Clear Sky Science · en
JAK-STAT signaling: molecular mechanism and targeted treatment in dento-maxillofacial abnormalities
Why jaw growth signals matter
Our faces and teeth don’t just “grow themselves.” They are shaped by a web of chemical signals that tell bone and tooth cells when to divide, harden, or be replaced. One of the most important of these systems is called JAK–STAT signaling. When this system is disrupted, children can develop serious dento‑maxillofacial abnormalities—problems with jaw size, facial shape, tooth formation, and bite—that often require years of braces and surgery. Understanding this signaling network may open the door to medicines that guide facial growth more precisely and spare future patients from invasive treatments.
How a cell’s message line shapes the face
JAK–STAT is a direct line from signals outside a cell to the genes inside its nucleus. Hormones and immune molecules in the bloodstream dock on receptors at the cell surface. These receptors activate JAK proteins, which in turn switch on STAT proteins. Once activated, STATs move into the nucleus and turn specific genes on or off. This tightly controlled relay tells bone‑forming cells when to lay down new tissue, bone‑eating cells when to remove it, and tooth‑forming cells when to build enamel and dentin. The review explains that built‑in brakes—such as SOCS and PIAS proteins—keep the system from running too hot or too cold, helping to prevent abnormal bone density or shape. 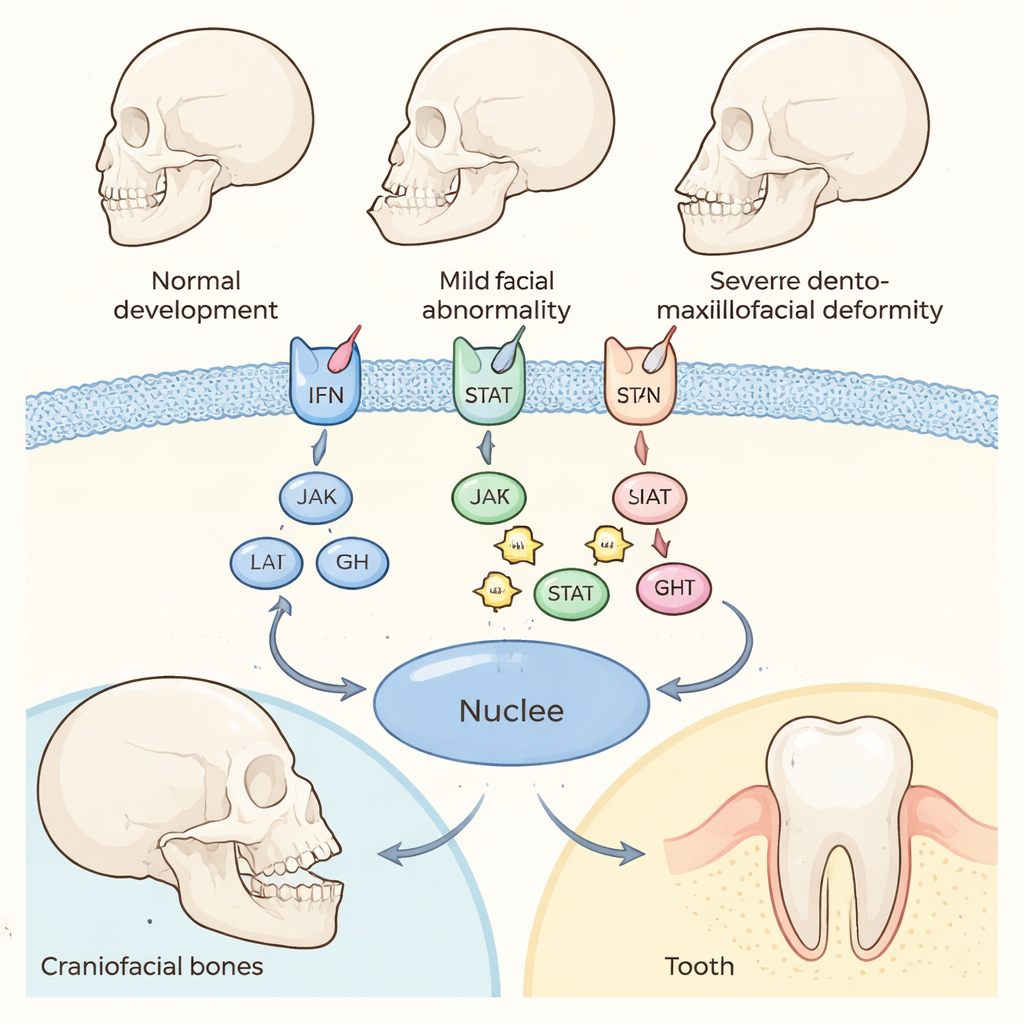
Building skull and jaws: two ways to make bone
The bones of the skull and jaws form in two main ways. In intramembranous ossification, stem cells change directly into bone‑forming osteoblasts, which build much of the skull and face. In endochondral ossification, stem cells first become cartilage, which is later replaced by bone; this process shapes the skull base and jaw joints. JAK–STAT signaling is woven into both modes. STAT3 and some