Clear Sky Science · en
KDM6B/Pdk1 glycolytic pathway-driven ZEB2 lactylation promotes cellular cementum formation
Why protecting tooth roots matters
Gum disease, or periodontitis, is one of the leading causes of tooth loss in adults. As the disease progresses, it quietly erodes the tissues that anchor teeth to the jaw. One of the most important of these is cementum, a thin, bone-like layer that coats the root surface and allows the tooth to “tie in” to the surrounding ligament and bone. Once cementum is lost, teeth loosen and may eventually fall out. This study asks a simple but powerful question: can we understand, in molecular detail, how cementum is built, so that one day we can help the body rebuild it and keep teeth in place for life?
From tooth-supporting cells to a healing target
Cementum is made by specialized cells called cementoblasts, which sit on the root surface and deposit new mineral. These cells behave a lot like bone-forming cells, but the signals that tell them when and how to build cementum have been poorly understood. The researchers focused on a molecule called KDM6B, an enzyme that edits chemical tags on DNA-packaging proteins and can switch genes on. By studying mouse teeth at different ages and cementoblasts grown in the lab, they found that KDM6B levels rise during the active phase of cementum formation and fall once growth slows. When they blocked KDM6B in cells or in mice, cementoblast activity dropped and the “cellular” part of cementum – the thicker, living portion near the root tip – became thinner, suggesting that KDM6B is a key promoter of healthy cementum growth.
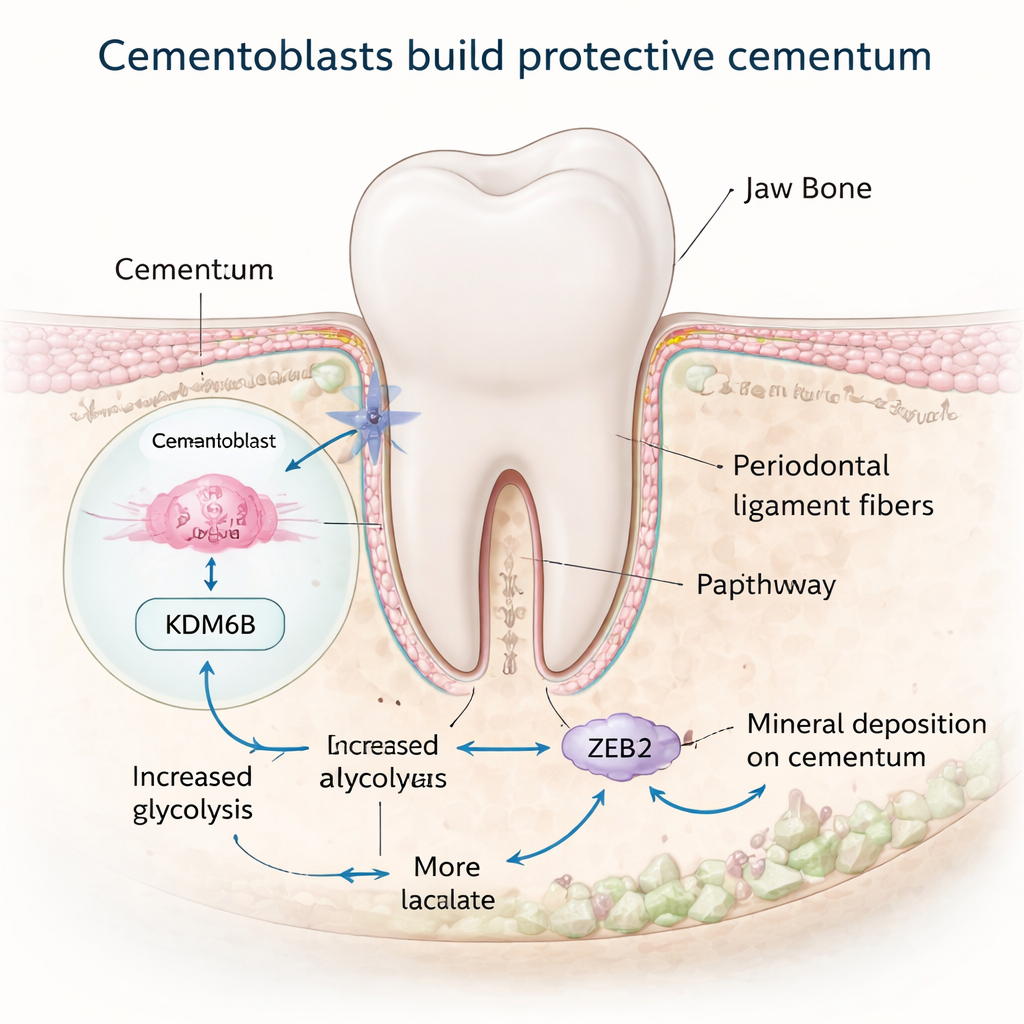
Rewiring cell metabolism to build mineral
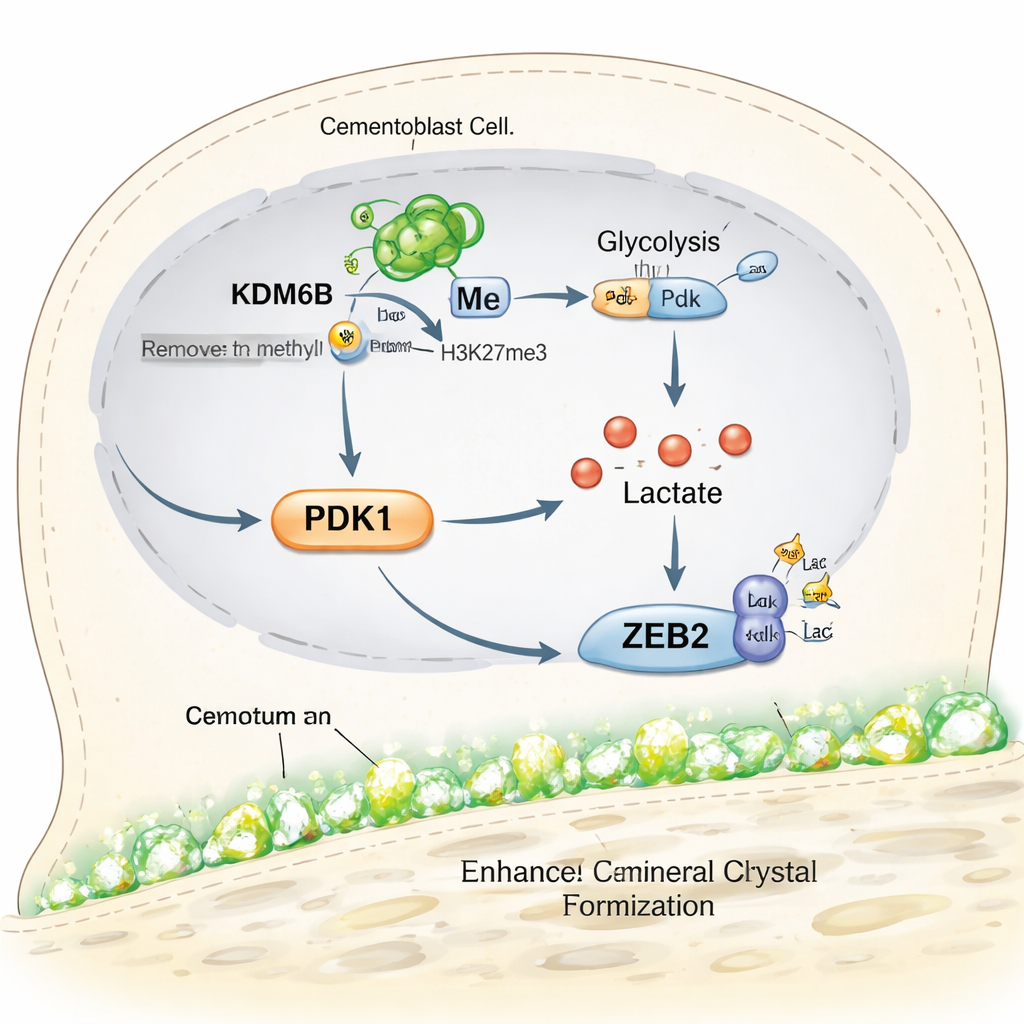
