Clear Sky Science · en
Regulatory mechanisms for Snail protein stability: ubiquitin–proteasome system and chaperone-mediated autophagy
Why Cancer Spread Hinges on a Fragile Protein
Cancer becomes truly life-threatening when cells break away from the original tumor and travel to distant organs. This spread, or metastasis, depends in part on a powerful "master switch" protein called Snail, which helps cells loosen their bonds and become more mobile. This article explains how our cells normally keep Snail on a short leash by rapidly breaking it down through two major waste-disposal systems. Understanding this balance offers new ideas for slowing or preventing metastasis.
From Tightly Packed Cells to Wandering Invaders
In healthy tissues, epithelial cells form orderly, brick-like layers that stay put and stick tightly to their neighbors. For a tumor cell to escape, it often goes through a makeover called epithelial–mesenchymal transition, or EMT, in which it loses its tight contacts and gains the ability to move and invade. Snail is a key driver of this makeover: when present in the cell nucleus, it turns off genes that maintain cell–cell adhesion and turns on genes that favor movement and invasion. High levels of Snail are linked to aggressive cancers and poor patient outcomes, so cells have evolved ways to keep Snail scarce and short-lived under normal conditions.
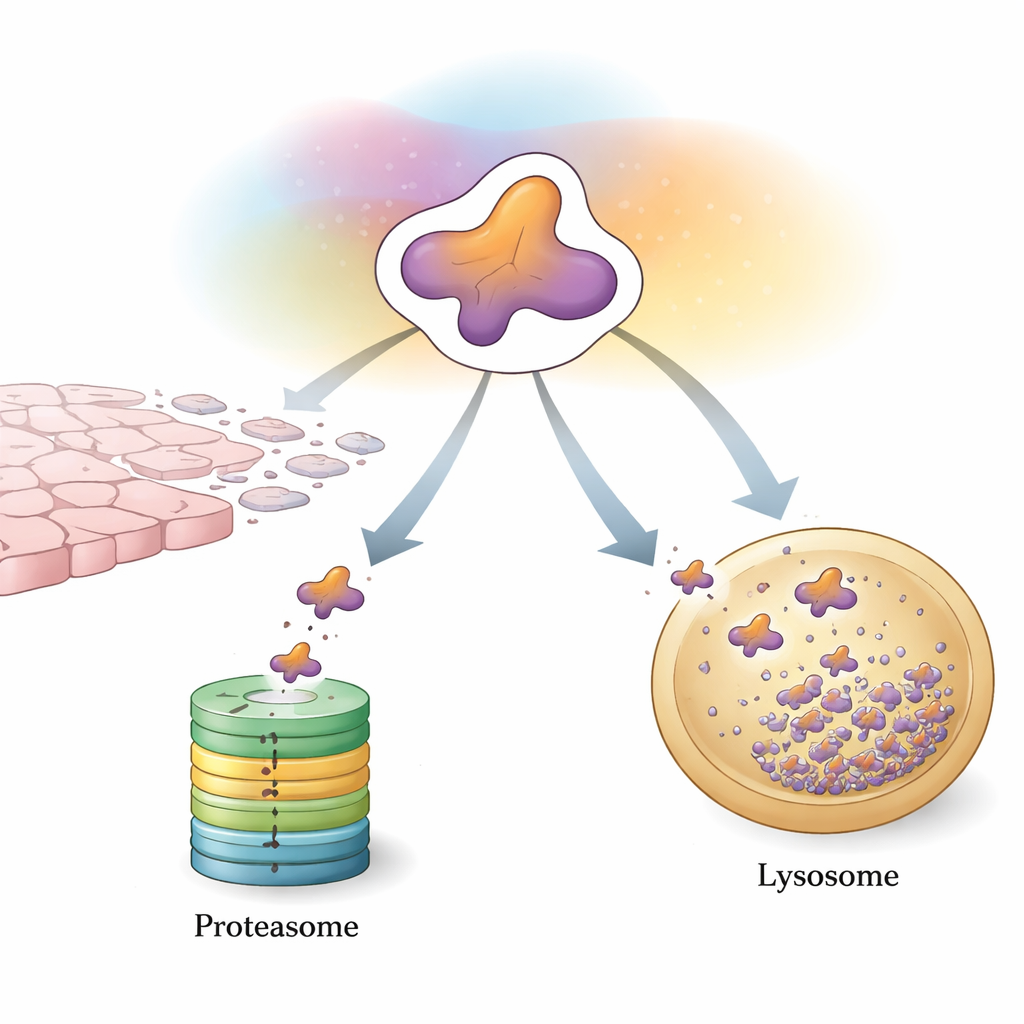
The Cell’s First Line of Control: Tag and Shred
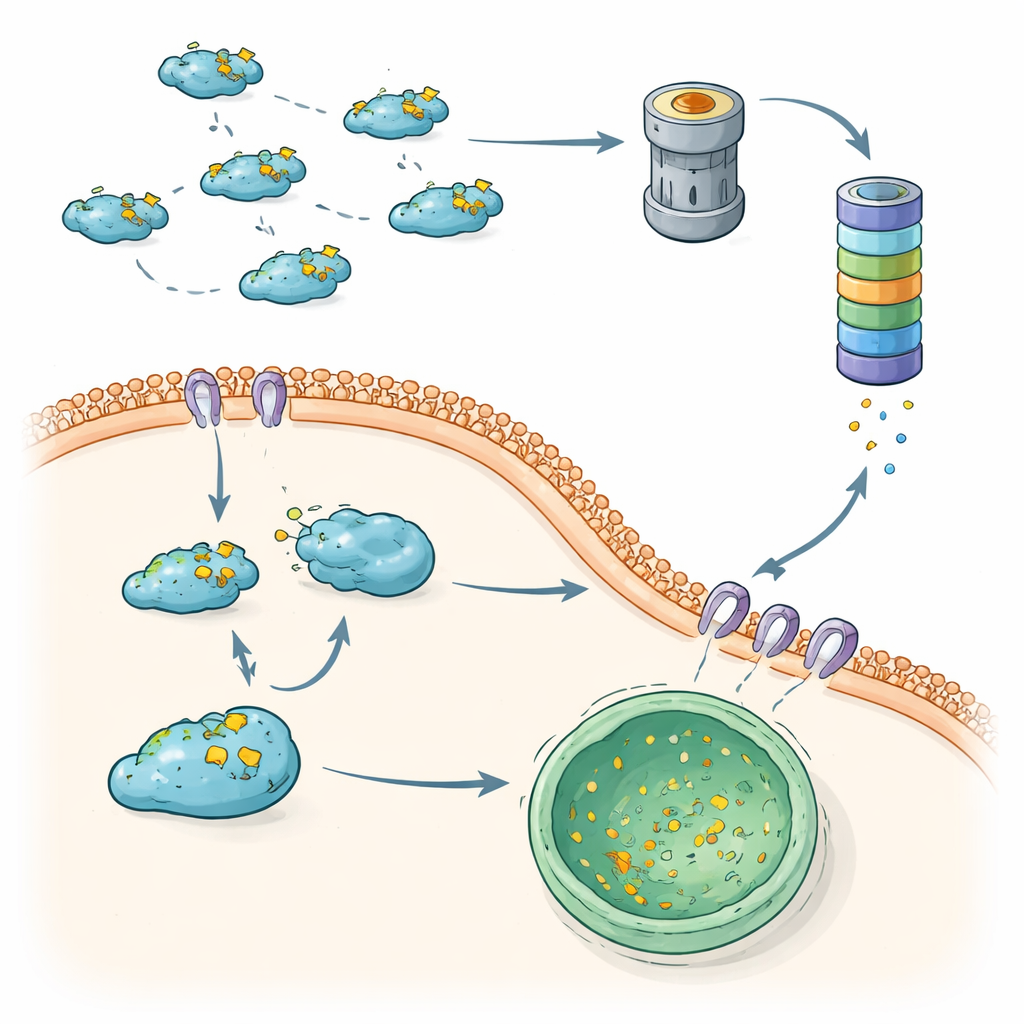
One main way cells control Snail is through the ubiquitin–proteasome system, a tag-and-shred machine for unwanted proteins. Specialized enzymes attach small “flags” made of a protein called ubiquitin onto Snail. When enough of these flags accumulate in a certain pattern, Snail is steered into the proteasome, a barrel-shaped complex that chops it into pieces. A large cast of helper proteins fine-tunes this process. Some enzymes attach degradation-promoting chains that quickly send Snail to the shredder, helping to restrain tumor growth and spread. Others add chains that instead stabilize Snail or boost its gene production indirectly, tipping the balance toward EMT and metastasis. Chemical tweaks such as phosphorylation and acetylation act like additional switches, controlling which helpers can bind Snail and whether it will be destroyed or spared.
Removing the Brakes: Enzymes That Rescue Snail
