Clear Sky Science · en
Identification of GLDN+ odontogenic stem cells as crucial for human tooth development and regeneration
Why Growing New Teeth Matters
Root canals save many teeth, but they do so by removing the living pulp inside. That pulp houses nerves, blood vessels, and repair cells that keep a tooth healthy. Once it is gone, the tooth becomes more fragile and loses much of its natural defense system. Scientists have long hoped to regrow living pulp instead of replacing it with inert fillings, but this requires precise control over the stem cells that build dentin and pulp during development. This study uncovers a previously unrecognized group of stem cells in developing human teeth that appear to be key architects of both the hard and soft parts of the tooth.
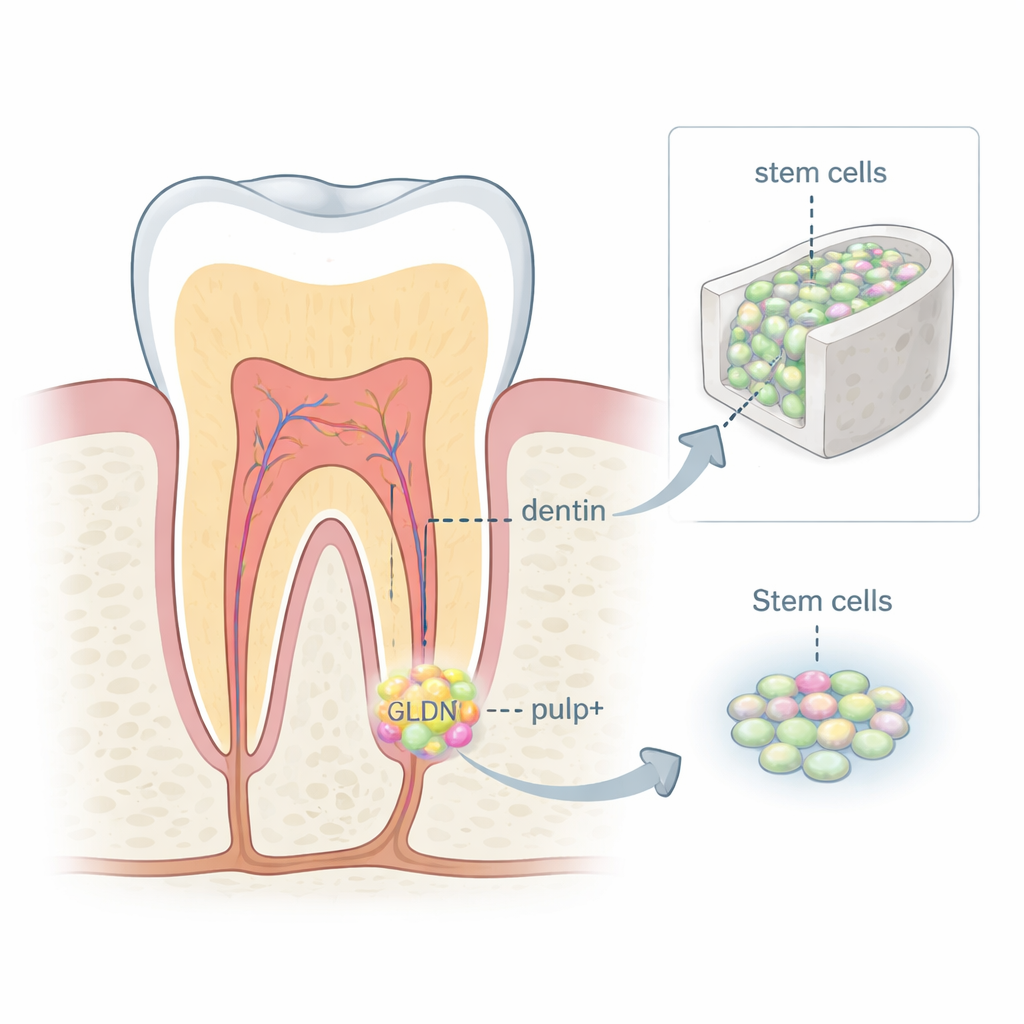
Hidden Builders Inside Young Teeth
During early tooth formation, a soft structure called the dental papilla sits beneath the future crown and root. It is packed with mesenchymal stem cells that eventually create dentin (the hard layer under enamel) and the dental pulp. Using single-cell RNA sequencing, which reads the activity of thousands of genes in individual cells, the researchers mapped all the cell types present in human dental papilla from developing wisdom teeth. They found that this tissue is far from uniform: it contains immune cells, blood vessel cells, nerve cells, and several distinct stem cell subgroups, each with its own genetic signature and likely role in tooth building.
