Clear Sky Science · en
Implantation awakens peri-implant osteogenic potential via Snx5-EGFR axis-mediated mechanical transduction
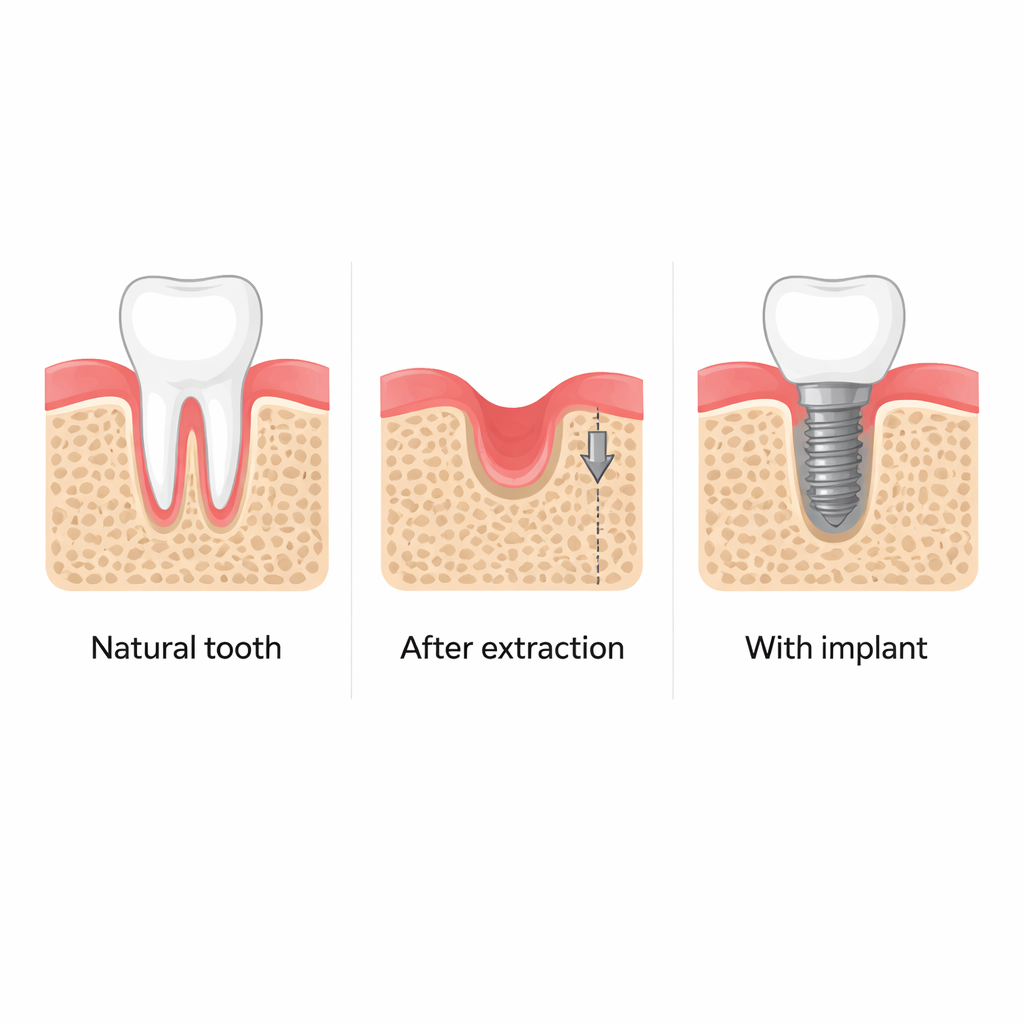
Why keeping jawbone after tooth loss matters
When a tooth is pulled, the surrounding jawbone often shrinks away, making it harder to place stable dental implants later and affecting facial appearance. Dentists have long noticed that simply putting an implant into a fresh socket seems to help preserve this bone, even before the implant is used for chewing. This study asks a simple but important question: how does the act of implant placement itself “wake up” the bone’s repair system, and can we tap into that process to improve jawbone regeneration?
Implants as quiet bone protectors
Clinical scans from patients and detailed experiments in mice confirm that placing a dental implant soon after extraction helps maintain the height and thickness of the surrounding jawbone. In contrast, sockets left to heal on their own lose substantial bone over several months. Around implants, the researchers found more immature bone-forming cells (osteoprogenitors), indicating that the tissue next to the metal post is biologically more active. This enhanced activity appears to be driven not by chewing forces, but by the unusual mechanical stresses created as a snug implant is screwed into slightly undersized bone.
A hidden switch inside bone stem cells
The team focused on a special group of bone marrow stem cells marked by a protein called LepR, known to be key suppliers of new bone in adults. By comparing gene activity in these cells from normal bone, implant-adjacent bone, and cells stressed in the lab, they homed in on one molecule, sorting nexin 5 (Snx5), whose levels consistently dropped when mechanical cues were present. In regions around implants, LepR cells with low Snx5 were more likely to carry markers of early bone-building cells, suggesting that dialing down Snx5 helps push these quiet stem cells toward an active bone-forming state.
When the switch is broken, bone can’t sense force properly
To test what Snx5 actually does, the researchers used mice and stem cells lacking this protein. Without Snx5, the stem cells showed a strong built‑in tendency to make bone, even without extra stimulation. But when the scientists applied mechanical stretch—mimicking the strains near an implant—normal cells ramped up their bone-forming activity, whereas Snx5‑deficient cells hardly responded. In living mice, implants in normal animals triggered clear new bone growth around the titanium post. In contrast, mice without Snx5 failed to gain extra bone after implantation, revealing that Snx5 is crucial for converting mechanical cues into additional bone repair rather than for baseline bone formation itself.
