Clear Sky Science · en
Cerebellar tonic inhibition orchestrates the maturation of information processing and motor coordination
Why this brain story matters
Learning to move with grace—from a child’s first clumsy run to an adult’s agile leap—depends on tiny brain circuits that quietly fine-tune every step. This paper uncovers how a subtle form of braking in the cerebellum, a region crucial for balance and coordination, continues to mature well into adolescence. By tracing changes from single cells to whole-body motion in mice, the authors reveal how support cells called astrocytes help transform stiff, linked movements into flexible, independent limb control.
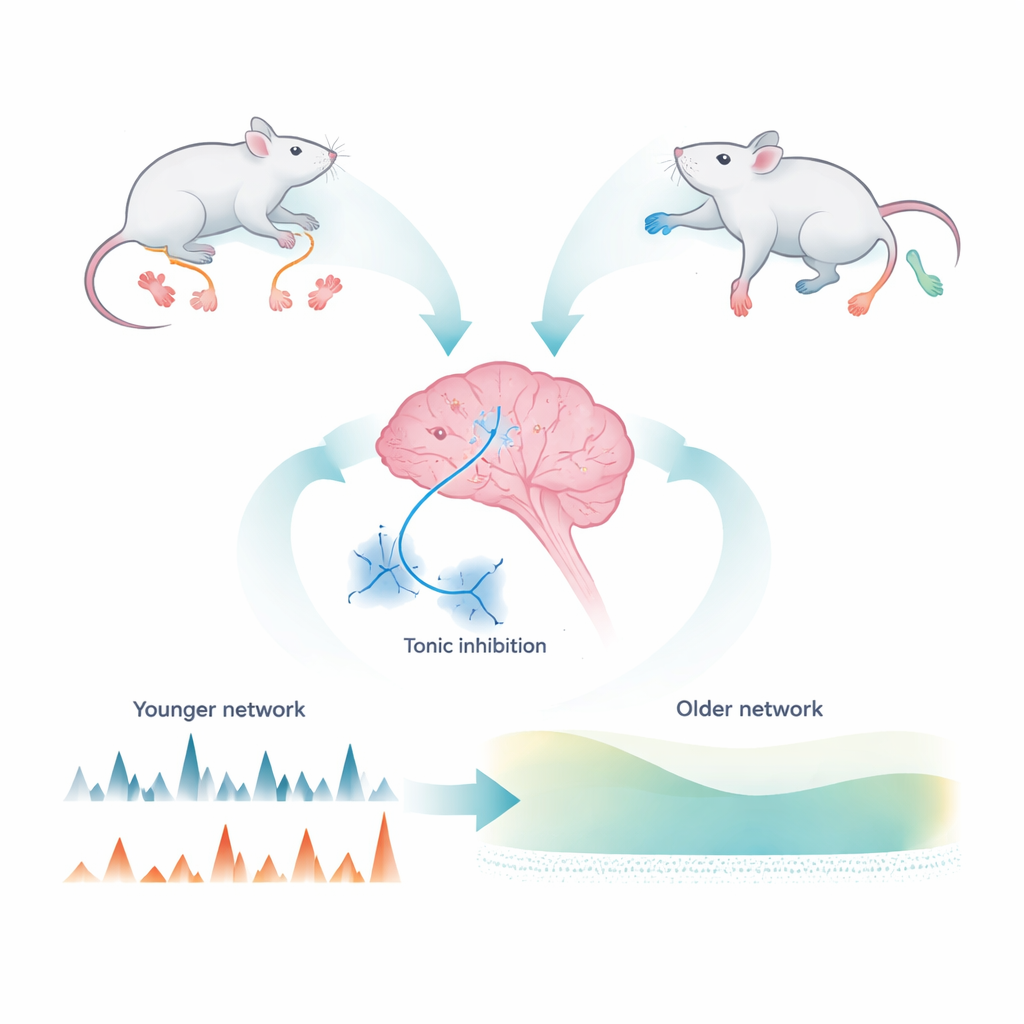
A quiet brake in the brain
Brain cells communicate not only through rapid bursts of signals, but also through a gentle, continuous background flow. In the cerebellum, this background braking—called tonic inhibition—dampens the activity of granule cells, the most numerous neurons in the brain. It is driven by the chemical messenger GABA bathing receptors that sit outside traditional synapses. Earlier work showed that this tonic brake helps sharpen the way granule cells encode incoming information, improving the clarity of motor signals. Yet, while the overall strength of this brake seemed stable with age, its precise origin was known to shift from early life to adulthood. The functional consequences of that shift remained a mystery.
From neuron-driven to glia-driven control
Using electrical recordings in thin brain slices from young (3–4 week) and adult (8–12 week) mice, the researchers teased apart where the background GABA comes from. In young mice, blocking action potentials sharply reduced the tonic current, showing that spillover from active synapses was the main source. In adults, the same block had little effect, even though the total tonic current was similar. Instead, adults showed greater uptake of GABA by transport proteins that clear the chemical from the space around cells, and a large, action-potential–independent component. By comparing normal mice with animals lacking a channel called Best1—found in astrocytes—they showed that more than half of this persistent inhibition depends on GABA released through these glial channels, especially in adulthood. Thus, over adolescence, the cerebellar brake shifts from being driven by ongoing neural chatter to being maintained by glial release and enhanced uptake.