Building mineralized tissue is energy-intensive, and many cells switch their metabolism into a special high-gear state while doing it. The team discovered that KDM6B helps cementoblasts rewire how they use sugar. Normally, these cells favor a pathway called glycolysis, which rapidly breaks down glucose and produces lactate. When KDM6B was reduced, genes involved in glycolysis were turned down, while those linked to slower, oxygen-dependent energy production were mixed or increased, and the cells made less energy overall. A central player in this shift was an enzyme called PDK1. Using genome-mapping tools, the researchers showed that KDM6B physically removes a repressive tag from the DNA region controlling the Pdk1 gene, allowing PDK1 levels to rise. Restoring PDK1 in KDM6B-deficient cells brought back many markers of mineral formation, revealing a KDM6B–PDK1 metabolic switch that powers cementoblasts.
Turning lactate from waste into a useful signal
Lactate is often thought of as a waste product of hard-working muscles, but recent work has shown it can also act as a signal that modifies proteins. Here, higher glycolysis driven by KDM6B and PDK1 increased lactate production in cementoblasts. The researchers then tracked a newer type of protein modification called lactylation, in which lactate-derived tags are attached to specific protein sites. They found that lactylation levels in cementoblasts rose during periods of active cementum formation and dropped in older mice and in KDM6B-deficient cells. Adding simple sodium lactate to the system boosted lactylation, revived mineral-related genes, and restored mineral nodule formation both in cultured cells and in a mouse model, even when KDM6B was blocked. This suggests that lactate itself can help rescue weakened cementum-building activity.
A molecular boost for a key cementum regulator