Finding the GLDN+ Stem Cell Subset
Among the different stem cell clusters, one group stood out. These cells strongly expressed a surface protein called gliomedin (GLDN), together with other markers linked to early tooth formation. The GLDN+ cells were located mainly around the area where the growing root meets the soft papilla, close to a thin epithelial structure that guides root shaping. Developmental analysis suggested that these GLDN+ cells arise from even earlier progenitors, then move toward the crown and root, where they mature into odontoblasts (dentin-producing cells) and cells that help form the pulp matrix. Microscopy of human tooth tissues across several stages showed that GLDN+ cells first increase near the forming dentin, then gradually decline as the root canal matures, hinting that they are most active during the window when pulp and dentin are being laid down.
Stem Cells That Build and Attract Blood Vessels
To test how special these cells really are, the team isolated GLDN+ and GLDN− cells from human dental papilla using cell sorting. Both behaved as mesenchymal stem cells, but the GLDN+ cells outperformed their counterparts: they formed more colonies, proliferated faster, migrated more readily, and produced more mineral deposits under conditions that encourage hard tissue formation. They also made higher levels of key dentin-related proteins. Perhaps even more important, when the researchers collected the liquid in which GLDN+ cells had been grown and applied it to human endothelial cells (the cells that line blood vessels), those endothelial cells migrated more and formed more tube-like vascular networks. This means GLDN+ cells not only build dentin-like tissue themselves, they also send signals that help assemble the vital blood supply of the pulp.
Regrowing Pulp in a Tooth Scaffold
The strongest evidence came from an animal model designed to mimic pulp regeneration. The scientists prepared hollowed, chemically treated dentin tubes from extracted human teeth and filled them with collagen gel containing either GLDN+ cells, GLDN− cells, or no cells at all. These constructs were implanted under the skin of mice. After four weeks, the GLDN+ group showed dense, organized pulp-like tissue inside the dentin tube, with a clear layer of odontoblast-like cells lining the inner dentin surface and a richer network of blood vessels and collagen fibers than the other groups. This demonstrated that GLDN+ cells can rebuild a vascularized pulp-dentin complex in a living environment, earning them the name “GLDN+ odontogenic stem cells.”
