Clear Sky Science · en
Erythropoiesis–inosine metabolic axis failure underlying retinal neurodegeneration in glaucoma: novel diagnoses and therapies
Why This Matters for Vision and Health
Glaucoma is usually described as a pressure problem inside the eye, slowly stealing sight. This study argues that the story starts much earlier and far away from the eyeball: in the bone marrow and in our red blood cells. The authors show that a breakdown in how we make and fuel red blood cells starves the retina of oxygen and energy, but also uncovers a surprising potential helper molecule—inosine—that might both diagnose and treat glaucoma.
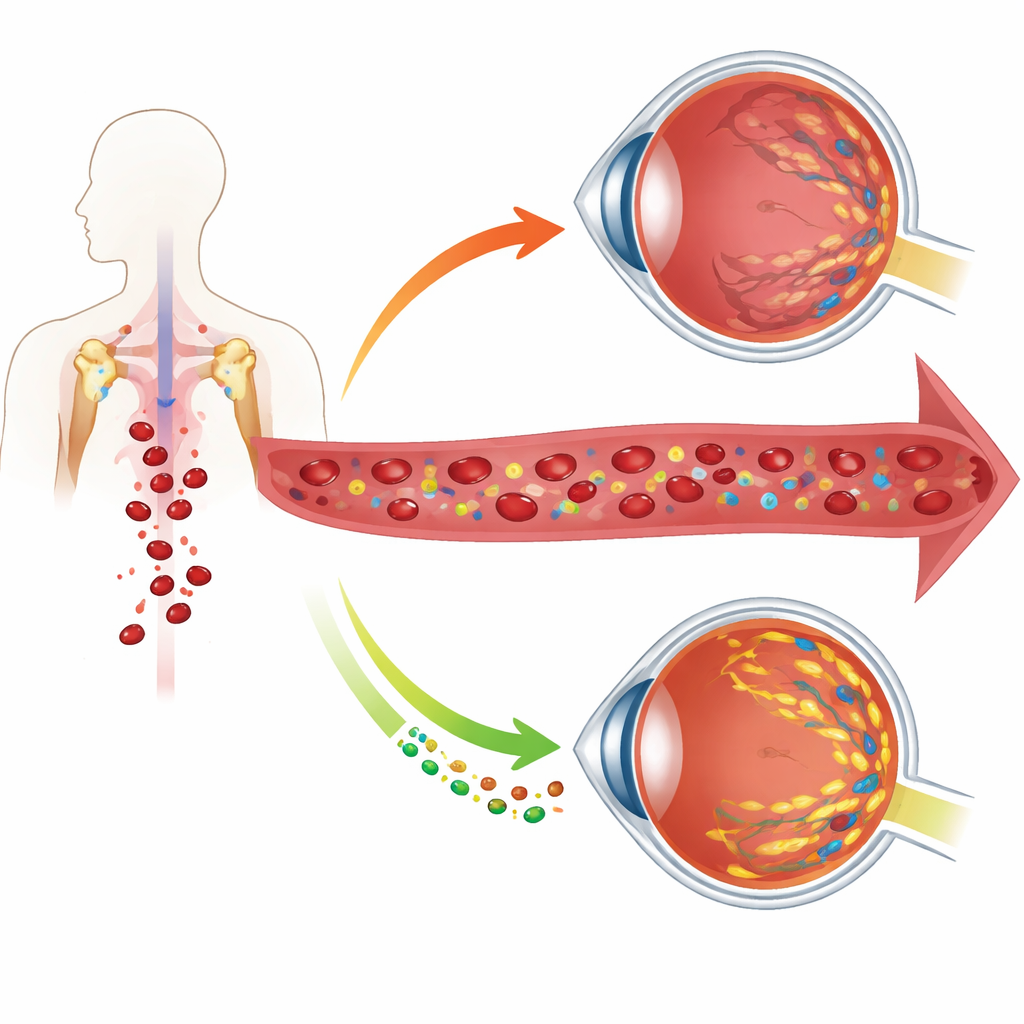
When Eye Disease Begins in the Blood
Using data from more than 127,000 people in the UK Biobank and a separate hospital cohort, the researchers found that people with glaucoma consistently have fewer red blood cells, lower hemoglobin and lower packed cell volume. These changes were seen across the two main clinical forms of glaucoma and were linked to worse eye pressure, thinner nerve layers in the retina, and poorer visual fields. In other words, glaucoma patients tend to have a mild but meaningful shortfall in the very cells that carry oxygen throughout the body, including to the eye.
Red Blood Cells Under Metabolic Stress
Red blood cells in glaucoma were not just fewer in number—they were biochemically exhausted. Detailed chemical profiling showed that their usual fuel, glucose, was not being used efficiently. The cells carried more glucose outside than inside, their main sugar transporter on the membrane was reduced, and key energy pathways were running low. At the same time, these cells had higher oxidative stress and lower stores of energy molecules such as ATP. As a stopgap, they shifted to burning another small molecule, inosine, to generate intermediates that help hemoglobin release oxygen more readily. This emergency switch, driven by an energy-sensing enzyme called AMPK, temporarily improves oxygen unloading but gradually drains systemic inosine reserves.
From Bone Marrow Failure to Retinal Damage
Because red blood cells make up most of the cells in our body, their increased appetite for inosine has consequences elsewhere. Mouse experiments in which the main inosine transporter on red cells (ENT1) was genetically removed showed that these animals developed age-related glaucoma: their eye pressure rose, their retinal ganglion cells died, and their red blood cells released oxygen poorly while generating more reactive oxygen species. Bone marrow studies revealed that lack of inosine signaling in early blood precursors impaired the production of new red cells, forcing the spleen to take on emergency blood-making duties. Together, these findings support a chain of events in which faulty erythropoiesis and maladapted red blood cells create a chronic oxygen shortage that ultimately harms the retina.