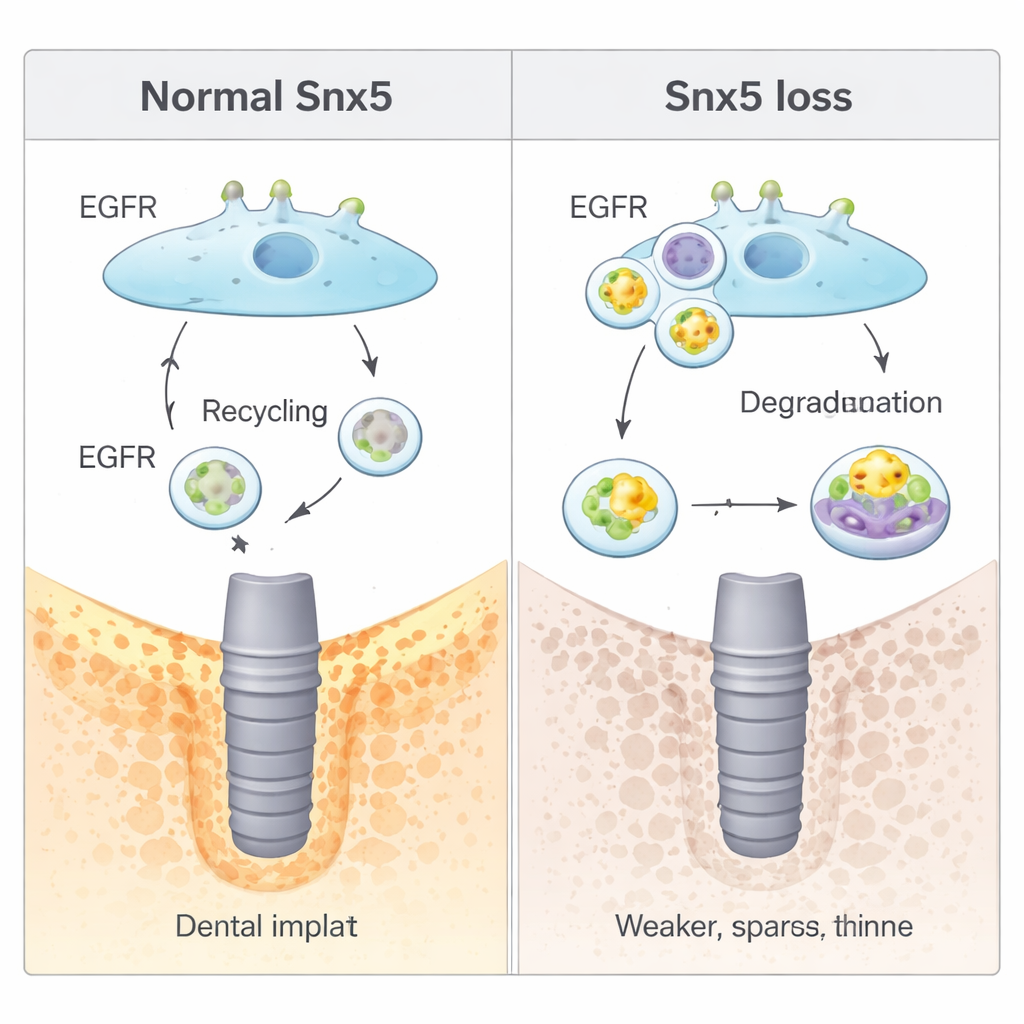
How a recycling pathway controls bone signals
The study then uncovered how Snx5 works at the molecular level. Many growth signals in cells travel through receptors on the cell surface that are constantly pulled inside and either recycled or destroyed. Snx5 belongs to a family of “sorting” proteins that guide this traffic. Here, Snx5 was shown to steer the epidermal growth factor receptor (EGFR)—a key controller of cell growth and bone differentiation—back to the cell membrane after it is internalized. When Snx5 was missing, more EGFR was shunted into cellular “digestive” compartments (lysosomes) and broken down. This reduced EGFR signaling unexpectedly boosted baseline bone formation but left the cells less able to adjust their behavior when mechanical forces changed. Drugs that artificially activated EGFR dampened the extra bone formation seen without Snx5, while drugs that blocked lysosomal degradation restored EGFR signals and again reduced bone-building.
What this means for future implant patients
To a layperson, these findings suggest that the jawbone contains a built‑in system that can sense the subtle push and pull created when an implant is placed and translate that into extra bone growth. Snx5 acts like a traffic controller that keeps a key growth receptor, EGFR, cycling at just the right level so bone stem cells can “hear” and respond to those mechanical whispers. When this controller is altered, bone may form more easily at rest but loses its fine‑tuned response to force. Understanding this Snx5–EGFR axis opens the door to new treatments—drugs or surface coatings on implants—that could adjust the bone’s mechanical sensitivity, helping preserve jawbone and improve implant success in challenging clinical situations.
Citation: Jiang, X., Weng, Y., Feng, Y. et al. Implantation awakens peri-implant osteogenic potential via Snx5-EGFR axis-mediated mechanical transduction. Int J Oral Sci 18, 18 (2026). https://doi.org/10.1038/s41368-025-00423-2
Keywords: dental implants, jawbone regeneration, mechanotransduction, bone stem cells, EGFR signaling