Clear Sky Science · en
A transient receptor potential vanilloid 1-dependent corneal–trigeminal neuroinflammatory circuit promotes corneal neuropathy
Sensitive Eyes and Lingering Pain
Many people with dry, irritated eyes are surprised to learn that their worst symptoms come not from the eye surface itself, but from the nerves that sense irritation and pain. This study in mice uncovers a hidden feedback loop between the eye and the brain’s sensory hub that helps explain why eye pain can become persistent and why it sometimes affects both eyes even when only one seems dry. By revealing a specific molecular gatekeeper in this loop, the work points toward new ways to calm painful eyes without simply adding more tears.

When the Eye’s Alarm System Misfires
The clear front window of the eye, the cornea, is densely packed with nerve endings that constantly monitor wetness, temperature, and the chemical make-up of tears. In dry eye disease, tears are reduced or become too salty, and patients often develop burning, stinging, or even debilitating pain. The authors focused on a molecular sensor on corneal pain fibers called TRPV1, best known as the receptor for the chili-pepper compound capsaicin. This sensor is tuned to heat and to signals released during tissue stress. The team asked whether overactivation of TRPV1 by dryness and damage could drive not only local irritation, but also deeper changes in the nerves that connect the eye to the brain.
A Nerve–Immune Circuit Linking Eye and Brain
Using mouse models in which tear production is surgically reduced, the researchers showed that dry eye strongly activates TRPV1-bearing nerve fibers in the cornea. This, in turn, changes gene activity and immune behavior in the trigeminal ganglion, a nerve cluster that houses the cell bodies of corneal sensory neurons. In normal mice, dryness led to a surge of inflammatory and immune-related genes there and altered the state of nearby macrophages, immune cells that wrap around sensory neurons. At the same time, the dry eyes themselves showed activated immune cells at the surface, reduced nerve density, and abnormal sensitivity: the cornea became less responsive to mechanical touch and to some chemical stimuli, yet more reactive to TRPV1 stimulation, suggesting that this particular sensor had become sensitized.
How One Sore Eye Can Affect the Other
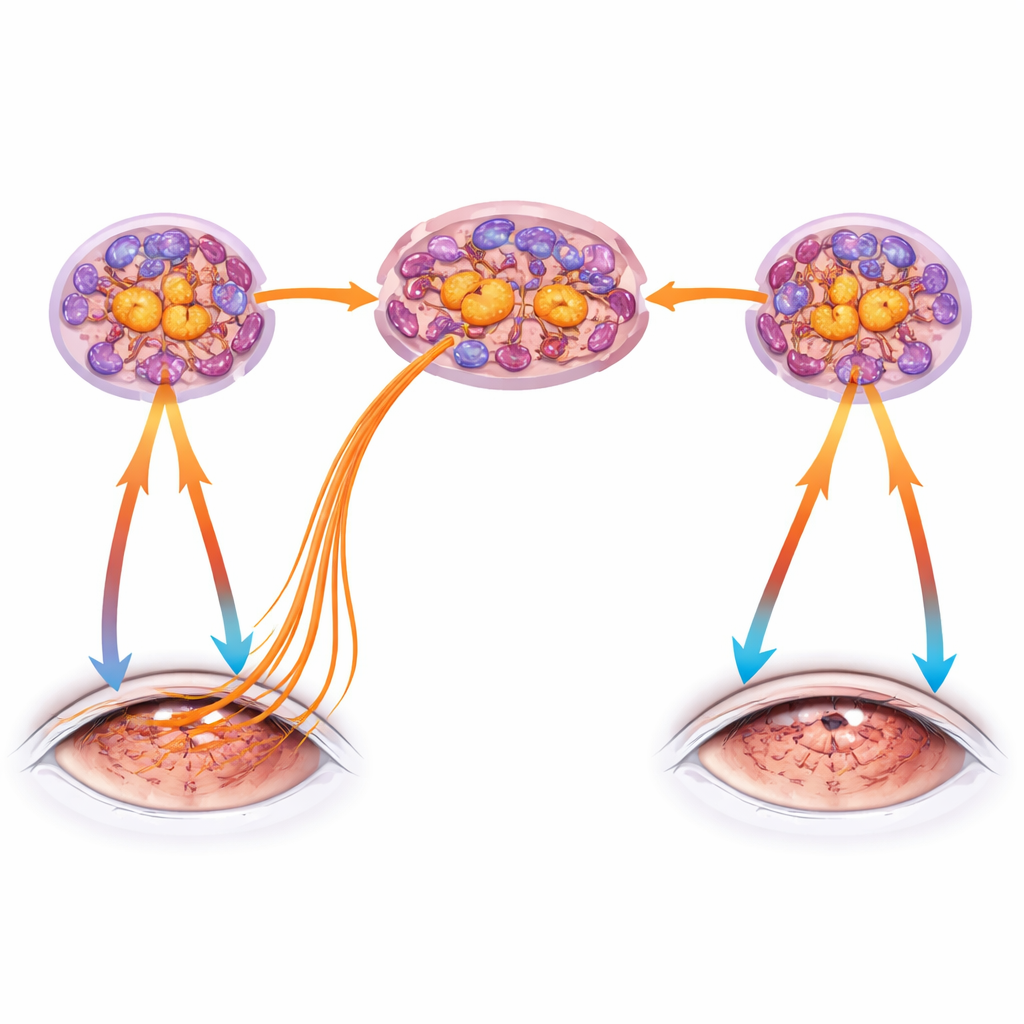
To tease apart direct effects of dryness from more remote nerve-driven signals, the team created a “one-sided” dry eye by removing a tear gland from only the right side. Strikingly, the untreated left eye remained normally moist and kept a healthy surface barrier, yet over time it developed subtle inflammation and clear signs of nerve dysfunction—thinning of deeper nerve fibers and altered sensitivity—mirroring but lagging behind the damaged side. These contralateral changes depended on TRPV1: mice genetically lacking this sensor were protected in both the dry and the untouched eye. RNA sequencing of trigeminal ganglia confirmed that unilateral dryness triggered an immune and inflammatory signature on the opposite side as well, pointing to a cornea–trigeminal–cornea circuit that can spread trouble from one eye to the other.
