Clear Sky Science · en
Blackening of copper pigments in wall paintings: impact of the fresco technique and the chemical composition of the pigments
When Bright Ancient Walls Turn Dark
Visitors to historic churches and villas often marvel at intense blues and greens painted centuries ago—only to find that many of these colors have dulled or turned almost black. This article explores why some of the copper-based pigments that once gave frescoes their vivid hues gradually darken, and why others stay surprisingly stable. Understanding this slow transformation helps conservators protect irreplaceable wall paintings before their colors are lost for good. 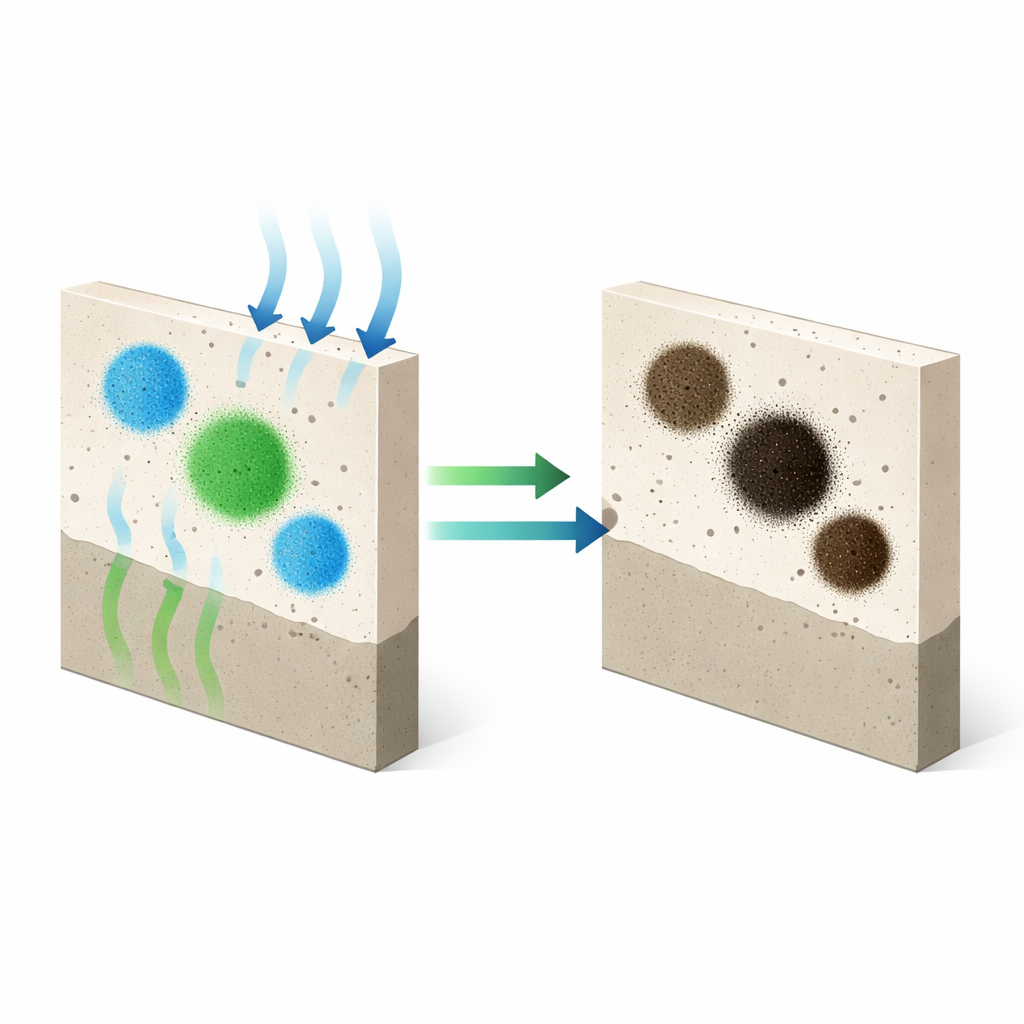
Why Copper Colors Were So Popular
For thousands of years, artists across the Mediterranean used copper minerals to create striking blues and greens on plaster walls. Natural stones like azurite (deep blue) and malachite (green), man-made versions called verditers, the famous Egyptian blue glassy pigment, and the greenish mineral chrysocolla all played starring roles. Later, a man-made copper acetate known as verdigris added brilliant, translucent greens. These pigments were often applied using the fresco technique, where color is brushed onto fresh, still-damp lime plaster so that paint and wall harden together into a single surface.