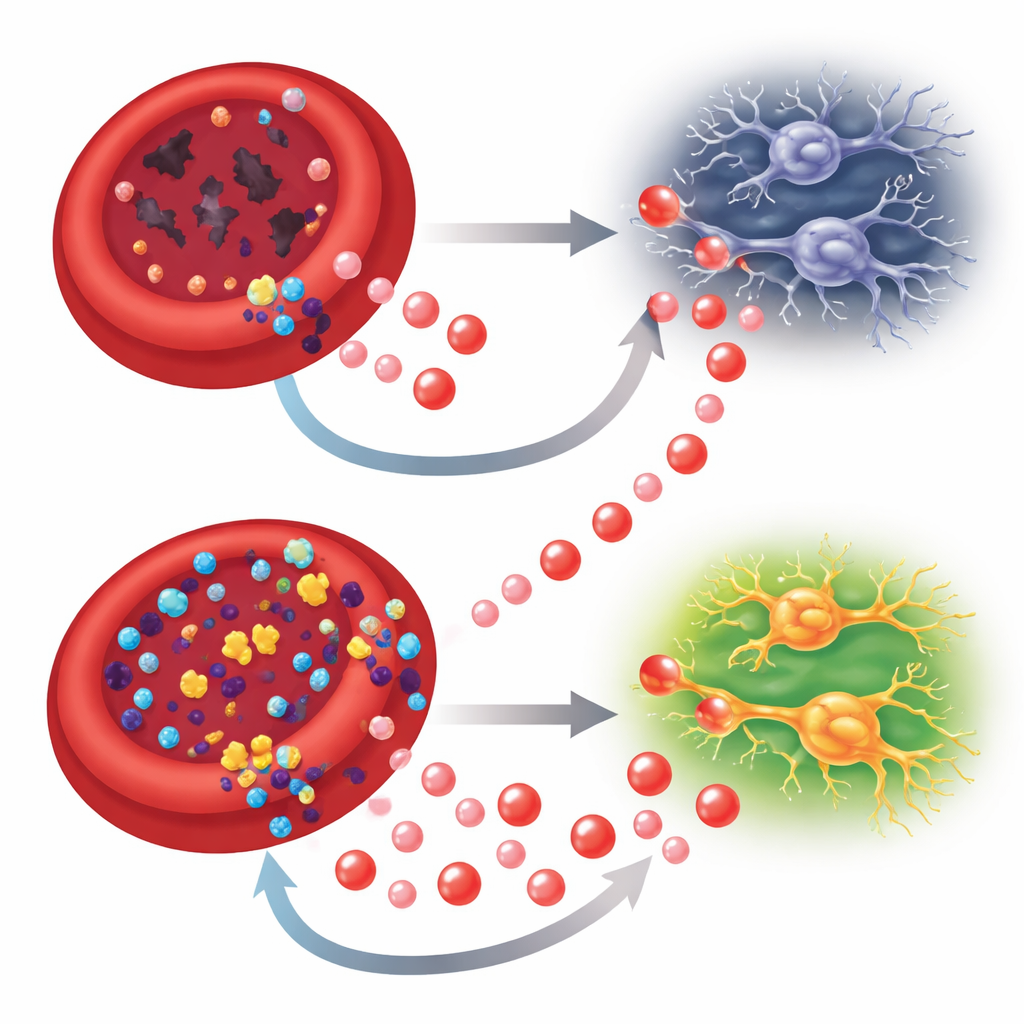
Inosine as a Metabolic Lifeline
The team then asked whether supplying extra inosine could break this vicious circle. In a mouse model of glaucoma induced by transiently raising eye pressure, daily injections of inosine improved red blood cell oxygen-releasing capacity, reduced oxidative stress, and restored red cell counts and hemoglobin levels—without shortening red cell lifespan. In the bone marrow, inosine nudged stem and progenitor cells back toward the red blood cell lineage and normalized late stages of red cell maturation. At the same time, treated mice showed better retinal oxygenation, thicker nerve layers, more surviving retinal ganglion cells, and stronger electrical and behavioral measures of vision. In cultured retinal neurons exposed to low oxygen and no glucose, inosine itself served as a backup fuel, feeding into several energy pathways, boosting ATP levels and lowering oxidative damage.
New Ways to Detect and Treat Glaucoma
By uniting large-scale human data with detailed animal and cell experiments, this work reframes glaucoma as a systemic blood–retina metabolic disorder rather than a purely local eye disease. A ten-metabolite signature in red blood cells, centered on inosine and its breakdown products, discriminated glaucoma patients from healthy subjects with accuracy comparable to standard eye tests, suggesting a future role for blood-based screening. At the same time, inosine emerges as a multi‑purpose rescue factor: it supports red blood cell production, improves their oxygen delivery, and directly fuels stressed retinal neurons. While clinical trials will be needed to test safety and efficacy in people, the study points to inosine metabolism as a promising new target for diagnosing and slowing hypoxia-driven neurodegeneration in glaucoma.
Citation: Chou, Y., Liu, W., Li, Y. et al. Erythropoiesis–inosine metabolic axis failure underlying retinal neurodegeneration in glaucoma: novel diagnoses and therapies. Exp Mol Med 58, 562–578 (2026). https://doi.org/10.1038/s12276-026-01654-x
Keywords: glaucoma, red blood cells, retinal ganglion cells, inosine metabolism, ocular hypoxia