Proving TRPV1 Is Sufficient to Drive Damage
Dryness is complex and activates many pathways, so the authors next asked whether TRPV1 alone could set this circuit in motion. They applied capsaicin topically to the cornea of one eye, stimulating TRPV1 without drying the surface. The treated eye showed marked degeneration of corneal nerves and reduced sensitivity, even though the epithelial barrier stayed intact. Remarkably, the untreated eye again developed milder but measurable loss of nerve endings and changes in sensitivity, confirming that strong activation of TRPV1 in one cornea is enough to provoke nerve injury and functional changes in the partner eye through the shared trigeminal network. Mice lacking T and B lymphocytes only developed changes in the treated eye, suggesting that full bilateral spread requires an adaptive immune response as well as nerve activation.
A Pain-Promoting Messenger and a Possible Treatment Angle
The study also examined substance P, a neuropeptide released from stimulated pain fibers that can further inflame tissues and enhance TRPV1 activity. Blocking substance P receptors with eye drops in the non-dry eye of unilateral dry eye mice did not harm the corneal surface but significantly blunted the loss of touch sensitivity and the heightened response to TRPV1 stimulation. However, it did not fully prevent structural loss of nerve endings, underscoring that several factors cooperate to damage corneal nerves. Together, the findings outline a vicious circle: dryness and injury activate TRPV1 in corneal nerves, which send danger signals into the trigeminal ganglion, recruit and reprogram immune cells there, and then drive back pro-inflammatory nerve signals and neuropeptide release to the eye surface, worsening nerve degeneration and spreading dysfunction between eyes.
What This Means for People with Dry, Painful Eyes
For non-specialists, the key message is that dry eye disease is not just a local problem of missing tears. It involves a self-reinforcing nerve–immune loop connecting the eye surface to sensory centers in the head and back again. TRPV1 sits at the entry point of this loop, acting as an over-sensitive alarm that, once triggered, helps maintain inflammation and nerve damage even if the original dryness is modest or one-sided. Therapies that dial down TRPV1 activity on corneal nerves, or that interrupt downstream messengers such as substance P, could therefore do more than soothe a dry surface—they may break a deeper pain circuit and protect the intricate nerve network that keeps our eyes comfortable and responsive.
Citation: Pizzano, M., Vereertbrugghen, A., Martinez Gomez, M.J. et al. A transient receptor potential vanilloid 1-dependent corneal–trigeminal neuroinflammatory circuit promotes corneal neuropathy. Exp Mol Med 58, 605–621 (2026). https://doi.org/10.1038/s12276-026-01653-y
Keywords: dry eye disease, corneal nerves, TRPV1, neuroinflammation, substance P