Fresh Plaster: A Tough Neighborhood for Pigments
In true fresco, pigments are mixed with water and laid over a wet lime-based mortar that is highly alkaline and full of moisture. As the plaster dries, it reacts with carbon dioxide in the air, turning lime into calcium carbonate and locking the paint in place. The authors recreated this process using eight different copper pigments applied to model wall sections, then followed the color changes over 28 days and again after a year. They also compared what they saw by eye with a battery of techniques—microscopy, X-ray methods, infrared light, and surface-sensitive X-ray photoelectron spectroscopy—to see which chemical and structural changes matched the darkening. 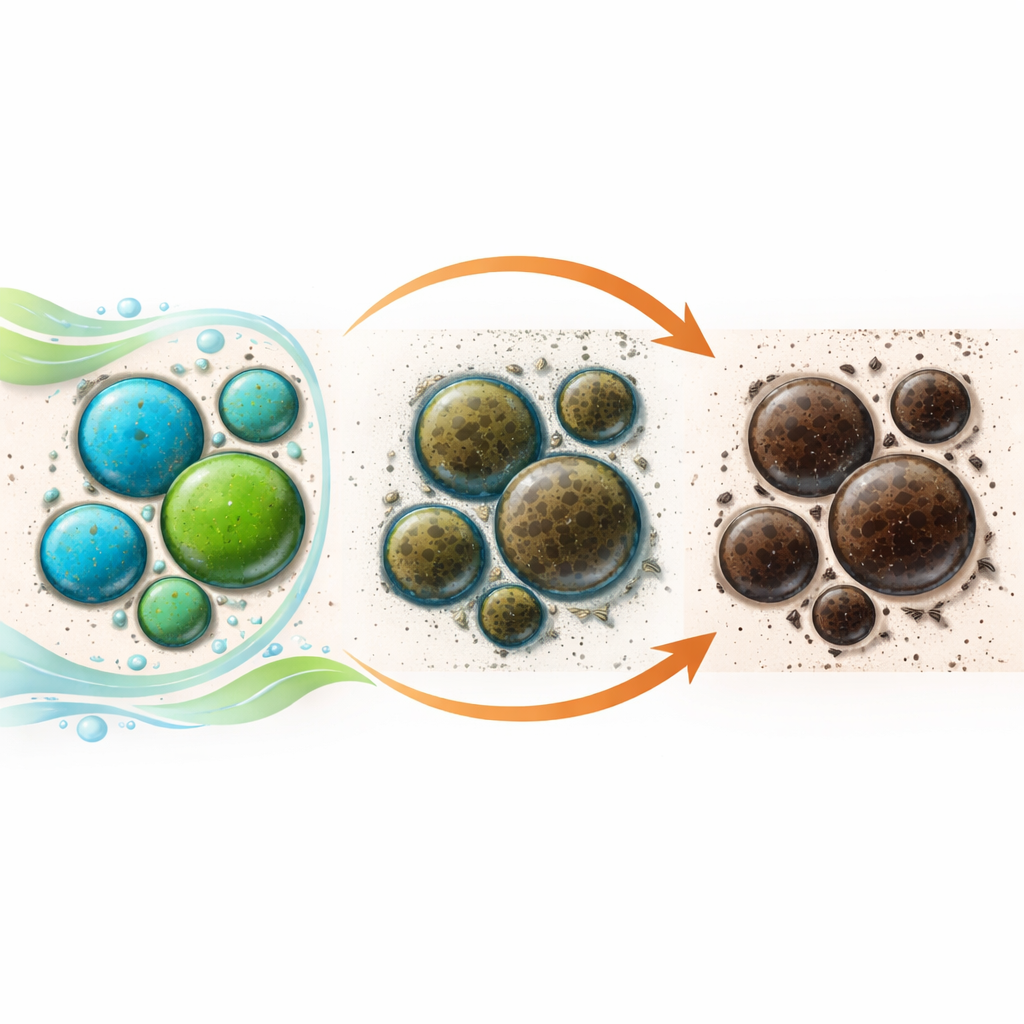
Which Pigments Darken—and How
The results showed that chemistry matters greatly. Copper carbonates (azurite and malachite, both natural and synthetic) blackened the most and showed pitted particles and “reaction halos” where pigment touched the alkaline plaster. Verdigris behaved differently: rather than forming a firm paint layer, it became powdery and patchy, with colors ranging from blue to brownish-black. In contrast, the copper silicates told a mixed story. Egyptian blue stayed remarkably stable—still blue and protected by a compact lime layer—while chrysocolla showed only slight darkening, mainly where tiny impurities rich in copper, cobalt, or iron were present. These findings suggest that not only the main pigment, but also its mineral impurities and particle size, influence how quickly and how severely colors shift.
Hidden Darkeners: Beyond the Usual Black Oxide
A long-standing explanation for blackening in such paintings is the formation of tenorite, a black copper oxide. The study did find this compound in some azurite-based tests, but not nearly often enough to explain all the darkening seen by eye. Instead, surface analyses revealed an increase in copper in lower oxidation states—forms of copper that are chemically more “reduced” than the original pigment. These reduced copper species, often poorly crystalline or nearly amorphous, can create very dark, light-absorbing surface layers that escape detection by standard crystallographic tools. The authors also saw evidence that carbon from the surrounding air—soot particles, volatile organic molecules, and other carbon-based contaminants—becomes trapped in the plaster during drying and may help drive this reduction of copper, especially under humid, alkaline conditions.
What This Means for Saving Painted Walls
In plain terms, the study shows that blackening of copper-based fresco pigments is not caused by a single, simple reaction. Instead, it arises from a combination of moisture, strong alkalinity, the exact pigment chemistry, tiny mineral impurities, and subtle changes in the copper’s electronic state. Carbonate and acetate copper pigments emerge as particularly fragile, while silicate-based pigments such as Egyptian blue are far more robust. For conservators, this means that correctly identifying the kind of copper pigment—and its origin—can guide choices about cleaning, treatments, and environmental control. Avoiding highly alkaline products and limiting exposure to pollutants and carbon-rich dust may help slow the darkening, giving historic blues and greens a better chance of surviving for future generations.
Citation: Jiménez-Desmond, D., Arizzi, A., Ricci, C. et al. Blackening of copper pigments in wall paintings: impact of the fresco technique and the chemical composition of the pigments. npj Herit. Sci. 14, 190 (2026). https://doi.org/10.1038/s40494-026-02461-3
Keywords: fresco wall paintings, copper pigments, color blackening, art conservation, Egyptian blue