The story does not end with tagging Snail for destruction. Another group of enzymes, called deubiquitinases, can strip off the ubiquitin flags, rescuing Snail from the proteasome. Many of these enzymes are switched on or strengthened in cancers, allowing Snail to escape breakdown and persist in the nucleus. Their actions depend strongly on cellular context: in some settings they directly stabilize Snail, while in others they influence where Snail sits in the cell or how it interacts with partner proteins. Signals from cancer-associated pathways, such as growth factors and stress responses, feed into this network, deciding whether Snail will be quickly removed or allowed to drive the EMT program.
A Second Safety Net: Selective Delivery to the Cell’s Recycling Center
Beyond the proteasome, cells rely on autophagy, a collection of pathways that ferry proteins to acidic compartments called lysosomes for breakdown. This review highlights a highly selective form known as chaperone-mediated autophagy (CMA) as a second major route for controlling Snail. CMA recognizes a short sequence within Snail and, with the help of escort proteins, delivers cytoplasmic Snail into lysosomes for destruction. In less aggressive breast cancer cells, this system keeps Snail largely outside the nucleus and prone to lysosomal removal. In more aggressive, triple-negative breast cancers, Snail tends to accumulate in the nucleus and sidestep CMA, strengthening its ability to trigger EMT and metastasis.
When Quality Control Fails, Metastasis Gains Ground
Taken together, these findings reveal that cells use two complementary “quality control” axes to restrain Snail: the ubiquitin–proteasome pathway and CMA-based lysosomal degradation. When both work properly, Snail is rapidly turned over, limiting EMT and helping prevent tumor cells from spreading. When either axis is weakened—through changes in tagging enzymes, deubiquitinases, or CMA components—Snail becomes more stable, accumulates in the nucleus, and strongly promotes invasive behavior. By mapping these interconnected breakdown routes, the article outlines new therapeutic possibilities: drugs that restore or boost Snail degradation, or that steer Snail back toward CMA or proteasomal destruction, could help reapply the brakes on metastasis without completely shutting down these essential protein-recycling systems.
Citation: Kim, M., Hong, KS., Kim, T. et al. Regulatory mechanisms for Snail protein stability: ubiquitin–proteasome system and chaperone-mediated autophagy. Exp Mol Med 58, 408–415 (2026). https://doi.org/10.1038/s12276-026-01667-6
Keywords: cancer metastasis, epithelial–mesenchymal transition, Snail protein, protein degradation, autophagy