How the changing brake reshapes network activity
Measuring all granule cells in a living animal is still technically daunting, so the team turned to large-scale computer models of the cerebellar input layer. They tuned the model using their slice data from young and adult, normal and Best1-deficient mice. Simulated input signals arriving via mossy fibers activated clusters of granule cells (“ON” clusters) while inhibitory cells called Golgi cells spread suppression to surrounding “OFF” clusters. In young-like networks, where tonic inhibition depended more on synaptic activity, this feedback loop generated strong oscillations that tightly linked different clusters, causing OFF cells to fire in patterns locked to ON activity. In adult-like networks, dominated by astrocyte-driven tonic inhibition, these internally generated oscillations weakened, and clusters became more independent. The main external inputs were still faithfully represented, but cross-talk between different input zones decreased, effectively raising the dimensionality and flexibility of the network’s information coding.
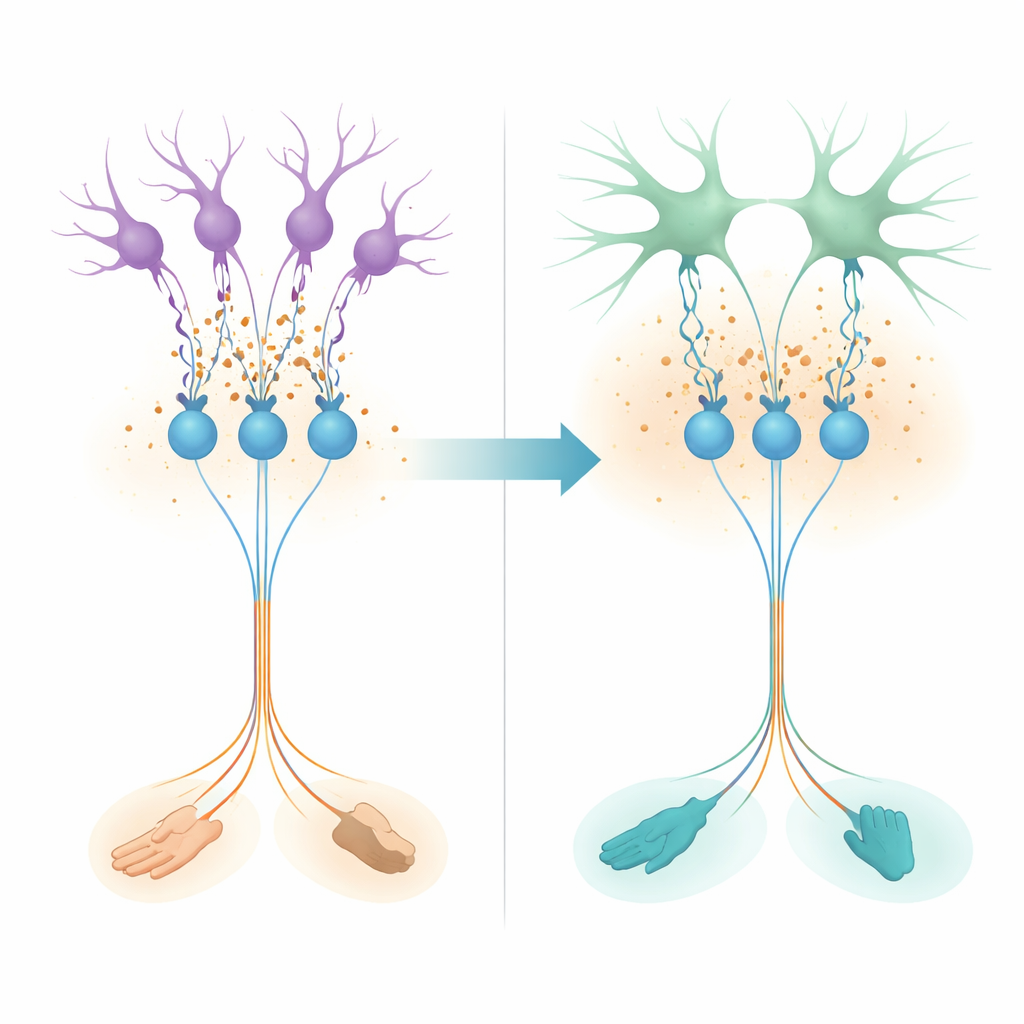
From circuitry to movement flexibility
To see whether this network-level change affects real behavior, the authors analyzed spontaneous movements in an open arena using a multi-camera 3D tracking system. They focused on how the angles of each limb changed during rapid whole-body motion and computed correlations between left and right legs. Contrary to the simple picture of perfect alternation, adult normal mice often moved both forelimbs or both hindlimbs together, especially during agile maneuvers such as hopping or sharp turns. This showed up as more frequent positive correlations between left and right limbs and a strong tendency for such in-phase movements to increase with turning speed. In adults lacking Best1, these flexible patterns were markedly reduced: their limb movements remained more stereotyped and constrained, even though standard measures of walking stability were often intact or even slightly improved.
What this means for growing into graceful motion
Taken together, the work shows that, during adolescence, the cerebellum does not simply “finish wiring up” and stop changing. Instead, the source and nature of its quiet background brake are rebalanced: astrocytes take over much of the job from synapses, while transporters and ionic conditions strengthen the overall inhibitory effect. This shift reduces internally imposed coupling between different granule cell clusters, giving separate body parts more freedom to move independently. In behavioral terms, that means fewer rigid, locked-together limb patterns and a richer repertoire of coordinated motions. The study suggests that astrocyte-driven tonic inhibition is a key, late-developing ingredient in the brain’s ability to trade off stability for flexibility, allowing mature animals—and by extension, possibly humans—to move not just reliably, but adaptively and with finesse.
Citation: Kwon, J., Kim, S., Woo, J. et al. Cerebellar tonic inhibition orchestrates the maturation of information processing and motor coordination. Exp Mol Med 58, 579–590 (2026). https://doi.org/10.1038/s12276-026-01657-8
Keywords: cerebellum, tonic inhibition, astrocytes, motor coordination, GABA