone‑ and cartilage‑linked signals involving bone sialoprotein (BSP) and the Thbs1–Sdc4 pathway were more prominent, and these were closely tied to new blood vessel growth at the cartilage–bone border. Areas rich in BSP overlapped with markers of angiogenesis, suggesting that invading vessels and recruited immune cells jointly drive cartilage erosion.
Tailored treatments for different joint stresses
To test whether these distinct signals really matter, the team treated mice during disease development with drugs that block specific pathways. In APC‑induced TMJOA, blocking TNF‑alpha with the drug etanercept clearly protected the cartilage, while a drug that targets neutrophil‑attracting receptors (Navarixin) made little difference. In contrast, in UAC‑induced TMJOA, Navarixin significantly eased cartilage damage, consistent with a disease process more dependent on neutrophil‑driven vessel invasion and remodeling, while etanercept had a weaker effect. These findings suggest that TMJ osteoarthritis is not a single disease but a collection of related conditions shaped by how the joint is stressed, and that effective therapies may need to be matched to the specific mechanical and cellular patterns at work in each patient.
What this means for people with jaw pain
For someone struggling with jaw clicking, pain or limited opening, this research underscores two key ideas. First, the jaw joint is maintained by a diverse community of cartilage and immune cells that can either protect or harm the tissue depending on how they are activated. Second, different bite problems may push this community toward distinct harmful pathways—one more driven by classic inflammation, another by abnormal blood vessel growth and bone‑cartilage remodeling. In the long run, this kind of single‑cell mapping may help dentists and doctors move beyond one‑size‑fits‑all treatments toward approaches that combine bite correction with targeted drugs chosen for the patient’s specific pattern of joint damage.
Citation: Wang, K., Li, Y., Xiang, S. et al. Single-cell transcriptional atlas reveals distinct immune-chondrocyte crosstalk mechanisms in temporomandibular joint osteoarthritis induced by different types of occlusal disorder. Int J Oral Sci 18, 22 (2026). https://doi.org/10.1038/s41368-025-00424-1
Keywords: temporomandibular joint osteoarthritis, single-cell RNA sequencing, chondrocytes, neutrophils, occlusal disorder