JAKs push stem cells toward osteoblasts and support healthy bone density, while STAT1 and STAT6 can restrain this process. In bone‑eating osteoclasts, other STATs help fine‑tune how quickly bone is removed. The mandible is particularly complex, combining both bone‑making modes in different regions, and early work suggests JAK–STAT activity may differ from one jaw segment to another.
From tooth bud to biting surface
Tooth development is another finely staged drama that depends on JAK–STAT. Early in gestation, mouth lining cells fold inward to form the enamel organ, which wraps around a core of soft tissue that will become the dental pulp and follicle. STAT3 helps enamel‑secreting ameloblasts make key matrix proteins and later manage iron transport that contributes to enamel hardness and acid resistance. Other STATs influence the dialogue between surface epithelium and underlying mesenchyme that determines root length and tooth position. When these signals go awry in animal models, incisors and molars show thin dentin, shortened roots, and delayed or defective enamel, echoing some human dental anomalies.
When signaling goes wrong: rare genes, visible faces
Inherited defects in JAK–STAT components produce striking face and tooth changes in several rare syndromes. Dominant‑negative mutations in STAT3 cause Autosomal Dominant Hyper‑IgE Syndrome (“Job’s syndrome”), marked by prominent forehead, asymmetrical face, high‑arched palate, and retention of baby teeth because their roots do not resorb properly. STAT3 gain‑of‑function mutations, in contrast, lead to a rounded face, tall forehead, and other skeletal problems. Faulty STAT5B signaling underlies certain forms of growth hormone insensitivity, with short stature and dentofacial differences, while STAT6 overactivity can produce enamel hypoplasia, a high palate, and joint hyperflexibility. Although these conditions are rare, they reveal which growth pathways sculpt normal craniofacial form.