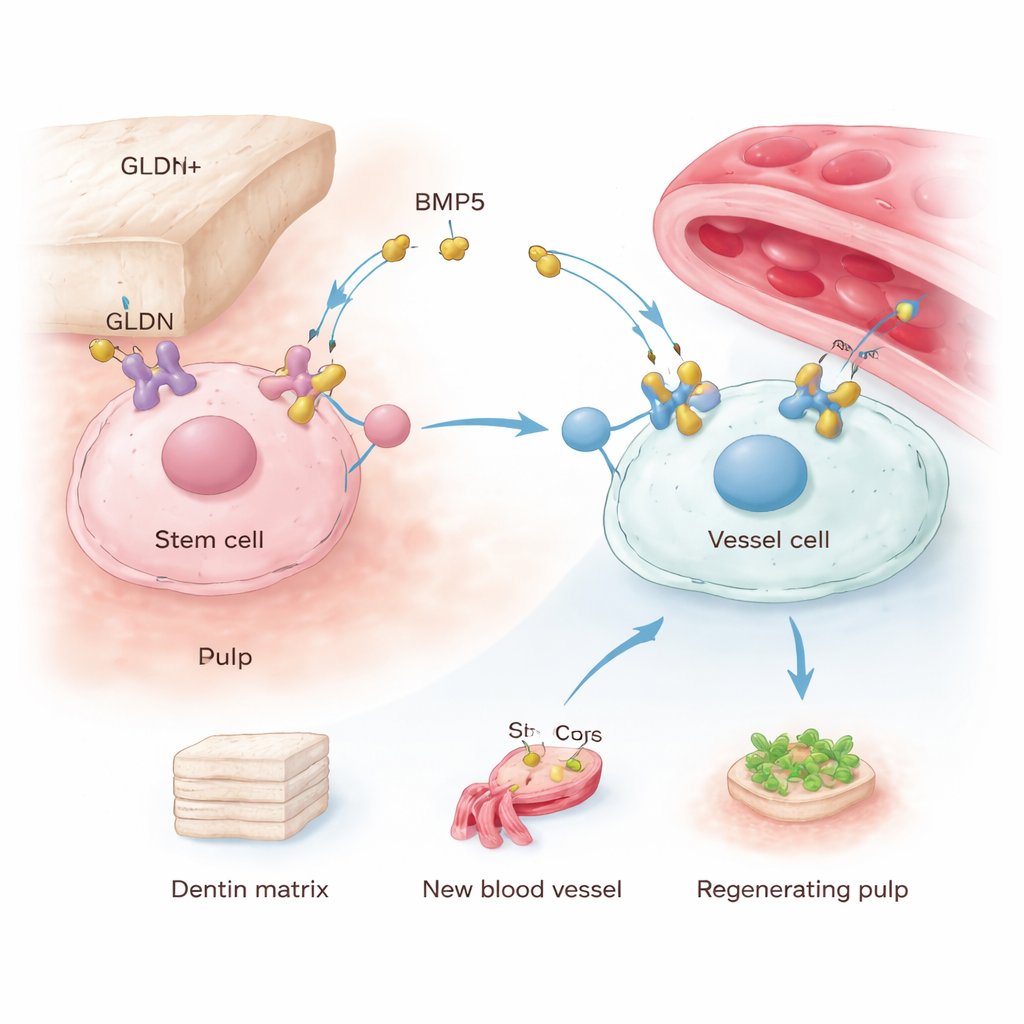
How GLDN Signals Drive Tooth Repair
Next, the researchers asked what makes these cells so powerful. By examining communication signals between GLDN+ cells and nearby blood vessel cells, they identified bone morphogenetic protein 5 (BMP5) as a key secreted factor. GLDN+ cells produced more BMP5 and showed higher activation of a downstream signaling cascade inside the cell, known through proteins called SMAD1/5/9, which is linked to bone and vessel formation. When GLDN in these cells was silenced, their growth, movement, mineralization, and ability to stimulate vessel formation all dropped, and BMP5 levels and SMAD activation fell. Directly reducing BMP5 had similar effects, whereas adding extra BMP5 to less potent cells boosted their mineralization and their support of blood vessel growth. Together, these experiments reveal a GLDN–BMP5–SMAD axis that helps GLDN+ cells maintain their identity and orchestrate both dentin production and angiogenesis.
What This Means for Future Dental Care
For non-specialists, the message is that scientists have pinpointed a highly capable subset of stem cells in developing human teeth that can build both the hard dentin shell and the living, blood-rich pulp within. These GLDN+ odontogenic stem cells use a specific signaling pathway, centered on BMP5, to renew themselves, form mineralized tissue, and attract blood vessels. In the long term, harnessing these cells—or mimicking their secreted signals—could enable treatments that rebuild living pulp in teeth damaged by decay or trauma, potentially offering an alternative to traditional root canals and opening doors to broader bone and neurovascular repair strategies.
Citation: Liao, C., Liu, J., Li, M. et al. Identification of GLDN+ odontogenic stem cells as crucial for human tooth development and regeneration. Int J Oral Sci 18, 20 (2026). https://doi.org/10.1038/s41368-025-00419-y
Keywords: dental pulp regeneration, odontogenic stem cells, GLDN, BMP5 signaling, tooth development