The team then asked which proteins were being “decorated” by lactate in a way that mattered for cementum. They homed in on ZEB2, a gene control protein already known to promote cementoblast mineralization. Using protein-mapping techniques, they identified specific spots on ZEB2 that carry lactylation marks and showed that altering one critical site weakened the cell’s ability to turn on mineralization genes. Further experiments demonstrated that the KDM6B–PDK1 pathway boosts ZEB2 lactylation and that supplying extra lactate can restore ZEB2’s activity when PDK1 is low. In essence, metabolism and gene control are linked: sugar breakdown fuels lactate production, which then fine‑tunes ZEB2 to keep cementum-building programs running strongly.
What this means for future tooth-saving therapies
Put simply, the study reveals a chain of events inside cementum-forming cells: the KDM6B enzyme opens up the Pdk1 gene, PDK1 pushes cells toward fast sugar use, that metabolism generates lactate, and lactate chemically tunes the ZEB2 protein to drive mineral deposition. When any step in this chain is blocked, cementum growth falters, but carefully adding back lactate can partly restore it. For patients, this work does not yet translate into a clinical treatment, but it highlights new, precise targets for drugs or biomaterials designed to regenerate cementum and stabilize teeth in advanced gum disease. It also suggests that similar metabolic “switches” could be harnessed to encourage bone healing elsewhere in the body.
Citation: Yang, Z., Wang, H., Xiao, J. et al. KDM6B/Pdk1 glycolytic pathway-driven ZEB2 lactylation promotes cellular cementum formation. Int J Oral Sci 18, 21 (2026). https://doi.org/10.1038/s41368-025-00420-5
Keywords: cementum regeneration, periodontitis, cementoblast metabolism, histone demethylase KDM6B, protein lactylation