Turning pathway insight into new treatments
Today, most dento‑maxillofacial abnormalities are treated mechanically—with braces, anchors, and jaw surgery that take years and can be costly. The review explores a more biological future. Drugs that already target JAK–STAT for blood and joint diseases, such as baricitinib, ruxolitinib, and imatinib, alter bone resorption and formation in animal models, sometimes improving osteoporosis or modifying orthodontic tooth movement. Natural compounds, designer proteins, and small RNAs can also nudge this pathway in specific cell types, for example by shifting immune cells from a bone‑destroying to a bone‑protective mode. 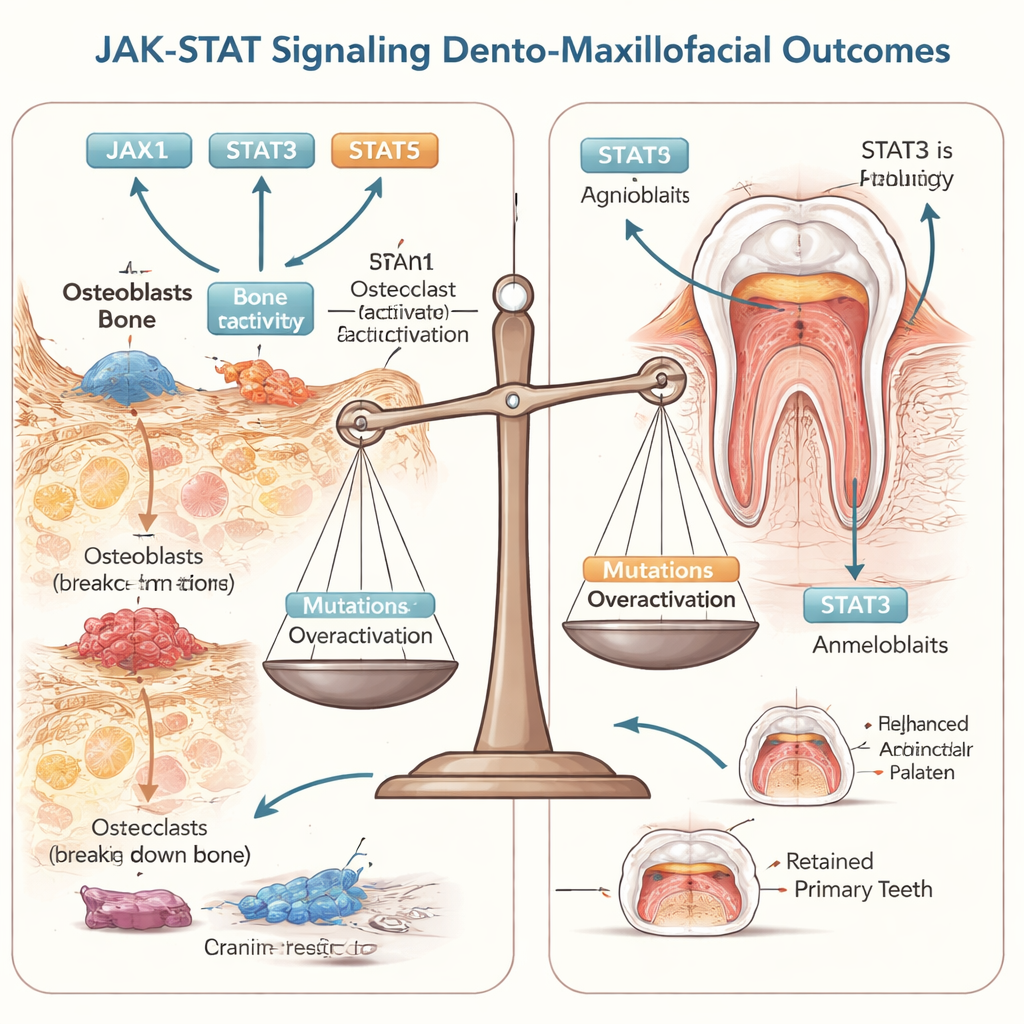
Looking ahead: toward precision growth guidance
The authors conclude that JAK–STAT signaling is a central control hub for how skull, jaw, and dental tissues grow, remodel, and respond to forces like chewing or braces. Because this same pathway also shapes immune defenses and long bones, any therapy must be carefully tuned so that adjusting facial growth does not harm the rest of the body. As scientists map which JAK–STAT switches operate in each craniofacial cell type, it may become possible to pair orthodontics with brief, localized drug treatments that guide bone and tooth cells more precisely. For patients, that could ultimately mean shorter, less invasive, and more predictable correction of complex jaw and bite problems.
Citation: Huang, Z., Cui, Y., Zhang, W. et al. JAK-STAT signaling: molecular mechanism and targeted treatment in dento-maxillofacial abnormalities. Int J Oral Sci 18, 24 (2026). https://doi.org/10.1038/s41368-025-00399-z
Keywords: JAK-STAT signaling, craniofacial development, jaw abnormalities, tooth formation, targeted bone therapy